Q: 8 g of potassium chloride is dissolved in a quart of distilled water. Calculate the pH value of the ...
A:
Q: Question 18 What is the pH of the solution given the following information? Methyl orange = yellow M...
A: Given : Methyl orange = yellow Methyl red=yellow Bromothymol blue = green
Q: Give a clear handwritten answer..make a structure formula for-(R)-4-chloro-2-ethyl-3-methyl-1pentene...
A: Structural formula is the formula or diagram which shows how the atoms of different elements are lin...
Q: Which two variables in the ideal gas law are directly proportional with volume? PV = nRT O Temperatu...
A: According to Charles law - For a definite mass of a ideal gas , Volume is directly proportional to t...
Q: Enter your answer in the provided box. For many years the extraction of gold-that is, the separation...
A:
Q: What is the concentration of an aqueous HI solution that has a pH of 2.26? 1.82 x 103 M 5.50 x 10-3 ...
A: Given that, pH of HI = 2.26 Then, [HI] = ?
Q: A solution of phosphoric acid (HsPO.) with a known concentration of 0.250 M H.PO, is itrated with a ...
A: Acid-base reaction : H3PO4 + 3 NaOH --> Na3PO4 + 3 H2O At equivalents point Ma Va / na ...
Q: Please provide the details on how: a. a Sunscreen or Sun Filter ingredient's SPF is measured? b. a...
A: (a) The sun protection factor (SPF rating) is a measure of the fraction of sunburn-producing UV ra...
Q: Consider the reaction: CuO (s) + H2 (g) = Cu (s) + H20 (g) Kp = 12.5 mm Hg at 900K %3D %3D A system ...
A:
Q: 32.0 g of the salt Ce(SO4)2. 2(NH4)2 SO4. 2H2O is dissolved in 500 mL of solution. Calculate (a) the...
A: Weight of Ce(SO4)2. 2(NH4)2 SO4. 2H2O = 32.0 g Molecular weight of Ce(SO4)2 = 632 g/mol number of m...
Q: HO CH,OH CH;ČNH Protein II CH,OH HO CH,OH OH IV Is the antigenic determinant reducing or non-reducin...
A: The reducing sugars are sugars that acts as reducing agent and reduces other species. The sugars ...
Q: Based from the nitrogen test experiment, what is the test for nitrogen?
A: Analytical chemistry is branch of chemistry in which we deal with detection of element in organic co...
Q: Silver sulfadiazine burn-treating cream creates a barrier against bacterial invasion and releases an...
A: As per the give reaction 1 mol Ag2O reacts with 2 mol of C10H10N4SO2 to give 2 mol of AgC10H9N4SO2 S...
Q: A 50 ml aqueous sample containing 0.015 M amine (Ka = 3.16 x 10-9) was adjusted to pH 9.0 and then e...
A: Liquid - liquid extraction
Q: LA balloon has a surface area of 133.2 square inches. If the gas inside is exerting a total force of...
A: In multipart questions, we solve only first three sub-questions according to the bartleby guidelines...
Q: Question 30 Which test can be used to differentiate maltose and sucrose? Benedict's Test Bial's Orci...
A: carbohydrate can be distinguish using various chemical test
Q: For the following compounds identify and draw freehand structural isomers and/ or stereoisomers wher...
A: Isomers are the compounds having same molecular formula but different structural formula or differen...
Q: Over the years, the thermite reaction has been used for welding railroad rails, in incendiary bombs,...
A: Mass of Fe = 10.1 g Atomic mass of Fe = 55.845 g/mol Number of moles of Fe = 10.1 / 55.845 = 0.1...
Q: Functional groups Compound CH CH, NH2 CH но NO 2 CHO ci -CEN
A: For the structure of amino acids, the carbon atom is surrounded by a hydrogen atom , a carboxyl grou...
Q: er is playing the r
A:
Q: (b) Using the cubic unit cells below, draw the directions and plane indicated on each figure. [101] ...
A: 1) (101 ) - Miller indices (1 infinity 1 ) - intercept Plane of Above intercept on Cube using x...
Q: Refer to the schematic diagram below, what is compound D? | Conc. H,SO, a-naphthol Conc. HS0, acetic...
A: The answer is as follows:
Q: Giorno Giovanna measured the mass of a product in order to calculate the amount of analyte present i...
A: For qualitative analysis different methods like Gravimetric method, Electroanalytical method, Spectr...
Q: What volume of oxygen gas is required to react completely with 1.99 mol of hydrochloric acid accordi...
A:
Q: 7. The ir and MS for a compound are shown below. Identify the compound. Clearly show your reasoning.
A: A question based on IR and mass spectrum that is to be accomplished.
Q: What do you expect to happen to the concentration of the sodium acetate salt formed when sodium hydr...
A: Concept - Sodium acetate, NaCH3COO, also termed as NaOAc. It is the sodium salt of acetic acid. It i...
Q: onsturct 3 jokes using functional group
A: A question based on functional group that is to be accomplished.
Q: Complete the balanced neutralization equation for the reaction below: HNO:(aq) + Ba(OH):(aq)
A: Neutralization reaction is the reaction between acid and base to form salt and water. And balance re...
Q: The empirical formula of compound R is ____________________
A: a. 32.2% of 5 g = 1.61 g 67.8% of 5 g = 3.39 g So, The sample 1.61 g Al and 3.39 g F Now, No of mole...
Q: Concentrated phosphoric acid is 85.0% by mass H3PO4. If the molarity of concentrated H3PO4 is 14.5 M...
A:
Q: In dialysis, large molecules are allowed to diffuse through a semi-permeable membrane. O True O Fals...
A: In dialysis, a chamber is used which has semi permeable membrane.
Q: Consider three molecules, H79Br and deuterated molecule D79B and H$1 Br. The lowest energy transitio...
A: The lowest energy transition occur for rotational levels.
Q: Heeded for this question. How many grams of PbBr2 will precipitate when excess ZnBr, solution is add...
A:
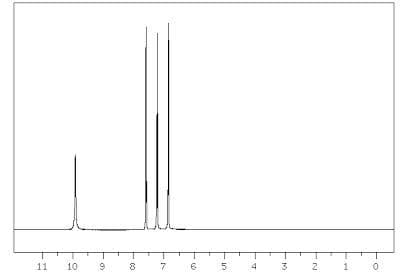
Q: compound C?
A:
Q: H H3C, „H H YCH2CH3 ČH3 A
A:
Q: (Bu Ме H Me H H H H H H Ме Me Me Me (Bu Y
A:
Q: Fill in the blank with the correct words: Energy is to break a bond and when a bond forms. required ...
A: Bond-breaking is an endothermic process, So energy is required.And when new bonds form or Bond-makin...
Q: What is the pH of a 2.3 x 10-3 M LIOH solution? O 11.4 O 2.82 O 2.64 O 11.2
A:
Q: the limiting reagent is IS
A:
Q: briefly explain how does genetic diseases or viruses connect to you?
A:
Q: 1) Give the IUPAC names for the following unsaturated hydrocarbons: CH a) b) CH, CH CH CH2– CH,
A: The IUPAC naming is done on the basis of following rules:- Longest chain is the parent chain Numbe...
Q: DDT, an insecticide harmful to fish, birds, and humans, is produced by the following reaction: 2C6 H...
A:
Q: Determine if the temperature will increase, decrease, or remain the same for each of the following s...
A: In an exothermic reaction, heat is released to the surrounding from the system. In an endothermic re...
Q: I need help with these three question please Which of the following is a Brønsted-Lowery base? A) CH...
A:
Q: O THERMOCHEMISTRY Calculating specific heat capacity 0/5 LANZCE V A chemist carefully measures the a...
A: Given, Note: 1000 mg = 1 g Mass of the sample (m) = 162.0 mg = 0.162 g Initial temperature (Ti) = 46...
Q: Which is known as B-D-glucopyranose? I. Choice I Which is the functional isomer of B-D-glucopyranose...
A:
Q: What are the components of the buffer solution when 0.20 mol H3PO4 is made to react with 0.40 mol of...
A: Here 0.2mol of H3PO4 is made to react with 0.4 mol of NaOH . We have to find phosphate components sp...
Q: Pwe Soe A student wanted to plate a nail with silver. She placed both the nail and a silver par in a...
A: Electroplating- it is a process in which one metal is coated on another metal with the flow of elect...
Q: EXERCISE NO.1 In each of the following solutions, identify the solute and solvent and their classifi...
A: Classification of mixture, compounds and elements. As per our guideline we have to answer first th...
Q: Complete the table below. Round each of your entries to 2 significant digits. You conjugate acid con...
A: In question 1 We are given either conjunction acid or base, we have to give the formula of rest one...
1
Step by step
Solved in 2 steps with 1 images


