Q: For each element determine the charge of the ion that will most likely form based on the data? What ...
A: Ionization energy: Ionization energy or ionization energy is the minimum amount of energy required t...
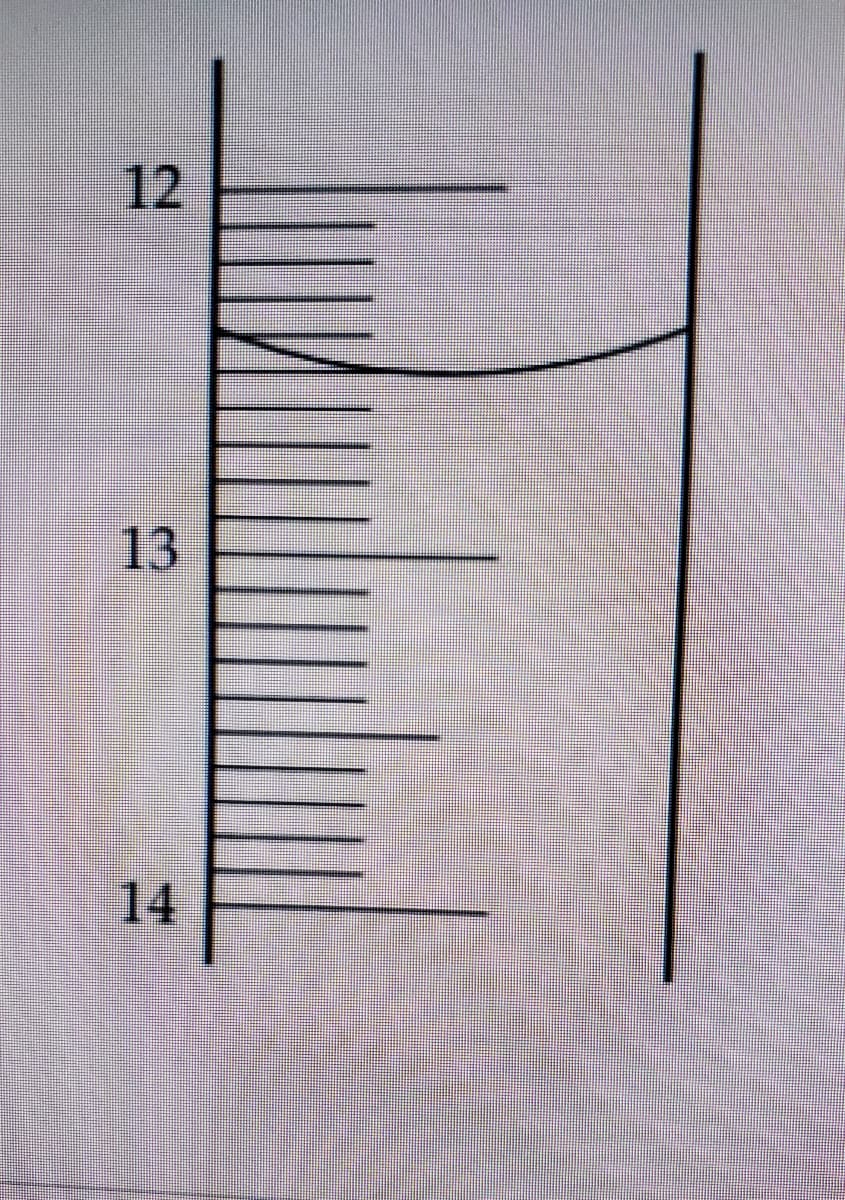
Q: In the analysis at 25 ° C of chloride using an ion selective electrode (ISE), two 100 mL solutions w...
A:
Q: Write a balanced equation for the following redox reaction in abasic solutionVO2+(aq) + NO(g) → V3+(...
A: The balanced redox reaction in basic medium is shown in the picture below:
Q: Balance the following oxidation-reduction occurring in acidic solution. MnO4–(aq) + Co2+(aq)→ Mn2+(a...
A: The unbalanced chemical equation is, MnO4–(aq) + Co2+(aq)→ Mn2+(aq) + Co3+(aq)
Q: References Use the References to access important values if needed for this question. A sample of ca...
A: Given, initial Temperature = 247°C Volume = 644 mL Final Volume = 546 mL Final temperature = ?
Q: 10) For the reaction between isopropyl 1-propyl (or 'n-propyl') ether and HBr, which type of reactio...
A:
Q: 12.Someone hands you a bottle of aluminum sulfate and asks you the following questions. a) What is ...
A: Aluminum sulfate is a compound with a molecular mass of 342.15 gmol It contains 2Al+3 and 3 SO42-
Q: Chemistry Question
A:
Q: 12. Draw the Lewis dot symbol for boron, B, how many unpaired electrons are there? A. 1 B. 2 C...
A: Lewis structure represents the bonds through electron dots.
Q: A container contains 5.0 L of a gas at 1520 mmHg. What will be its volume if the pressure increases...
A: According to Boyle's law, pressure is inversely proportional to volume. Mathematically it can be wri...
Q: Please answer my question fast and be carfulle
A: Carbonyl compounds and imide generally undergo a process called tautomerization. Tautomerization is ...
Q: I need number 11 please
A: According to Graham law of diffusion, The rate of diffusion of a molecule is inversely related with ...
Q: Osmotic pressure depends on all except which of the following? A. Atmospheric pressure B. The mo...
A: "Osmotic pressure is defined as is the minimum pressure which is needed to be applied to a solution ...
Q: Draw geometrically clear picture of the most stable enol form of the compound shown below, and point...
A:
Q: 1). Fill in the missing intermediates and product in the following mult-step synthesis:
A: 1) Toluene reacts with conc. H2SO4 is sulphonation reaction 2) second reaction is chlorination 3) ...
Q: what is the equation for relating moles to volume? In other words, what is Avogadro’s law?
A: Avogadro's law is an empirical relation of volume of gases with their number of moles.
Q: Which compound will undergo decarboxylation (-CO2) most easily when heated? (a) CO2H (b) НО-С CO2H (...
A: Ans
Q: A cobalt block was heated in a calorimeter to increase its temperature by 35.5 °C. The energy produc...
A:
Q: Use the cell notation to answer the question: Pt(s) | Sn2+(aq), Sn4+(aq) || Ag+(aq) | Ag(s) What ele...
A: We will identify the cathode
Q: Is a carbon atom microscopic or macroscopic?
A: Ans
Q: The molecular orbital energy diagram for F2 is shown below. Based on this diagram, what is the bond ...
A: The bond order of the molecules measures the number of bonds present between the atoms. The atoms ma...
Q: 20. How many domains are there around the central atom in H2O? A. 1 B. 2 C. 4 D. 3
A: To tell the number of domains around the central atom.
Q: 10. Can you help me solve this
A: Molar mass of a compound is equal to the sum of atomic masses of all the elements in that compound. ...
Q: Write an equation for each of the following reactions. Use molecular and structural formulas and cla...
A: Types of reactions depend reactants and reagent.
Q: Pinacol rearrangement reaction • Propose a mechanism for each of the following reactions а. H* но OH...
A:
Q: uces 18.15 grams of Fe(MW= nol) when 50.00g of Fe203 (MV nol) reacts with 10.00g of Al (M nol) in re...
A: Fe2O3 + 2Al -----> Al2O3 + 2Fe A) 1mole i.e. 159.69g Fe2O3 gives Fe = 2× 55.485g 50g Fe2O3 gives ...
Q: A 92.0 gram gold bar was heated in an experiment and 3,150 J of energy was transferred to the bar. W...
A: Given, mass of gold bar = 92.0 g Specific heat, c = 0.126 J/g°C Q = 3.150 J
Q: What is the entropy change when silver chloride form? The net ionic equation is Ag+ (aq) + C...
A:
Q: 11. What is the bond angle around the central atom that is trigonal planar? A. 180 degrees B. 12...
A: In a trigonal planar geometry, a central atom is bonded to three groups and each bond angle is equal...
Q: The hydrogenation reaction of the following hydrocarbon in the presence of mickle produces? pls che...
A: Given Product = to be determined
Q: Organic compounds are always highly water soluble. True False
A: Ans
Q: What is the concentration of NaOH(M)? Thank you!
A:
Q: 24) What is the correct order of reagents to achieve the following synthesis? A) i. Br2, FeBr3; ii. ...
A: Ans
Q: Please answer my question fast and be carfulle
A: When phenol is alone in the tube the concentrat of OH- ion is constant. When HCl is added to it, the...
Q: For each chemical reaction listed in the first column of the table below, predict the sign (positive...
A:
Q: A 2.98 mol of a gas occupies a volume of 3.92L. What is the volume if 1.19mol of the gas is released...
A: We will use gas law V1,n1,n2 is given
Q: AI III 皿 I ON III () II ) ON excess HNO,/H,SO4 21) What is the major product of the following reacti...
A: Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitr...
Q: Butyric acid (HC4H7O2) is responsible for the odor in rancid butter. A solution of 0.25 M butyric ac...
A:
Q: The standard reaction free energy AG°=-1181. kJ for this reaction: 4 H2O2(1) + PbS(s)→P6SO4(s) + 4 H...
A: Given reaction : 4 H2O2 (l) + PbS (s) → PbSO4 (s) + 4H2O (l) ∆G° = -1181 kJ ...
Q: Which of the following statements is TRUE? A. Dynamic equilibrium occurs when the rate of the forw...
A: A. Dynamic equilibrium occurs when the rate of the forward reaction equals the rate of the reverse r...
Q: Plutonium-238, an α emitter and a compact heat source, has a half-life of 86 years. Coupled with a P...
A: We wil use first order kinetics formula to get the answer
Q: Write an equation for each of the following reactions. Use molecular and structural formulas and cla...
A: The equation for the above reactions are given in attached image.
Q: For the reaction 2 CH4 (g) → C2H6 (g) + H2 (g)at 3547 K, Kc = 0.0235.a) If the initial concentration...
A: Kc is the equilibrium constant which is equal to the of equilibrium concentrations of product divide...
Q: 7. For the molecule SF4, what is its hybridization? A. sp3 B. sp3d2 C. sp3d D. sp2
A:
Q: Determine the color of light emitted from an electron in a hydrogen atom that falls from n = 6 to n ...
A:
Q: The caffeine molecule is shown below. Indicate the electron geometry AND the molecular shape around ...
A: To determine electron geometry and molecular shape of the given compound
Q: 100.0 mL of a 0.500 M solution of KBr is diluted to 500.0 mL. What is the new concentration of the s...
A: Consider the given information is as follows; Initial concentration M1 = 0.500 M Initial volume V1 =...
Q: , she fills a reaction vessel with some mixture of the reactants and products at a constant temperat...
A: The following formula is used: ΔG=ΔH - T ΔS T = 83°C = 273+83 = 356K
Q: Chemistry Question
A: This is an electrophilic aromatic substitution reaction. In this reaction an electrophile substitute...
Q: The simplest alkyne is used in ripening fruits. True False
A: Alkynes are organic compounds containing carbon and hydrogen atoms and triple bond. The simplest al...
Step by step
Solved in 2 steps


