Q: Provide the name/chemical formula for the compounds below Ca(NO3)2 * 4 H2O…
A: Answer - 1. Ca(NO3)2 * 4 H2O - The name of the given compound is - Calcium nitrate tetrahydrate…
Q: Write the chemical formulas for the following compounds or ions. a) b) c) d) e) nitrate ion aluminum…
A: We are to write the formulas of given compounds and ions (A) nitrate ion (B) aluminium oxide (C)…
Q: Fill in the gaps in the following table, assuming each columnrepresents a neutral atom. Symbol…
A: 79Br is the symbol of bromine. Its mass number, as represented by the superscript to the left of its…
Q: write the chemical formula for the following compounds g. magnesium phosphide h. potassium sulfide…
A: Chemical formula for the following compounds= g. magnesium phosphide = Mg3P2 h. potassium…
Q: 1) Give the formulas of the following compounds: a) potassium dihydrogen phosphate b) sodium cyanide…
A: We'll answer the first question and up to 3 sub-parts since the exact one wasn't specified. Please…
Q: 23 e 26 р 8 n 28
A: The number of protons is equal to atomic number. The number of electrons in a neutral atom is also…
Q: 3 p*
A: The given ion is having 3 protons, 2 electrons and 4 neutrons. The atomic number of an element is…
Q: Write the chemical formulas for the following compounds:
A: A chemical formula is defined as an expression which shows the elements present in a compound and…
Q: Determine whether each statement is true or false. If false,correct it. (a) The Fe2+ ion contains 29…
A: An atom is primarily composed of three subatomic particles. They are electron, proton and neutron.…
Q: Write the name of the following elements, ions, or compounds: (please answer all questions in order)…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Does each chemical formula represent an element or a compound? a. H 2 b. H 2O 2 c. S 8 d. Na 2CO 3…
A: An element is made up of same type of atom whereas a compound is made up of more than 1 type of…
Q: Write the symbol for every chemical element that has atomic number less than 58 and atomic mass…
A:
Q: Fill in the gaps in the following table, assuming each columnrepresents a neutral atom. Symbol…
A: Table:
Q: Write the formulas of the following compounds:(a) chlorine dioxide(b) dinitrogen tetraoxide(c)…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Write the formulas for the attached
A: Since this question contains multiple subparts so i am solving only first three. Please repost the…
Q: Which of the following ionic compounds is incorrectly named? O Fe2O3, diiron oxide O Zn(NO3)2, zinc…
A:
Q: Write the correct chemical name for each compound.a. NaIb. Fe(N O 3 ) 3c. Sr(OH ) 2d. CoC l 2e.…
A:
Q: Name the following compounds or formulas: Titanium (V) chloride. Trinitrogen Pentoxide. Ag2SO3.…
A:
Q: 1. Using only the periodic table, write the formula and charge for the typical ion of each of the…
A: As per multiple question rule, I am explaining question 1 only.
Q: 3) Give the formulas for the following compounds: a) iron (III) oxide b) strontium carbonate c)…
A: Note : As per our guidelines we are supposed to answer first 3 parts. Please repost other parts as…
Q: Fill in the gaps in the following table, assuming each column represents a neutral atom: 1 3 4 112…
A: For an atom, Mass number = no.of protons + no.of neutrons Atomic number = no.of protons No.of…
Q: When radium becomes an ion, what is its charge? O There is not enough information to answer this…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 17. Zinc dichromate Mg(SCN). Ca(HCO;)z Fe ls 18. Magnesium thiocyanate 19. Calcium bicarbonate 20.…
A:
Q: Fill in the gaps in the following table,assuming each column represent a neutral atoms. Symbols…
A: Given table: Symbol 65 Zn Protons 38 38 92 Neutrons 58 58 49 Electrons…
Q: Fill in the table for the following atoms or ions. Please pay careful attention to the difference…
A: Complete the following table ---
Q: Write the names or chemical formulas of given compounds; Mg(HCO3)2, KI, Iron(III)chloride, nitric…
A: the solution is as follows:
Q: Fill in the gaps in the following table, assuming each columnrepresents a neutral atom.
A: Atomic Number of an element (Z) =total number of proton present in the nucleus…
Q: In which of the following are the name and formula correctly paired? manganese(II) chlorate:…
A:
Q: 66. Write the formula for each of the following compounds: a. zinc chloride b. tin(IV) fluoride C.…
A: We know that Molecular formula : molecular formula consists of the chemical symbols for the…
Q: Provide the symbol of the following monoatomic ions, given the number of protons and electrons in…
A:
Q: 72.691g 84.321g FeSO4 + Potassium phosphate à Iron (II) phosphate + K2SO4 Write…
A:
Q: Write formulas of the two ions in each compound and then write the formula of the compound. a)…
A: A question about writing the formula for ions of salt, which is to be accomplished.
Q: (c) Complete the following table: Name(s) of lon(s) formed during ionization Pd2+ Pd4+ magnesium…
A: Given incomplete table is : Complete the given table = ?
Q: Phosphorus-32 is often used to image biological tissue. Mark any/all that apply to phosphorus-32. It…
A: Phosphorus -32 , means molar mass of phosphorus is 32 .and molar mass is written at the top of…
Q: Which of the following elements fo Gold O Fluorine Hydrogen Sulfur Oxygen O Helium Olodine Antimony…
A:
Q: Give the name and formula for the acid derived from each ofthe following anions:(a) hydrogen…
A: Give the name and formula for the acid derived from each ofthe following anions:(a) hydrogen…
Q: Which of the following elements is not a representative element? O Nickel Cadmium Silicon O Barium
A: I A, IIA, IIIA, IV A, VA, VI A, VII A, VIII A elements are called representative elements. Between…
Q: Here are the colour-coded scale models of 10 types of atoms. Hydrogen (H) Nitrogen (N) Fluorine (F)…
A:
Q: 3 e 3 p* 4.
A: According to the question, the ion is having 3 electrons, 3 protons and 4 neutrons. We need to…
Q: Write the formulas for the following chemical compounds A) ammonium sulfate B) cobalt(III) C) carbon…
A: Compound is a material form by chemical bonding of two or more elements.
Q: The isotope of an unknown element, X, has a mass number of 79. The most stable ion of the isotope…
A: Mass number of an isotope represents the sum of protons and neutrons in its nucleus. In an atom,…
Q: Fill in the gaps in the following table, assuming each columnrepresents a neutral atom.
A: Given,
Q: 2.89 Give the systematic names for the formulas or the formulas for the names: (a) CoO; (b)…
A: Systematic name: It is the nomenclature of a chemical compound to describe the structure of a…
Q: Write the formulas of the following compounds:(a) rubidium bromide(b) magnesium selenide(c) sodium…
A: For ionic compounds, identify the cation and the anion, along with the charge, then these 2 must be…
Q: Be sure to answer all parts. For each neutral atom, fill in the blanks in the table below. 121…
A: The given table is filled by using the following two expressions: Atomic number of the element =…
Q: Write the formula for each of the following binary compounds: a) silver (I) oxide b) aluminum…
A: According to Bartleby questions and answers guidelines we are supposed to solve only the three sub…
Q: Potassium ion Copper (I) ion Aluminum ion
A: Since you have posted a question with sub-parts , we are entitled to answer the first three…
Q: Name the following ionic compounds: (d) CaSO3 (e) Cu(OH)2 (f) Fe(NO3)2
A: Ionic compound are formed by donating and accepting of electrons between cation and anions, Ionic…
Express your answer as a chemical symbol or as an ion.
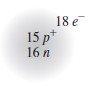
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


