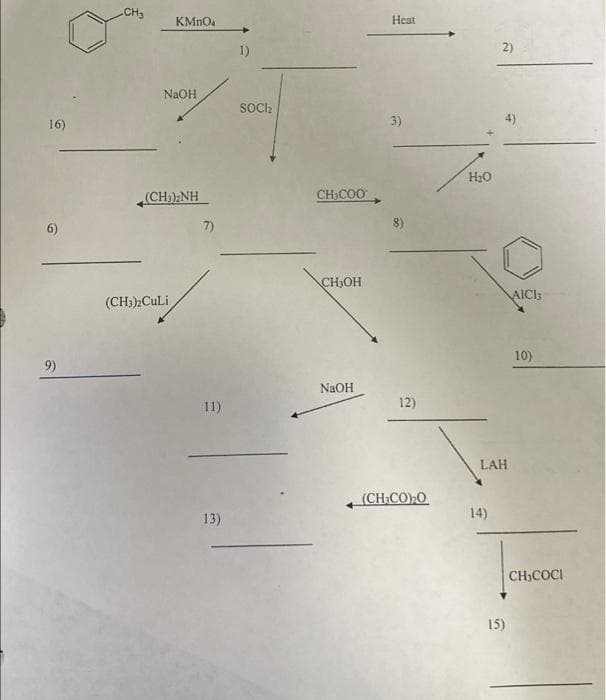
16) 6) 9) CH₂ KMnO4 NaOH (CH))NH (CH₂)2Culi 7) 11) 13) 1) SOCI₂ CH₂COO CH₂OH NaOH Heat 3) 8) 12) , (CH,CO)O H₂O 2) LAH 14) 15) AICI 10) CH₂COCI
Q: Hi! I need an explanation for this. More so, a technique. Technically, we're just separating the…
A: Friedel-Craft acylation reaction is a special type of reaction in which an acyl group is bonded to…
Q: CH3ONA, CH3OH heat H Na O Br:
A: This is an example of the elimination reaction.Methanol is protic polar solvent which promotes SN1…
Q: Standard Reduction Potentials at 25 °C Half-reaction Eº(volts) Au³+ + 2e Au+ 1.290 Aut+eAu 1.680…
A: A disproportionation reaction is a reaction in which a single chemical species undergoes both…
Q: Predict the effect of increasing temperature the following reaction A(1)+B (1) C (1) AH=5.4 kj/mol…
A: The question is based on the concept of Le-chatlier principle.It States that when a system at…
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A:
Q: Draw the product of the reaction shown below. Use dash and/or wedge bonds to indicate…
A:
Q: Macmillan Learnin At 1 atm, how much energy is required to heat 87.0 g H₂O(s) at -24.0 C to H₂O(g)…
A:
Q: 2. Draw two resonance structures for the structure shown below. Use curved arrows to indicate…
A: If the Lewis structure of a molecule or ion cannot be explained by a single structure then more than…
Q: Q8. Draw three representative segments of the polymer formed from the following monomer.
A: Polymers are made of basic organic molecules known as monomers. These monomers attach with in…
Q: d. -C CH 1. 1 eq. CH3CH₂MgBr 2. H3O* 3. DMSO, COCI2, (CH3CH2)3N, CH2Cl2
A: The given reaction scheme is shown below.We have to give the final product of the given reaction.
Q: Which is the most stable species? (A) (B) OA OB OC OD (C) (D)
A: Order of stability of radicals: Allylic = Benzylic > 3° > 2° > 1°
Q: .O-CH3 حمد سینہ H OH ОН
A: Functional group is a group of atoms having distinctive chemical properties responsible for the…
Q: Draw the correct products for the reaction. 1. Cl₂, NaOH 2. H₂O* Product A+ Product B
A: Given is organic reaction.In this reaction, the reactant is acetophenone.Let's see products of this…
Q: The compound below can be prepared with an alkyl iodide and a suitable nucleophile. Identify the…
A:
Q: owing orbitals cannot exist according to the quantum theory: 2s, 2d, 3p, 3f. 4f. and 5s. (Select all…
A: We are given following orbitals :2s , 2d , 3p, 3f , 4f, 5sWe have to answer which of them doesn't…
Q: If benzoic acid (C6H5COOH) is 0.42% ionized in a 0.80 M solution, what is the Ka of benzoic acid?
A: Answer:When a weak acid solution is added in water then its partial ionization takes place and an…
Q: Order the compounds made of the following molecules by increasing melting point. HO HO 0= FO HO HO…
A: Melting point - Melting point is the point at which substance changes from a solid to a liquid.In…
Q: The following ball-and-stick molecular model is a representation of acetaminophen, the active…
A: In the skeletal formula, each covalent bond between carbon-carbon or carbon-heteroatom is…
Q: Show inetmediates and electron flow
A: Methoxide (MeO-) acts as a nucleophile and attacks the electron-deficient carbonyl carbon. The…
Q: Draw the Lewis structure (with formal charges) of the indicated chemical species. If there is…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A: Given :1. HBrO2 2. BrO31-We need to draw the lewis structure with formal charge.
Q: atomic mass electronegativity electron affinity ionization energy heat of fusion 190.23 2.20 106.1…
A: Answer:The energy required to remove an electron from an isolated gaseous atom is called as…
Q: Please explain the procedure to analysis aromatic compound in river water using HPLC?
A: To explain : Procedure for the analysis aromatic compound in river water using HPLC.Procedure…
Q: draw a sketch of the predicted NMR of this compound
A:
Q: What is the equilibrium constant expression for each reaction below? 3NaBr(aq) + CuN(s) →…
A: (1) Equilibrium constant expression = ?
Q: Solid lead(II) nitrite is slowly added to 50.0 mL of a ammonium bromide solution until the…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: Periodic table is the arrangement of elements in the order of atomic number. The arrangement is done…
Q: atomic mass electronegativity electron affinity ionization energy heat of fusion 85.468 0.82 46.9…
A: Answer:Energy required to remove an electron from an isolated gaseous atom is called as ionization…
Q: Give detailed mechanism Solution with explanation needed..don't give Handwritten answer
A: Markovnikov's rule is used for addition of unsymmetrical reagent to the unsymmetrical alkenes and…
Q: Which is the proper IUPAC name of the major organic product of the reaction provided? A B OC Cl₂, hv…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: ased on the numbering pattern, please assign all peaks to the carbon specstroscopy
A: We have to label each single carbon in the given structure. 13 C-NMR is generally used to detect the…
Q: 1. Uranium hydride is formed by the reaction of hydrogen gas with solid uranium. The equilibrium…
A: Balanced chemical reaction is given as below:Equilibrium pressure is given as:The equilibrium…
Q: Question 11 How many possible products can the following reaction produce? CH₂CH3 CH₂CHCH=CHCH; +…
A:
Q: Calculate the cell potential for the galvanic cell in which the reaction Ti(s) + Cu²+ (aq) = Ti²+…
A: The given red-ox reaction is Ti(s) + Cu2+(aq) Ti2+(aq) + Cu(s)Given: The concentration of Ti2+,…
Q: Combustion of glucose (CHO) is the main source of energy for animal cells: CHO (s) + 60₂(e) -…
A: Maximum number of ATP's that can be created is calculated below.
Q: Complete the structure of NutraSweet/aspartame (C14H18N2O5) by indicating the positions of the…
A: Given :Incomplete structure of aspartame (C14H18N2O5).We need to complete the structure of aspartame…
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: The objective of the question is to predict the entropy change associated with the change in the…
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A: Hydrogen bond is a special type of bond which is formed between the most electronegative atoms (F,…
Q: In the laboratory, a general chemistry student measured the pH of a 0.532 M aqueous solution of…
A:
Q: 15. For the equilibrium established below A(g) B (g) has equilibrium constant K = 0.50. Suppose the…
A: Given:Initial concentration of A = 1.0 MInitial concentration of B = 0.0 MKc = 0.50
Q: Draw the most prominent ionization state for phenylalanine (pKa for acidic hydrogens shown below) at…
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Q: Which of the following represents a step in the mechanism for the alkoxymercuration of…
A: Hydration of alkene proceeds by an electrophilic addition reaction mechanism to form an alcohol.…
Q: Calcium carbonate (CaCO3) reacts with stomach acid (HCl, hydrochloric acid) according to the…
A:
Q: Draw the organic product(s) of the following reaction. 1 eq. NaNH2, NH3()) H-CEC-H • You do not have…
A: From the hydrocarbons, alkanes and alkenes do not show acidic properties, but the terminal alkyne…
Q: What is the balanced chemical equation for the chemical reaction depicted in the figure? Be sure…
A: Information of question
Q: solution A iron (II) nitrate zinc sulfate sodium chloride solution B sodium hydroxide manganese(II)…
A: First, write the balanced reaction between compounds and check the solubility of the product using…
Q: Draw all of the expected products for the following solvolysis reaction: Br XBOH ? EtOH heat Draw…
A: 3° alkyl halide undergo E1 or SN1 pathway.More substituted alkene will be the major product.
Q: Predict the products of this organic reaction: br + NaOH Specifically, in the drawing area below…
A: Esters are formed when carboxylic acid reacts with alcohol. So on hydrolysis in acidic medium…
Q: 10. CHALLENGE. Calculate AHrxn. N2H4(1) + O2(g) → N2(g) + 2 H₂O(1) given: 2 NH3(g) + 3 N2O(g) → 4…
A:
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Step by step
Solved in 4 steps with 3 images


