13.48 The cell Pt|H;(1 bar)|HBr(aq)|AgBr(s)|Ag|Pt' at 25°C with HBr molality 0.100 mol/kg has E =0.200 V. Find the activity coefficient y of HBr(aq) at this molality.
Q: Calculate the activity coefficient of K+ in 0.050 M KNO3. pls show the solution, thank u
A: Given-> Molarity of KNO3 = 0.050 M
Q: Calculate the solubility at 25 °C of PbCO 3 in pure water and in a 0.0020 M Pb (NO3), solution.…
A: Given ; Ksp of PbCO3 =7.4 x 10-14 Let the solubility of PbCO3 in pure water is S mol/L…
Q: ST5G.2 - Estimate the solubility of CaF2(aq) [in units of PICOmolar] in the presence of 0.701 molar…
A: Here we have to determine the solubility of CaF2 in picomolar in presencee of 0.701M of AlF3. Dta…
Q: Consider the equilibrium reaction: AB2(s) ⇌ A2+(aq) + 2B-(aq). What is the molar solubility of…
A: Step 1: AB2 (s) ↔ A2+ (aq) + 2B- (aq) YB2 (aq) ↔ Y2+ (aq) + 2B- (aq)
Q: Calculate the solubility and molar solubility of Cu3(PO4)2 (MW=380.58 ; Ksp=1.4x10-37).
A:
Q: The partial pressure of O₂(g), referred to as P(O₂(g)), in the lungs is 0.210 atm. The Keq of oxygen…
A: To calculate the molar concentration of oxygen dissolved in blood at 310.0 K, given that the…
Q: Calculate the solubility at 25 °C of AgCl in pure water and in a 0.0170 M AgNO3 solution. Round both…
A:
Q: For the reaction N2(?) + 3 H2(?) ⇌ 2 NH3(?): a. Write an expression for standard reaction Gibbs…
A: ΔrG = ∑i=1nNi μi Where μi is the chemical potential of the species Ni is the particle number…
Q: If the cell constants of 0.2 cm^-1 and the molar conductivity of H+ and Cl- are known, 350…
A:
Q: The dissociation vapor pressure of a salt A2B(s) ⇌ A2(g) + B(g) at 367°C is 208 kPa but at 477°C it…
A:
Q: Calculate the activity coefficient of K* in 0.050 M KNO3.
A: Given: 0.050M KNO3 To find: activity coefficient of K+.
Q: What is the molar solubility (in mols/L)of silver chloride in 2.00 M NH3(aq) given that Ksp of AgCI…
A:
Q: Calculate the % relative error in solubility by using concentrations instead of activities for…
A: The activity coefficient is the factor that relates the concentration of the substance in the ideal…
Q: Outline the change in composition of a solution of the salt of a triprotic acid as the pH is changed…
A:
Q: The standard reaction enthalpy of Zn(s) + H2O(g) → ZnO(s) + H2(g) is approximately constant at +224…
A: Given: The standard reaction enthalpy is +224 kJ.mol-1 The standard reaction Gibbs energy is +33…
Q: At 250C, Ksp for AgBr is 5.0 x 10-13. Calculate the solubility of AgBr in g/L at 25 oC (a) in pure…
A: Given : Ksp of AgBr = 5.0 X 10-13 Molar mass of AgBr = Atomic mass of Ag + Atomic mass of Br = 108…
Q: 7. The bisulfate ion, HSO4', is a weak acid with Ka = 0.012. HSO4 (aq) H*(aq) + SO42(aq) a)…
A: Using the relation,∆G°=-RTlnKGiven,Ka=0.012
Q: Calculate the masses of (a) Ca(NO3)2 and, separately, (b) NaCI to add to a 0.150 mol kg-1 solution…
A:
Q: Calculate the value of the reaction quotient, Q, of silver iodate (AgIO3) when 10.0 mL of 0.011 M…
A:
Q: Calculate K for each salt in an aqueous solution at 25 °C in which u = 0.0100 M. Use the table of Kp…
A: CONCEPT: K'sp =Concentration solubility product constantK'sp =Ksp γx γyKsp = Thermodynamic…
Q: 4. What is the solubility in mol/L of barium sulfate in water if its Ksp is 1.5x10⁹?
A: We know that the solubility product of ionic compounds can be defined as the solubility product is a…
Q: 10p in recent years, technology has developed considerably in analytical devices as in many areas…
A:
Q: Calculate the activity coefficient of K+ in 0.050 M KNO3.
A:
Q: Calculate the solubility (in g/L) of CaSO,(s) in 0.450 M Na, SO, (aq) at 25°C. The Ksp of CaSO, is…
A:
Q: What is the solubility of CO2 in a soft drink (which is mainly water) that was bottled under a CO2…
A: From the Henry's law, we can calculate the solubility. P =KS S is solubility in mol/L. K is…
Q: The equilibrium constant of the reaction 2 C3H6 (g) ⇌ C2H4(g) + C4H8(g) is found to fit the…
A: Standard Gibbs free energy: It is the sum of standard enthalpy and the standard entropy multiplied…
Q: Calculate the solubility (in g/L) of CaSO,(s) in 0.400 M Na, SO,(aq) at 25°C. The Ksp of CaSO, is…
A: When Na2SO4 ionizes in water, it dissociates to give two moles of Na+ and one mole of SO42-ion.
Q: The solubility of Ag,CrO, in water is 8.00.6 x 10-5m at 298 K and its solubility in 0.04 m NaNO3 is…
A: Solubility is a phenomenon where the solute get dissolved in a solvent to form a solution. The…
Q: 2- The concentration of SO, in a saturated solution of Hg2SO4 is 5.54x10-3 mol/L at a certain…
A: Given- Concentration of SO42- = 5.54 × 10-3 Mole/L Ksp of the compound=?
Q: If the cell constant for conduction is 0.2 cm1 and the molar conductivity of H* and Cl is 350…
A:
Q: A solution of a fictitious silver salt, AgX, in pure water has a concentration of silver ions of…
A:
Q: 5. The limiting molar conductivities, A°, of KCl, KNO; and AgNO3 are 149 S cm2 mol', 145.0 S cm2…
A:
Q: Consider an aqueous solution of pyridine, C5NH5(aq), with F = 5.0 mM, at 1 bar and at 298 K.…
A: According to the Bronsted-Lowry acid-base theory, an acid is that which gives H+ ions and the base…
Q: The dissociation vapor pressure of a salt A2B(s) A2ig) + B(g) at 367°C is 208 kPa but at 477°C it…
A:
Q: Calculate the solubility of silver acetate.1 (CH;COOA9) in water at 25 °C, assuming * .the DHLL to…
A: The solubility product constant is the equilibrium constant for the dissolution of a solid substance…
Q: Aniline, C6H7N (molar mass = 93.13 g/mol), is a weak base that is used as a precursor in the…
A: 924 mg of aniline is dissolved in enough water to make a total volume of 1.00 L. pKb of aniline…
Q: State the limits to the generality of the expression for estimating the pH of an amphiprotic salt…
A:
Q: Write the expression for the solubility-product constant for CaF2, and look up the corresponding Ksp…
A: The solubility product of a sparingly soluble salt represents the product of the molar…
Q: Calculate the value of the equilibrium constant, K., for the reaction AgBr(s) + 2 NH, (aq) =…
A:
Q: 4. Given the following data, calculate the concentration (molarity) of Sr2+ in a saturated aqueous…
A: Introduction : Molarity can be defined as the number of moles of a solute in grams present in 1…
Q: Calculate the molar solubility of SnCOH)2 ( Kçp =3.0 X10) in 5.0M NH,N0, solution- %3D Tkb for…
A: Henderson- Hasselbalch equation The equation that is used to find the concentration of a buffer…
Q: Neglecting any effects caused by volume changes, what is the effect on the ionic strength by the…
A: a) Addition of iron(III) chloride to HCl, ionic strength of HCl increases. b) Addition of…
Q: Write out the equilibrium expression for the solvation of Ag2CrO4 in water
A: The equilibrium expression for the solubility of an ionic compound in water can be written on the…
Q: CaF2(s) → Ca2*(aq) + 2 F´(aq) Part B which has the following standard enthalpy and entropy of…
A: We have to calculate Gibbs energy Change (∆G) for the reaction. CaF2(s) →…
Q: Sodium acetate, NaCH3CO2, of mass 7.4 g is used to prepare 250 cm3 of aqueous solution. What is the…
A:
Q: Explain the common-ion effect.
A: The common ion effect is an effect that suppresses the ionization of an electrolyte when another…
Q: Calculate the solubility (in g/L) of CaSO,(s) in 0.500 M Na, SO, (aq) at 25°C. The Ksp of CaSO, is…
A: Given that : Ksp of CaSO4 = 4.93× 10–5 Concentration = 0.500 M Na2SO4
Q: The Ksp of Ca3 (PO4 ) 2 is 1.3 × 10−26 . Estimate the solubility of this salt in units of g. L −1 .…
A:
Q: Potassium acid phthalate, KHC8H4O4 ( MM=204.2 ), reacts with sodium hydroxide on a 1:1 molar basis.…
A: Given:: mass of Potassium acid phthalate = 0.4823 g, molar mass = 204.2 g mol-1 titrant NaOH…
Q: 4. The equilibrium constant of a reaction A(g) = 2B(g) was found to fit the expression 4375 +1.75…
A:
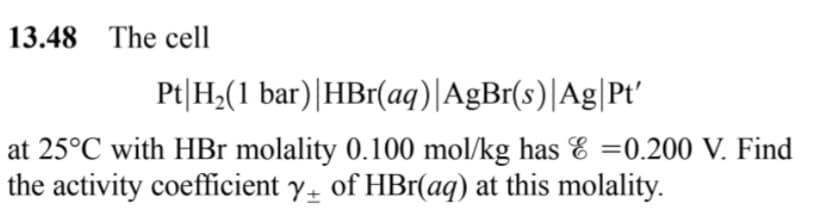
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- For a salt MpXq define the mean activity coefficient, where n= p+q, in terms ofthe separate activity coefficients of the ions.The standard reaction Gibbs energy for the reactionH2(g) + (1)/(2)O2(g) → H2O(I) is -237.13 kJ mol-1 at 25 °c. Determine the equilibrium constant in terms of concentration, Kc, at this temperature.7.5(a) The standard reaction enthalpy of Zn(s) + H2O(g) → ZnO(s) + H2(g) is approximately constant at +224 kJ mol−1 from 920 K up to 1280 K. The standard reaction Gibbs energy is +33 kJ mol−1 at 1280 K. Estimate the temperature at which the equilibrium constant becomes greater than 1.
- Lactic acid (CH3 - CH(OH) - COOH) is a weak acid and therefore a weak electrolyte.found in "cut milk". The freezing point of an aqueous solution 0.01 m of lactic acid is 0.0206 ºC. Knowing that in aqueous solution the following equilibrium occurs:CH3 -CH(OH) -COOH(ac) = CH3 -CH(OH) -COO(-)(ac) + H(+)(ac)and that, therefore, there are three species (solutes) in solution, calculate their percentage of ionization.Lactic acid (CH3 - CH(OH) - COOH) is a weak acid and therefore a weak electrolyte. found in "cut milk". The freezing point of an aqueous solution 0.01 m of lactic acid is 0.0206 ºC. Knowing that in aqueous solution the following equilibrium occurs: CH3 -CH(OH) -COOH(ac) = CH3 -CH(OH) -COO(-)(ac) + H(+)(ac) and that, therefore, there are three species (solutes) in solution, calculate their percentage of ionization.Estimate the aqueous gram-solubility in g/L of mercury(I) chloride: Hg2Cl2(s) ↔ 2Hg+ (aq) + 2Cl– (aq), Ksp = 1.43×10–18 The molar mass of Hg2Cl2 is 472.06 g/mol.
- Calculate the Gibbs free energy of the reaction from mixing 50 mL each of 0.50 M Ag+(aq) solution and 1 M of NaCl(aq) to form AgCl(s) at 25°. The Ksp of AgCl is 6.0 × 10—11 at 25 °C.Calculate the Gibbs free energy of the reaction from mixing 50 mL each of 0.50 M AgNO3(aq) solution and 1 M of NaCl(aq) to form AgCl(s) at 25°. The Ksp of AgCl is 6.0 × 10—11 at 25 °C.Thermodynamic data can be used to predict the solubilities of compounds that would be very difficult to measure directly. Calculate the solubility of mercury(ll) chloride in water at 25 °C from standard Gibbs energies of formation.
- The dissociation vapour pressure (the pressure of gaseous products in equilibrium with the solid reactant) of NH4CI at 427 °C is 608 kPa but at 459 °C it has risen to 1115 kPa. Calculate (a) the equilibrium constant. (b) the standard reaction Gibbs energy, (c) the standard enthalpy, (d) the standardentropy of dissociation, all at 427 °C. Assume that the vapour behaves as a perfect gas and that ΔHΘ and ΔSΘ are independent of temperature in the range given.The reaction A(g) + B(g) ↔ C(g) + D(g) has ΔGrxn° = -28.62 kJ mol-1 and Kp = 0.63 at 980 °C. A rigid cylinder at that temperature contains 1.2 atm of A, 0.20 atm of B, 0.30 atm of C, and 0.27 atm of D. What is the reaction Gibbs energy?Consider an aqueous solution of pyridine, C5NH5(aq), with F = 5.0 mM, at 1 bar and at 298 K. Pyridine is a weak Brønsted-Lowry base. Structure of pyridine, Write all the relevant chemical reactions among the species

