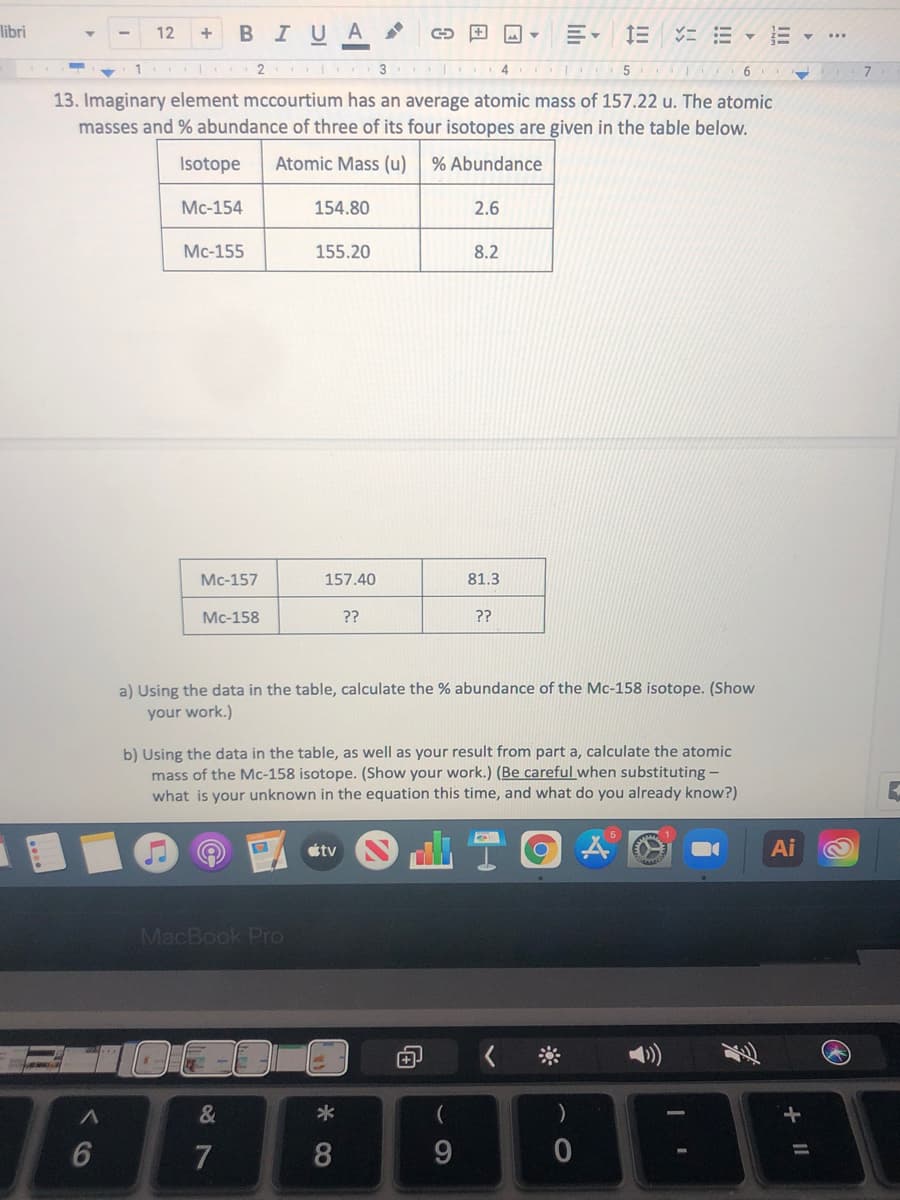
13. Imaginary element mccourtium has an average atomic mass of 157.22 u. The atomic masses and % abundance of three of its four isotopes are given in the table below. Isotope Atomic Mass (u) % Abundance Mc-154 154.80 2.6 Mc-155 155.20 8.2 Мc-157 157.40 81.3 Мc-158 ?? ?? a) Using the data in the table, calculate the % abundance of the Mc-158 isotope. (Show your work.) b) Using the data in the table, as well as your result from part a, calculate the atomic mass of the Mc-158 isotope. (Show your work.) (Be careful when substituting - what is your unknown in the equation this time, and what do you already know?)
13. Imaginary element mccourtium has an average atomic mass of 157.22 u. The atomic masses and % abundance of three of its four isotopes are given in the table below. Isotope Atomic Mass (u) % Abundance Mc-154 154.80 2.6 Mc-155 155.20 8.2 Мc-157 157.40 81.3 Мc-158 ?? ?? a) Using the data in the table, calculate the % abundance of the Mc-158 isotope. (Show your work.) b) Using the data in the table, as well as your result from part a, calculate the atomic mass of the Mc-158 isotope. (Show your work.) (Be careful when substituting - what is your unknown in the equation this time, and what do you already know?)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter2: Chemical Formulas, Equations, And Reaction Yields
Section: Chapter Questions
Problem 18P: A gaseous binary compound has a vapor density that is 2.53 times that of nitrogen at 100°C and...
Related questions
Question
Transcribed Image Text:libri
12
+
BIUA
1
3
5 I 6
7
13. Imaginary element mccourtium has an average atomic mass of 157.22 u. The atomic
masses and % abundance of three of its four isotopes are given in the table below.
Isotope
Atomic Mass (u) % Abundance
Мc-154
154.80
2.6
Mc-155
155.20
8.2
Mc-157
157.40
81.3
Mc-158
??
??
a) Using the data in the table, calculate the % abundance of the Mc-158 isotope. (Show
your work.)
b) Using the data in the table, as well as your result from part a, calculate the atomic
mass of the Mc-158 isotope. (Show your work.) (Be careful when substituting –
what is your unknown in the equation this time, and what do you already know?)
TO AC
étv
Ai
MacBook Pro
&
6
7
8.
II
+ II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning