Q: 3500 3000 2500 2000 1500 1000 500 Wavenumbers (cm-1)
A: IR spectrum is used to analyze functional groups present in the compound.
Q: 4.8 0 0.8 0.6 0.4 0.2 20 4000 40 3000 INFRARED SPECTRUM 2000 Wavenumber (cm-1) 42 MASS SPECTRUM 60…
A: From the above given data,we can assume that the compound is bromoethene ( CH2=CHBr ).
Q: What are the functional groups with wave number of the IR Spectrum below . The unknown is C8H8O2
A: From given formula the DBE value is calculated.
Q: Using IR Spectroscopy to Determine the Types of Bonds in a Compound What types of bonds are…
A: For compound A, two important picks, above the 1500 cm-1, are observed, ~3000 cm-1 and ~1700 cm-1…
Q: Label the IR peaks draw the structure of the compoundand give reasoning
A:
Q: FTIR Common Name: BP: IUPAC Name: MP: MICRONS 100 foo 90 .05 -0.1 70 60 50 40 30 20 10 -20 4500 4400…
A:
Q: 100 80 - 60 40 20 10 15 20 25 30 35 40 45 50 55 60 m/z Absorbance / % 50- 100- 4000 3000 2500 2000…
A: Mass spectra gives an information about the molecular mass and possible fragments of unknown. IR…
Q: INFRARED SPECTRUM 0.8F 0.4 2000 1000 3000 Wavenumber (cm-1) Transmitance
A: IR spectrum of a compound provides detailed explanation about the functional groups present in it.…
Q: Transmittance (%) 100% 75% 50% 25% 0% 4000 3500 3000 مل ОН 2500 2000 Spectrum 3B 1750 سمه 1500…
A: The bonds of molecules absorb the infrared radiation and vibrate. IR spectroscopy detects the…
Q: INFRARED SPECTRUM 0.8 0.6 0.4 2000 1000 3000 Wavenumber (cm-1) Relative Transmittance
A:
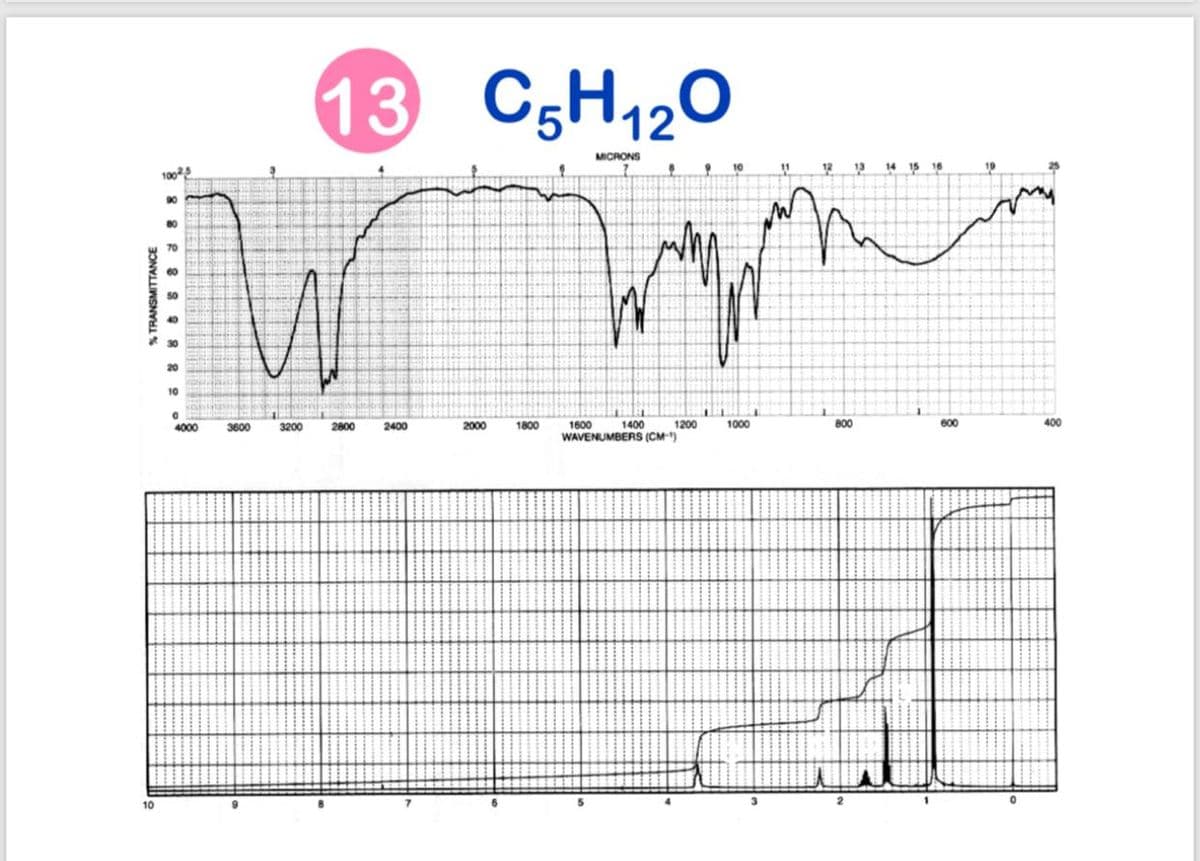
Q: 5) C3H120 13C NMR IR ppm 100 199.61 159.85 126.86 80 36.11 34.40 60 32.83 27.71 %T 40 20 4000 3000…
A:
Q: 2000 1500 1000 WAVENUMBERS PPM Peaks: 1.23 ppm (singlet, 6H), 2.24 (singlet, 3H), 4.62 (singlet, 1H)…
A: All the details are explained in handwritten solution.
Q: Wavelengh m) 10 12 13 14 15 16 26 27 28 29 ! 300 3600 3400 3200 3000 2800 2600 2400 23200 2000 1S00…
A: Spectra 1 : 2800 cm-1 and 2700 cm-1 aldehyde C-H stretch. 1730 cm-1 carbonyl stretch. Therefore the…
Q: 100 80 60 40 (a) 20 CH;CH,CH,C-NH, 4000 3500 3000 2500 2000 1800 1600 1400 1200 1000 800 600…
A:
Q: 100 43 71 50 27 114 58 20 40 60 80 100 120 mlz D00 3800 3600 3400 3200 3000 2800 2600 2400 2200 2000…
A: Mass spectrometry is an important tool for the determination of the molecular mass of the compound…
Q: wavelength, micrometers 2.6 2.8 3 3.5 4 4.5 5 5.5 6 7 8 9 10 11 12 13 14 1516 100 80 60 40 3800 3400…
A: The given spectrum is corresponding to cyclohexane (option A).
Q: 1000 500 250 100 50 Ò CPS 400 300 200 100 1H NMR 60 MHz 200 Integral = 3 80 40 CaH;O2Br Integral = 1…
A:
Q: Identify the following compounds from its molecular formula and its IR and 1H NMR spectra:
A:
Q: Which one of the following five compounds produced the IR spectrum shown below?
A: The predictions from IR spectrum are as follows: Alcohol/Phenol O-H Stretch at 3550-3200 (broad, s)…
Q: What types of bonds are responsible for the absorptions above 1500 cm−1 in the IR spectra for…
A: a) The given IR spectra for compound A shows three absorptions above 1500 cm-1. The absorption at…
Q: 2.2 You know that the wavenumbers of the rotational transition J = 1← 0 for 1H35Cl and 2H35Cl are…
A: Given, wavenumbers of the rotational transition J = 1← 0 for 1H35Cl and 2H35Cl are 20.8784 and…
Q: BRUKER 50 54 2221 08 3500 3000 2500 2000 1500 1000 500 Wavenumber cm-1 1342. 82 VV 1670.50 3082.42…
A:
Q: Two products, A and B, are obtained from the reaction of 1-bromobutane with NH3. Compound A reacts…
A: One equivalent of 1-bromobutane reacts with NH3 to give compound A. The reaction is shown below.…
Q: transition "3d -> 2p" can be allowed? 3d : n=3 , l=2 , ml = +1, +2, 0, -1, -2 2p : n=2, l=1, ml =…
A: n= principal quantum number tell size of shell l= azimuthal quantum number tells no of subshell m=…
Q: D. Spectrum [1] 100 1650 cm 50 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm) %…
A: Note: according to our guidelines we can only provide question 1 answer.
Q: 70- H3CO. NH2 Cr H3Co C,H19CIN,O, Mol. Wt. 350.79676 3800 3600 3400 3200 3000 2800 2600 2400 2200…
A: IR Spectrum helps to identify different functional groups. Specific functional group give particular…
Q: The rotational spectral lines of 12C160 appear at higher frequencies than the rotational spectral…
A: Rotational spectroscopy deal with absorption and emission of electromagnetic radiation in microwave…
Q: 100 100- %T I %T II 50 50- 4000 3000 2000 1500 1000 500 4000 3000 2000 1500 1000 500 Wavenumbers…
A: IR mainly used to determine functional groups that are present in molecule. For alkene c=c…
Q: Spectrum 28 100% 75%- 50%- 25% 0% 3500 2500 1750 1250 750 4000 3000 2000 1500 1000 50 Wavenumber…
A: Concept: Characteristics IR bond for Corbonyl: intensed bond near1715 cm-1 characteristic IR bond…
Q: UNKNOWN A ON Agel plates 52 50 48 46 44 42 40- 38- 36 34- 32 30- 28 26 24 22 20- 18 16- 14 12 10-…
A: The IR spectra shows the different type of stretching and bending vibration frequency as peaks. From…
Q: Five compounds are shown of the IR spectra below. Indicate which of the five compounds is…
A: The peak at 1700-1680 cm-1 indicates the presence of C=O group, the peak at 1600 - 1450 cm-1…
Q: INFRARED SPECTRUM 0.8 0.6 oo OH 0.4 0.2 3000 2000 1000 Wavenumber (cm-1)
A:
Q: 3. Fill in the following for the correspondence between % transmittance and absorbance: l0.0 %T…
A:
Q: What types of bonds are responsible for the absorptions above 1500 cm−1 in the IR spectra for…
A: a) The given IR spectra for compound A shows three absorptions above 1500 cm-1. The absorption at…
Q: Which one of the following five compounds produced the IR spectrum shown below?
A: Band at 3400 cm -1 is due to hydroxy group of alcohol, since for carboxylic acid hydroxy group will…
Q: %Transmitance 3345.22 1665.76 1597.577.05 1493.7248.84 1202.89 9 T001.62 1068.b08.08 965.16 019.23…
A: IR peak corresponds to different functional groups present in a molecule.
Q: 100 100- II se 4000 3000 2000 1500 1000 500 4000 3000 2000 1500 1000 s00 Wavenumbers icm-11…
A: d) o-xylene shows the IR spectrum VI.
Q: Spectrum (2] 100 50 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm) % Transmittance
A: In given IR spectra we can see broad peak at ~3400cm-1 , which indicates presence of OH group. So…
Q: 100 50 966 500 2000 1500 2000 3000 4000 wavenumber am Transmittance $%T
A:
Q: Each of the IR spectra shown below is the spectrum of one of the following compounds. Identify the…
A: Each of the functional group produces its own characteristic peak in the IR spectra, and it is very…
Q: Please help.me.to identify these peaks
A:
Q: BRUKER 50 || IS|| 3高さ 888 3500 3000 2500 2000 1500 1000 500 Wavenumber cm-1 Transmittance [%] 09 06…
A: It is trans-cinnamic acid. The characteristics peak in IR spectrum is as follows : a. Weak peak at…
Q: INFRARED SPECTRUM 0.8 0.6 OH 0.4 OH 0.2 3000 2000 1000 Wavenumber (cm-1) TRANSMITTANCE
A: In first IR, the correct answer is D. A strong peak observed around 1680-1690 cm-1 is attributed…
Q: How do I go from frequency (eV) to wavenumber (cm-1)?
A: Frequency and wave number are two characteristics of a wave which provide the details regarding the…
Q: What types of bonds are responsible for the absorptions above 1500 cm−1 incompounds A and B ?
A: The types of bonds are responsible for the absorptions above 1500 cm−1 in compounds A and B have to…
Q: Transmittance [%] 45 50 3800 3600 3400 3200 3000 2800 2600 2400 2200 2000 1800 1600 1400 600 1200…
A:
Q: The bond length of CO is 112.82 pm. What resolution is needed (in wavelength or wavenumber) if you…
A: Interpretation - To determine the wavelength or wavenumber to able to desire to distinguish the L…
Q: Intensity (a.u.) 4000 3352 cm¹ 3500 3000 UNKNOWN B 1710 cm¹ 1668 c cm¹ 2500 Wavenumber (cm¹¹) 2000…
A: Ir spectroscopy helps in the identifiaction of organic compounds.It helps in determining functional…
Q: 0.8 0.6 0.4 0.2 3000 2000 1000 Wavenumber (cm-1)
A: IR mainly explaines the functional groups that are present in molecules Alcohol: O-H stretching ,…
Step by step
Solved in 2 steps with 2 images

- Molar concentration of the dye stock solution: 2.52e-5 mol/L Operating Wavelength: 630 nm Solutions(Dilutions) Absorbance (A)(a.u.) 1 0.073 2 0.118 3 0.258 4 0.477 Using linear regression determine the absorbance/concentration relationship for the dye. [dye] =_____ x AShow that: Cp-Cv= -T(dv/dT)^2 (dp/dv)Convert the following Transmittance to absorbance: T=0.305
- Given the following data find the Molar absorption coefficient for Fe(SCN)^2+}Fe(SCN)X2+, (M−1 cm−1), if the length is 2.24cm. FIND THE UNROUNDED VALUE. Unrounded Rounded Molar absorption coefficient for \ce{Fe(SCN)^2+}Fe(SCN)X2+, (M−1 cm−1) Sample FROM PRELAB EXPERIMENTAL DATA CALCULATED VALUES 0.000300 M Absorbance Fe(SCN)X2+]equ, (M) --> for next bar 1 1.50 0.210166 0.0000300 2 3.50 0.466178 0.0000700 3 7.00 0.912604 0.000140 4 10.00 1.348948 0.000200 SLOPE, (M−1) INTERCEPT R^2R2 Unrounded 6.66606 × 103 1.20727 × 10-3 0.998979Calculate the resolution required to resolve the following peaks; i) CH3N+ (MW = 28.0187) and N2+ (MW = 28.0061) ii) C2H4+ (MW = 28.0313) and CO+ (MW = 27.9949) iii) C3H7N3+ (MW = 85.0641) and C5H9O+ (MW = 85.0653) iv) 116Sn+ (At W = 115.90219) and 232Th2+ (At W = 232.03800)Convert the accompanying transmittance data to absorbances: (a) 25.5% (b) 32.8% (c) 0.085
- The drug pentobarbital is a sedative. It was extracted from a urine sample during a particular ultraviolet-visible spectrophotometric forensic analysis procedure. The extraction solvent was evaporated and the residue reconstituted in a small volume of the transparent solvent methanol. The pentobarbital solution thus obtained had a percent transmittance (%T) of 91 at the relevant wavelength of maximum absorbance. If the urine analyte concentration were halved, what would its percent transmittance be? (You may assume that the Beer-Lambert law holds). Can you show all the steps of the solution5.4.5 Chemical microanalysis5.4.5.1 Exploitation of characteristic X-raysElectron probe microanalysis (EPMA) of bulk samples is now a routine technique for obtaining rapid, accurate analysis of alloys. A small electron probe (100 nm diameter) is used to generate X-rays from a defined area of a polished specimen and the intensity of the various characteristic X-rays measured usingeither wavelength-dispersive spectrometers (WDS) or energy-dispersive spectrometers (EDS). Typically the accuracy of the analysis is š0.1%. One of the limitations of EPMA of bulk samples is that the volume of the sample which contributes to the X-ray signal is relatively independent of the size of theelectron probe, because high-angle elastic scattering of electrons within the sample generates X-rays. The consequence of this is that the spatial resolution of EPMA is no better than ¾2 µm. In the last few years EDX detectors have been interfaced totransmission electron microscopes which are capable of operating…Potassium dichromate and potassium permanganate have overlapping absorptionspectra in 1M H2SO4 . K2Cr2O7 has an absorption maxima at 440 nm, and KMnO 4 has a band at 545nm (the maximum is actually at 525 nm, but the longer wavelength is generally used whereinterference from K2C2O7 is less). A mixture is analyzed by measuring the absorbance at these twowavelengths with the following results: A 440 =0.405, A 545 =0.712 in a 1-cm cell. The absorbances ofpure solutions of K2Cr2O7 (1x10 -3 M) and KMnO 4 (2.00x10 -4 M) in 1 M H 2 SO 4 , using the same cell gavethe following results: A Cr,440 = 0.374, A Cr,545 = 0.009, A Mn,440 =0.019, A Mn,545 = 0.475. Calculate theconcentrations of dichromate and permanganate in the sample solution.
- A)What absorbance value corresponds to 80% transmittance? B)What is the new % transmittance value if the absorbance value in A increases by 0.250?A solution containing 6.94x10^-8 M CrQ3 (Q=8 hydroxyquinolate ion) has a percent transmittance of 31.4 in a 1.00cm cell. The A of the solution is: a. -1.50 b. 68.6 c. 0.164 d. 0.503A photon of wavenumber 100 cm-1 has a wavelength of



