11.24) The cyclic form of D-glucose (shown on right) is a cyclic hemiacetal. Recall that a hemíacetal is a molecule that contains both an OR group and an OH group that are bonded to the same carbon. Carbons that are bonded to both an OR group and an OH group are called hemiacetal carbons. Carbon number 1 in the cyclic form of D-glucose meets this criterium. The OH that is bonded to carbon number 1 is obvious, but the OR may not be immediately obvious to you. However, note that, beginning at carbon number 1 and moving counter-clockwise, as indicated by the red arrow in the structures shown on the right, the OR bonding pattern is seen. QUESTION: Which of the molecules shown below contain a hemiacetal carbon? a) Hemiacetal Carbon. CH₂OH H O OH H H OH OH CH₂OH b) H HO CH₂OH H CH₂OH H OH H OH H O 0 H OH H OH H O CH₂OH c) H, HO CH₂OH H OH H O H ОН H HO H H O Hemiacetal carbon. CH UM H OH H H D-glucose (cyclic form) OH CH₂OH R 0. H 0 OH H ОН OH H OH H H OH
11.24) The cyclic form of D-glucose (shown on right) is a cyclic hemiacetal. Recall that a hemíacetal is a molecule that contains both an OR group and an OH group that are bonded to the same carbon. Carbons that are bonded to both an OR group and an OH group are called hemiacetal carbons. Carbon number 1 in the cyclic form of D-glucose meets this criterium. The OH that is bonded to carbon number 1 is obvious, but the OR may not be immediately obvious to you. However, note that, beginning at carbon number 1 and moving counter-clockwise, as indicated by the red arrow in the structures shown on the right, the OR bonding pattern is seen. QUESTION: Which of the molecules shown below contain a hemiacetal carbon? a) Hemiacetal Carbon. CH₂OH H O OH H H OH OH CH₂OH b) H HO CH₂OH H CH₂OH H OH H OH H O 0 H OH H OH H O CH₂OH c) H, HO CH₂OH H OH H O H ОН H HO H H O Hemiacetal carbon. CH UM H OH H H D-glucose (cyclic form) OH CH₂OH R 0. H 0 OH H ОН OH H OH H H OH
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter17: Conjugation And Molecular Orbital (mo) Theory
Section: Chapter Questions
Problem 12E
Related questions
Question
100%
I need help with number 24 because I struggle with my homework. I don't understand. Can you explain to me, please?
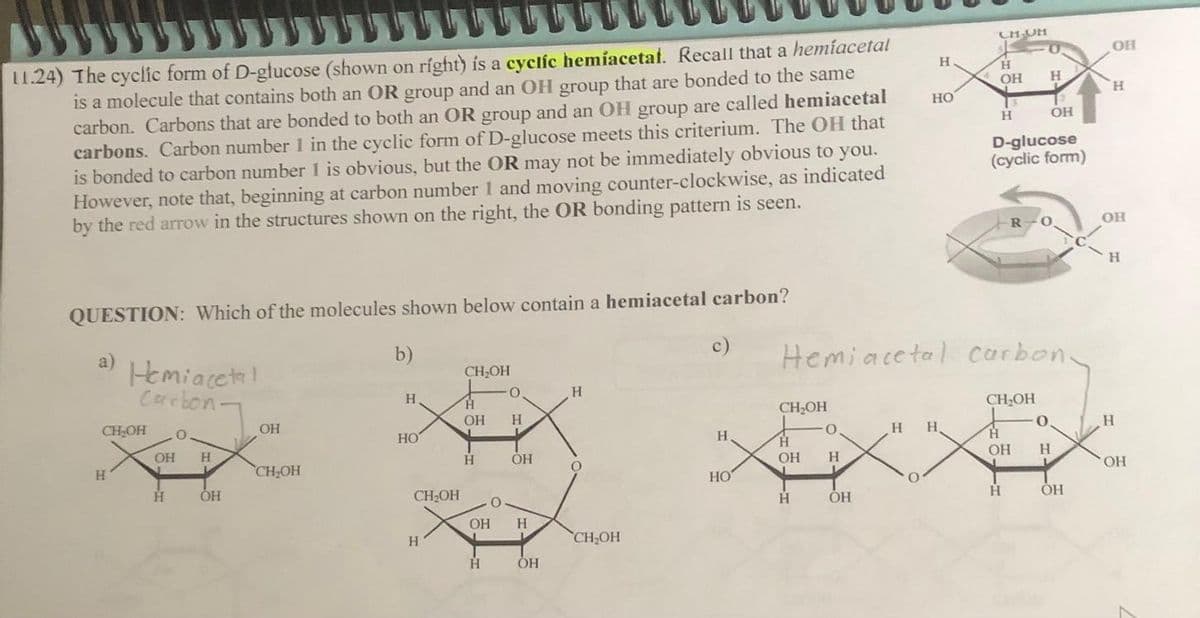
Transcribed Image Text:11.24) The cyclic form of D-glucose (shown on right) is a cyclic hemiacetal. Recall that a hemíacetal
is a molecule that contains both an OR group and an OH group that are bonded to the same
carbon. Carbons that are bonded to both an OR group and an OH group are called hemiacetal
carbons. Carbon number 1 in the cyclic form of D-glucose meets this criterium. The OH that
is bonded to carbon number 1 is obvious, but the OR may not be immediately obvious to you.
However, note that, beginning at carbon number 1 and moving counter-clockwise, as indicated
by the red arrow in the structures shown on the right, the OR bonding pattern is seen.
QUESTION: Which of the molecules shown below contain a hemiacetal carbon?
a)
Hemiacetal
Carbon-
CH₂OH
H
O
OH H
L
H
OH
OH
CH₂OH
b)
H
HO
CH₂OH
H
CH₂OH
H
OH
H
0.
H
H
OH
OH H
OH
H
O
CH₂OH
c)
H
HO
CH₂OH
H
OH
H
O
H
OH
H
HO
O
H H
CH UM
Hemiacetal carbon-
H
OH
H
D-glucose
(cyclic form)
R
H
CH₂OH
+
H
OH
H
O
-0.
OH H
OH
OH
H
OH
H
H
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning