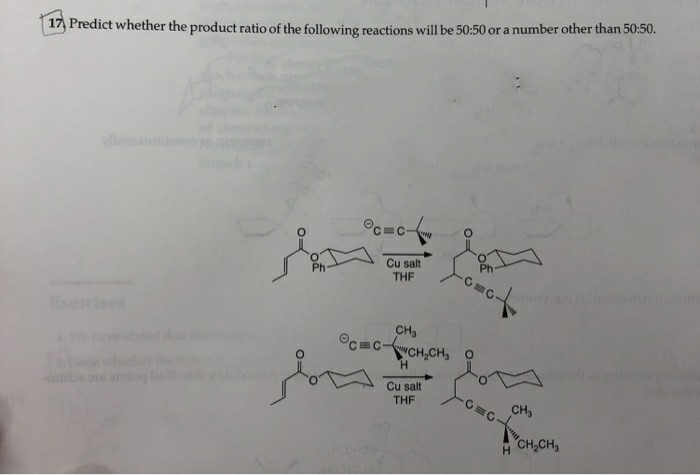
17, Predict whether the product ratio of the following reactions will be 50:50 or a number other than 50:50. Cu salt THE ercises CH OCCCH,CH, O H. Cu salt THF CH, CH,CH,
Q: 3
A: Given: Amount of starch has produce = 2.50×103 kg percent of starch in potato = 1.83 % let 1.83 kg o...
Q: I just asked this question and didn't get the right answer. Me and the helper both got 33.2 mg. This...
A: Given : Value in grams = 3.32 X 10-5 g
Q: Palmitate and Behenate are saturated fatty acids with 16 and 22 Carbons respectively. Which would yo...
A: Concept: The melting point (Tm) of fatty acids depends on the number of the carbon atom and the degr...
Q: Calculate the osmotic pressure of each of the following aqueous solutions at 20.00°C: 32.0 mL of g...
A: As you nor specified so I am giving answer of first question as per guidlines Osmotic pressure=CRT M...
Q: The density of zinc is 7.1 g/cm3g/cm3 What is its density in kilograms per cubic meter Express your ...
A: Given, Density of Zinc = 7.1 g/cm3 We need to convert its density in kilograms per cubic meter (kg...
Q: Is CH3CH2CH2CH2OLi more soluble in water than CH3CH2CH2CH2OH?
A: The compounds given are 1) CH3CH2CH2CH2OLi 2) CH3CH2CH2CH2OH
Q: Write the condensed electron configurations for atoms of the following elements, using abbreviations...
A: The condensed electron configurations for atoms of the following elements, using abbreviations for n...
Q: The energies, ? , for the first few states of an unknown element are given in the table in arbitrary...
A: The final energy of electron is the sum of energy in the atom and the energy in the photon and its e...
Q: Determine the molar solubility for Ag₂SO₄ (Ksp = 1.5 × 10⁻⁵) in a solution that already contains 0.2...
A: solunb
Q: Problem (1) Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomer...
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question s...
Q: A) Calculate the mass of a gold-197 atom using the information from question 9 and the masses of the...
A: The ratio of mass to the volume of the solution is known as density.
Q: Q1
A: An oxidation number of an element can be positive, zero, or negative. The oxidation number of an ele...
Q: A student has a sample of CaSO4 hydrate and it weighs 0.4813 grams. He heats it strongly to drive of...
A: Given: mass of CaSO4 hydrate = 0.4813 g mass of anhydrous CaSO4 = 0.3750 g
Q: Write the balanced COMPLETE ionic equation for the reaction when (NH₄)₃PO₄ and Na₂SO₄ are mixed in a...
A:
Q: 10. 0.287 M 20. 0.189 M 30. 0.141 M 40. 0.112 M Use this data to answer the following questions. Wri...
A: reaction : 2[SO3]→2SO2+O2 rate of a reaction : rate=k[r]a where k is rate constant a is order of th...
Q: is 2-butanone a correct IUPAC name?
A: The IUPAC name given is 2-butanone.
Q: Which of the following factors (maybe more than one) contributes to high viscosity for a liquid? low...
A: Viscosity is the opposing force that acts on the liquids. It opposes the flow of the fluids. The fac...
Q: Draw a stepwise mechanism for the following reaction
A: Acetals are produced when an aldehyde molecule reacts with alcohol molecules. The generic formula of...
Q: Under certain conditions, the substance hydrogen peroxide (H2O2) can be broken down to form water an...
A: The decomposition of hydrogen peroxide takes place as follows: 2H2O2 (aq)→ 2H2O(l) + O2 (g) [Balan...
Q: If 39.4 g of AgNO3 react with 28.6 g of H2SO4 according to this UNBALANCED equation below, what is t...
A:
Q: Determine the molarity of each of the following solutions from its osmotic pressure at 25°C. Include...
A: Given: Osmotic pressure = π = 0.605 atm Van't Hoff factor for the solution = i = 2.47 Temperature = ...
Q: The heat of vaporization for benzaldehyde is 48.8 kJ/mol, and its normal boiling point is 451.0 K. U...
A: Given: Heat of vaporization of benzaldehyde = 48.8 kJ/mol Normal boiling point of benzaldehyde = 451...
Q: An evacuated reaction vessel is filled with 0.80 atm of N₂ and 1.68 atm of Br₂. When equilibrium is ...
