150 to 140 130 120 110 100 NaNO 90 80 70 60 50 KCI 40 Nac 30 20 KCIO Ceso, O 10 20 30 40 50 60 70 80 90 100 10 Temperature (C) 35 grams of potassium chlorate are dissolved in 100 grams of water. The solution is heated until all of the solid dissolves, and then cooled to 40°C. How many grams of potassium chlorate precipitate out? O 20 grams O 35 grams 15 grams none Grams of solute per 100 g H0
150 to 140 130 120 110 100 NaNO 90 80 70 60 50 KCI 40 Nac 30 20 KCIO Ceso, O 10 20 30 40 50 60 70 80 90 100 10 Temperature (C) 35 grams of potassium chlorate are dissolved in 100 grams of water. The solution is heated until all of the solid dissolves, and then cooled to 40°C. How many grams of potassium chlorate precipitate out? O 20 grams O 35 grams 15 grams none Grams of solute per 100 g H0
ChapterU6: Showtime: Reversible Reactions And Chemical Equilibrium
Section: Chapter Questions
Problem 10STP
Related questions
Question
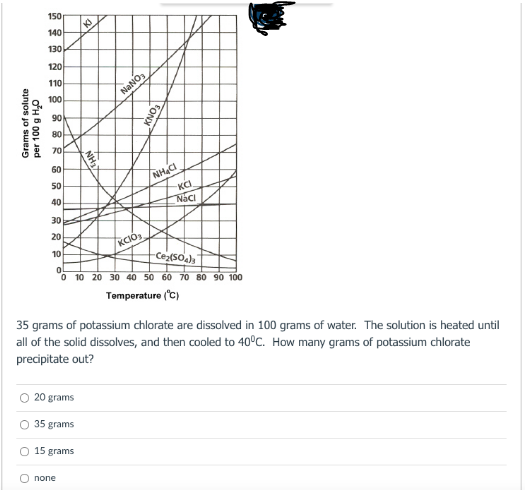
Transcribed Image Text:150
to
140
130
120
110
100
NaNO
90
80
70
60
50
KCI
Naci
40
30
20
KCIÓ
10
Cez(so)
O 10 20 30 40 50 60 70 80 90 100
Tomperature ('C)
35 grams of potassium chlorate are dissolved in 100 grams of water. The solution is heated until
all of the solid dissolves, and then cooled to 40°C. How many grams of potassium chlorate
precipitate out?
20 grams
35 grams
15 grams
O none
FON
NH
Grams of solute
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning