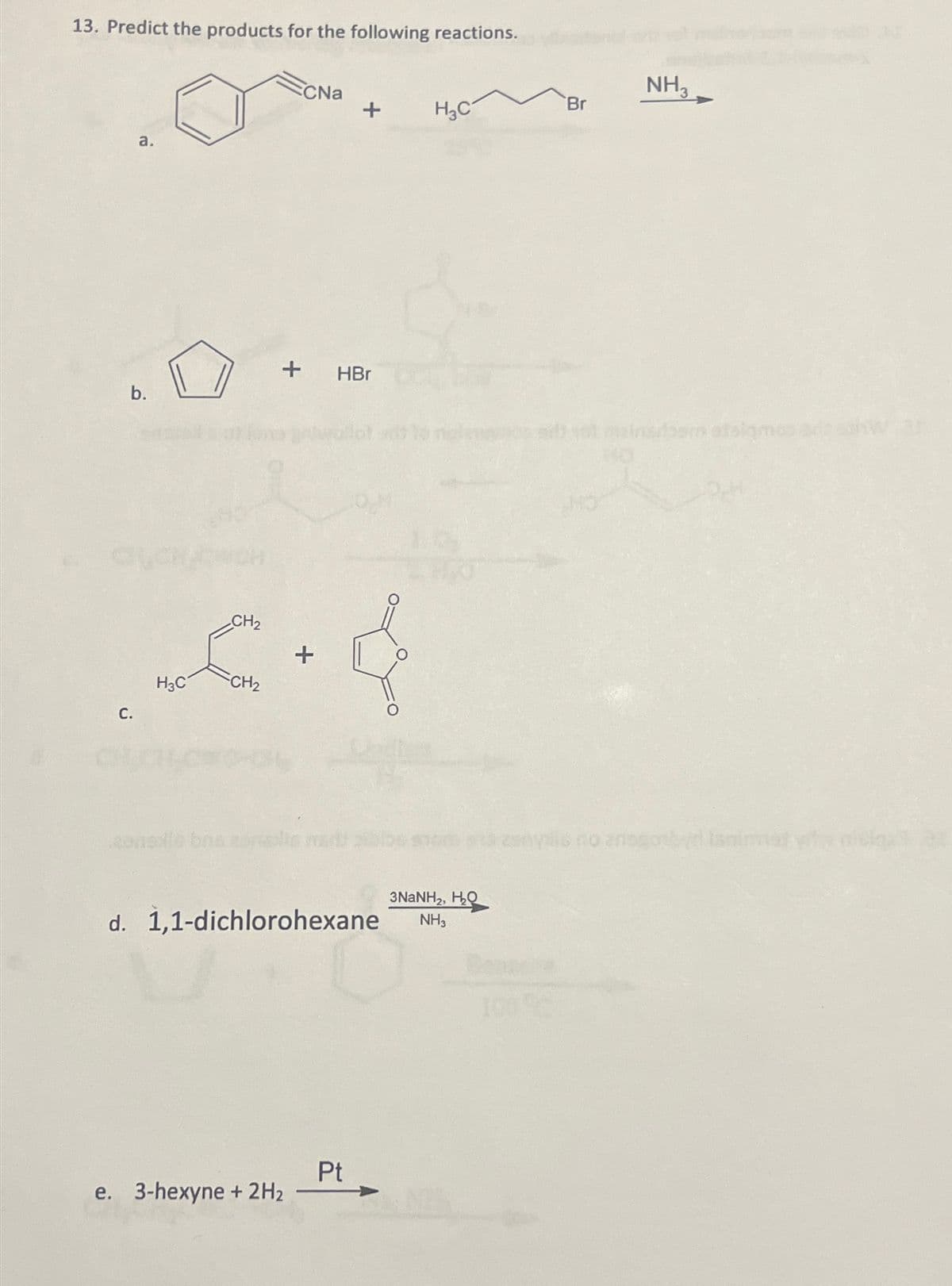
13. Predict the products for the following reactions. a. b. C. H3C CH₂ CH₂ CNa + HBr zonele bns zonsolin v e. 3-hexyne + 2H₂ + d. 1,1-dichlorohexane Pt H₂C 3NaNHz, HQ NH3 Br zsnysis NH3
Q: 4-Give the IUPAC names of the following compounds: (a) CH,CH₂C=CCOOH (d) CH, COOH (b)…
A:
Q: 15. The thiazole ring of 12 is metabolized to a toxic metab- olite. Suggest two approaches you would…
A: Thiazoles (1,3-thiazoles) are 5-membered heterocyclic compounds that can be considered as derived…
Q: gg H ܐܘ )q(
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: he lowest electronic levels for 14N 160 have the following characteristics: Calculate, for 5000 k,…
A: The objective of the question is to calculate the fraction of molecules in the 3 electronic levels,…
Q: H30+
A: -> Acidic hydrolysis of ethers involves breaking the ether linkage in presence of an acid…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. Br2 (1 equiv) FeBr3 Q Go…
A: The reactant is an aromatic compound. Its name is 4-methyanisole. In the presence of reagent, the…
Q: The scientist who first proposed the order of electron distribution within subshells was Einstein…
A: Electrons within an atom are distributed in specific subshells following the Hund's rule.. It states…
Q: 31 Show below is the change of glucose from the linear form to the ring form, which typically occurs…
A: The objective is to determine the correct statement of the change of glucose from linear to ring…
Q: Describe, using curly arrows, how limonene is biosynthesized from geranyl pyrophosphate. ham OH OH…
A: The objective of the question is to explain the biosynthesis of limonene from geranyl pyrophosphate…
Q: For the ammonia formation reaction, N2 (g) + 3H2 (g) 2 NH3(g) The equilibrium constant Kc, is 2.4 x…
A: The equilibrium constant(Kc) for a reaction is the ratio of the equilibrium concentrations of the…
Q: Add curved arrow(s) to draw step 2 of the mechanism. Modify the given drawing of the product as…
A: Information about the question
Q: A student determines the heat of dissolution of solid cesium chloride using a coffee-cup calorimeter…
A: Given:Mass of CsC = 7.38 g,Mass of water = 111.00 g,Initial temperature = 25.00 °C,Final temperature…
Q: b) H3C OH Oxone, KCl CH3CO₂Et
A: This is an example of oxidation of alcohol to ketone
Q: Draw all possible isomers of c4h7No
A: Given is organic compound. The molecular formula of given compound is C4H7NO.Isomers are compounds…
Q: When 8.50g of solid potassium hydroxide is added to 500.0g of water in a calorimeter, the…
A: The values are provided in questionmass of potassium hydroxide = 8.50 g mass of water = 500 g…
Q: 1. Design a synthesis of the following epoxide from the indicated alkyne. Be sure to consider how to…
A: The given product isIn the epoxide formed, the two methyl groups are opposite to each other. So,…
Q: Describe, using curly arrows, how limonene is biosynthesized from geranyl pyrophosphate. ham OH OH…
A: The objective of the question is to explain the biosynthesis of limonene from geranyl pyrophosphate…
Q: Predict the major product of the following reaction. HBr peroxides • You do not have to consider…
A: The given reaction is shown below.We have to predict the major product of the given reaction.The…
Q: Answer the following questions 1) What will happen if the Al - sample dipped in water for a week?
A: Given:What happens when aluminium sample dipped in water for a week.The deterioration of a material…
Q: CH₂OH OH HỘ CH2 OH ОН О Identify this molecule. [Spelling counts.] CH₂OH OH CH₂OH OH HỘ CH2 OH -0-…
A: The IUPAC name of the compound…
Q: Match each set of numbered hydrogens on the given cyclohexanone oxime structure to their topicity…
A: All parts of this question are solved in below steps.Explanation:Step 1: Step 2:
Q: What is the IUPAC name of the product for the following reaction? CH₂CH₂CH₂Br, FeBr; ?
A: This is an example of Friedel-Crafts alkylation
Q: One mechanism for the destruction of ozone in the upper atmosphere is Os (9) + NO(g) → NO₂(g) +…
A: The objective of the question is to identify the catalyst and intermediate in the given reaction…
Q: (10) 2,2-Dimethyl-5-14. bromophenylaminu methylene) - 1,3-dioxan - 4.6-dione…
A: To predict the 1H NMR peaks for the given compound, we consider the types of hydrogen atoms present…
Q: In europe gasoline is sold by the liter. Assume that it takes 14 gallons of gasoline to fill the…
A: Dimensional analysis is used to convert a physical quantity from one type of unit to some other type…
Q: Write the net ionic equation for the precipitation of lead(II) phosphate from aqueous solution: +…
A: Net ionic equation includes the "participating" ions and omit spectator ions. spectator ions remain…
Q: Problem 16.3 Each of the following alcohols has been prepared by reaction of a Grignard reagent with…
A: The above given alcohol are prepared by using Grignard reagent and ethylene oxide. General reaction…
Q: the configuration of each chiral center in the following compound: 6 Cl Apakah In the boxes below,…
A: Organic compounds are compounds that contain carbon and hydrogen atoms. We have been asked to…
Q: Assuming that benzaldehyde is the limiting reagent for the synthesis of stilbene, that stilbene is…
A: The objective of the question is to calculate the overall percentage yield of diphenylacetylene from…
Q: - The decomposition of dinitrogen pentoxide obeys the rate-law expression is 0.30 M, what is…
A: The concentration of N2O5 after 2,6 minutes will be 0.24 (M). Explanation:Step 1: The rate constant…
Q: www
A:
Q: The equilibrium constant Kc = 4.0 at a certain temperature. A reaction is carried out starting with…
A: b) 1.3 MExplanation:
Q: Identify this compound with its IUPAC name.
A: The IUPAC name of the compound…
Q: 19. What is the major product of the sequence of reactions shown below? SCH a) b) c) d) CH Fr. O…
A: Bromination of alkanes is the substitution reaction which involves the replacement of hydrogen by…
Q: 4. Describe how the following compounds could be prepared from cyclohexanone using an enamine…
A: When amine compound react with carbonyl compound in presence of acid catalyst, imine formation which…
Q: b) OH OH 1. Na2Cr2O7, H2SO4 (aq) LOH, H', heat 2.
A: We have to predict the major organic products.
Q: 1) Give the IUPAC names of the following carboxylic acid derivatives. нель CI CI سلمل له سلہ que ly…
A: We have to write the IUPAC names of the given structures.
Q: Indicate, by letter, the strongest acid and the weakest acid of those below. COOH The strongest acid…
A: The acidity of an acid is determined by its ability to donate a proton (H+) in a chemical…
Q: Use the data to estimate the Keq of the forward reaction.
A: We have to find Keq
Q: Draw the major product of the following reaction. H NH₂ Na(CN)BH3 pH-6
A: ReactionProduct: Need to be drawn
Q: A weak monoprotic acid is titrated with 0.100 MNaOH. It requires 50.0 mL of the NaOH solution to…
A: Given:Calculate the pKa of the weak acid.The strength of an acid in aqueous solution is determined…
Q: AcOH
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 1. The atomic number of lithium is 3. How many neutrons does an atom of lithium have if it is…
A: The objective of these questions is to understand the atomic structure of different elements and…
Q: identify starting reagent
A: The objective of the question is to identify the starting reagent AcOH in the context of chemistry.
Q: Which of the following are intermediates in the following
A: Firstly all acid-base reaction takes place.In the next step negative charge of oxygen gets back and…
Q: 3. Predict the products of the following intramolecular reactions. (Hint: only 1 of the 2 bottom…
A: Product: Need to be determine
Q: Please don't provide handwritten solution ...
A: The concentration of A2- or [A2-] in the given solution is calculated as 1.55 x10-10…
Q: For the cationic polymerization of ethyl vinyl ether initiated with a water/aluminum chloride…
A: Cationic polymerization is a process during which a cationic initiator interacts with monomer which…
Q: 5. What is the product of the reaction shown? CH3OCH2 О OCH3 (A) CH3OCH2 Н CH3O H Н CH3O (B) HOCH2…
A: This is an example of hydrolysis of acetal to hemiacetal
Q: Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is S₂(g) + C(s)…
A: The objective of the question is to find out how many grams of CS2(g) can be prepared by heating…
Step by step
Solved in 3 steps with 1 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?In the reaction below, 3-Buten-2-one reacts with 2,3-Dimethyl-1,3-butadieneand 1,3-Butadieneat 140°C and 30°C, respectively. Explain why these two dienes react at different temperatures.Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…
- Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other. a. What was the major product? b. Why was more of one product obtained than of the other?Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?
- give the reagents for parts a-pGiven: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)In the beginning of this video https://www.youtube.com/watch?v=9Ng6Zv9oLzk, what reason is given on why the dienophile likes the diene?
- When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidWhat would the final products look like? Pls specify stereochemistry if needed