A: Use relation of Kp and given values Calculate kp Kp= 82.4
Q: A solution is made initially with 0.200 M HIO₃ (Kc = 0.170). Once the equilibrium below is establish...
A: The equilibrium reaction (dissociation reaction) taking place can be written as, => HIO3 (aq) ---...
Q: Choose the most appropriate systematic (IUPAC) name from the choices below that accurately describes...
A: B is the correct option CH3 and OH are opposite side so they are anti position Name will be trans-...
Q: Point Group Worksheet Using models, determine the point group for each of the following molecules. I...
A: I am supposed to Answer only the first three sub questions please do repost for the rest.
Q: Write a balanced chemical equation based on the following description: propanol, C₃H₇OH(l) burns in ...
A: The reactants given are 1) Liquid propanol i.e C3H7OH (l) 2) Oxygen (from air) i.e O2 (g) And the ...
Q: Complete the balanced dissociation equation for the compound below in aqueous solution. If the compo...
A: The balanced dissociation equation for the compound below in aqueous solution
Q: 17
A: Given : Concentration of Barium hydroxide i.e Ba(OH)2 = 0.0100 M And volume of Ba(OH)2 solution = 50...
Q: Write structures for the following compounds. 3-ethyl-4-methylhexane 4-tert-butyl-2-methylheptane ) ...
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for...
Q: 4
A: Mass= ? Density=1.23g/ml Volume=31.2ml Mass=density×volume
Q: What product is formed when D-Altrose is treated with C6H5CH2NH2, mild H+
A: An imine is formed when an aldehyde or ketone reacts with a primary amine. The reaction is catalyzed...
Q: a. Calculate the Gibbs energy of formation for a generic compound AB2 in kJ/mol given the following ...
A: The formation reaction of molecule AB2 can be written as, => A (l) + B2 (g) ---------> AB2 (s)...
Q: Methanol (CH4O) is an organic solvent with a boiling point of 64.5°C. What is the concentration, in ...
A: When a solute is added to a pure solvent , many changes occur in the properties of those solutions. ...
Q: How would incomplete transfer of solute from a beaker to the volumetric flask affect the concentrati...
A: If we do not dissolve complete Solute in solution then mole of Solute in solution will be less And ...
Q: What is the empirical formula of a compound composed of 23.5 g of potassium (K) and 4.81 g of oxygen...
A:
Q: Fertilizer sample 1.150 g containing SO4 -2 ion is dissolved in 500ml dist. Water. 100ml of the solu...
A: %(mass/mass) is a unit of concentration that represents the presence of the mass of solute (in gram)...
Q: What is the molality of a 5.117 M solution of phosphoric acid with a density of 1.254 g/mL?
A:
Q: Draw all the isomers of C,H10: Clearly show stereochemistry if stereoisomers are possible. Step 1: S...
A: In linear form stereochemistry can show on double bond if grouping is different on double bond
Q: Enter a balanced nuclear equation for the production of lead-212, used in the treatment of some canc...
A: Radioactive decay refers to the breakdown of an unstable nucleus to a stable nucleus with the releas...
Q: A different experiment has a copper unknown which has two moles of copper for every one mole of unkn...
A: Given : Mass percent of copper in the compound = 78.35 % And there are 2 moles of copper for every ...
Q: 1. Assume that the hydrocarbonlike interior cavity of the SDS micelle has a diameter of - 3 nm and t...
A: Gibb’s free energy (∆G)– Formula is, ∆G=∆H-T∆S Where, ∆G= Gibb’s free energy ∆H=change in entha...
Q: Draw the structure for 2-ethylcyclopentanecarbaldehyde :
A: The Structure of 2-ethylcyclopentanecarbaldehyde is given as:
Q: 16
A:
Q: A 15.630 gg milk chocolate bar is found to contain 9.815 g of sugar. What will be the amount of suga...
A: Given : Mass of sugar = 9.815 g And mass of bar = 15.630 g The mass of reduced bar = 4.000 g
Q: Part A 13 How many moles of atoms are in 6.00 g of C? Express your answer numerically in moles. • Vi...
A: Given : Mass of 13C = 6.00 g
Step by step
Solved in 2 steps with 2 images

- Prove that the Keq for the reaction between Pb2+ and HF is 12.4. (Ksp, PbF2 = 6.76 x 10-4 and dissociation constant of HF is 3.7 x 10-8)Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.complete this reaction Fe³= ........agent (from-to) S2O3= .........agrnt (from-to).
- Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g . Calculate the percentage yield Week 2 -》 Formation of the tlide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxwaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g .Calculate the percentage yield Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. Calculate the percentage yieldChoose the answer that best represents the products of the following reaction. Potassium carbonate is reacted with phosphoric acid. A. 2 K3PO4 (aq) + 3 H2O (l) + 3 CO2 (g) B. K2CO3 + H3PO4 C. 3 K2CO3 + 2 H3PO4 D. 2 K3PO4 (aq) + 3 H2CO3 (aq)
- The reported melting point for this product is 159-161 degree C. What does a lower melting point (97-100) of the product suggest about the reaction outcome?Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The pKa of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acid
- Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…What is the percent dissociation of 0.150 M triethylamine? (The Kb value for triethylamine is 5.25 × 10-4.) a. 5.92 %b. 5.75 %c. 39.4 %d. 5.75 x 10-30 %Predict and draw the structural formal of the product(s) of each of the reactions below. Butanone+H2 -------> Pt

