18 g of unknown organic sample was dissolve in 758 mL of Dicloromethane (DCM). The boiling point of benzene was increased by 2.29°C. Determine the molecular weight of the unknown sample? Kb of DCM = 2.42°C/m Bb of benzene = 39.6 °C density of benzene = 1.33 g/mL at 25 °C Round your answer to the nearest whole number, no units required.
18 g of unknown organic sample was dissolve in 758 mL of Dicloromethane (DCM). The boiling point of benzene was increased by 2.29°C. Determine the molecular weight of the unknown sample? Kb of DCM = 2.42°C/m Bb of benzene = 39.6 °C density of benzene = 1.33 g/mL at 25 °C Round your answer to the nearest whole number, no units required.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 40E: In lab you need to prepare at least 100 mL of each of the following solutions. Explain how you would...
Related questions
Question
Transcribed Image Text:Question 1
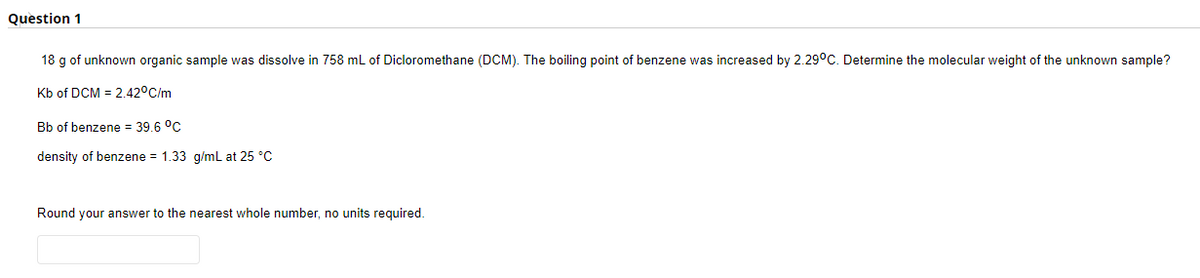
18 g of unknown organic sample was dissolve in 758 mL of Dicloromethane (DCM). The boiling point of benzene was increased by 2.29°C. Determine the molecular weight of the unknown sample?
Kb of DCM = 2.42°C/m
Bb of benzene = 39.6 °C
density of benzene = 1.33 g/mL at 25 °C
Round your answer to the nearest whole number, no units required.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
