Q: 3. Which of the following species are capable of hydrogen-bonding among themselves? (a) C2H6, (b)…
A: For hydrogen bond to be formed, compound should have N,O or F and there should be H attached to it
Q: A certain reaction is first order in N₂ and second order in H₂. Use this information to complete the…
A:
Q: Incorrect. Predict the major product(s) that are expected when the following compound is heated with…
A: In this question, HBr is concentrated that means it is in excess amount so that you will get…
Q: Glassware/Tool Function/Uses Do’s Donts 1. Test Tube Brush 2. Test Tube Rack…
A: These are laboratory equipment commonly used in chemistry experiments: Test Tube Brush: a small…
Q: A. Write the Xnotation for each atomic depiction: (a) Z 18e 18p* 20nº (b) 25e 25p* 30n (c) 47e 47p*…
A:
Q: Consider solutions of Cr(NO3)3-9H2O in water. The solubility of Cr(NO3)3-9H₂O in water at 15 °C is…
A: Given that the solubility of Cr(NO3)3.9H2O in water is 208 g per 100 g of water at 15oC. Saturated…
Q: Problem 3. The following solvents cannot be used in the preparation of organometallic reagents such…
A: Organometallic reagents are chemical compounds that contain a metal-carbon bond, where the carbon…
Q: Thalidomide exists in two enantiomeric isomers as shown below. ((R) N۰۰۰ ((S) If a solution is…
A: Given that thalidomide exists in two enantiomeric isomers as shown below A solution is prepared by…
Q: 1. Write the name for each of the following compounds: (a) MgO (b) PbO (c) Al2S3 (d) AgBr
A: Since you have asked for question 1 only, we are providing you a solution for it
Q: 4. Write the formulas of the following compounds: (a) phosphorus triiodide (d) diphosphorus…
A: Answer-4 When an atom forms more than one type of compound with the same other element in than for 1…
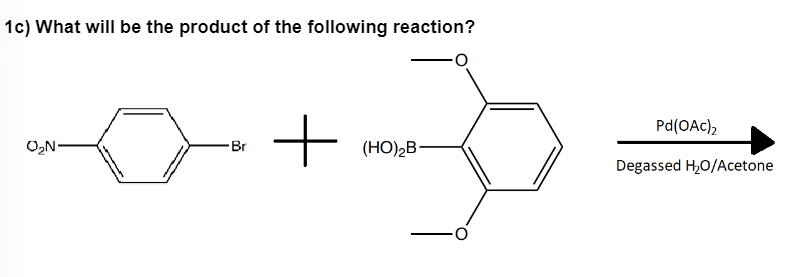
Q: 1h) What will be the product of the following reaction? -Br + (HO)₂B Pd(OAc), Degassed H₂O/Acetone
A:
Q: 3. Identify the hybridization and the approximate bond angles around each atom marked with an arrow…
A: Geometry shape is approximate arrangements of all bond pairs and loan pairs around the atom which…
Q: A certain reaction was found to have a rate constants 7.04x10-3 min-1 at 152°C and 5.54×10-2 min-1…
A:
Q: 2. Draw a Lewis structure and predict the types of bond in each molecule. a) Mg N₂ b) SrC₂ c) POF,…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Name all of the functional groups present in this structure. но. OH
A: Given compound
Q: In this section, describe the compound, ethanol. You must include the following information:…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: In the laboratory a student finds that it takes 71.6 Joules to increase the temperature of 11.6…
A:
Q: For a chemical equilibrium with AS rxn = -12.4 J/K and AH = -5.7 kJ, the equilibrium is spontaneous…
A:
Q: 5. Calculate the amount of heat released when 100.0 g of water at 50.0 °C is converted to ice at…
A: Given data Specific Heat (C) for ice = 2.03 J/g-°C Heat of Fusion for ice, ∆Hfusion = 6.01 KJ/mol…
Q: What is the molar concentration of potassium chloride (Formula Weight: 74.55 g/mole) in a 25% (w/v)…
A:
Q: Which statement best describes the structure that results from the following curved arrow: |- ? O…
A:
Q: A 1.25 L vessel at 23°C contains three gases: N2, H2, and O₂. The system contains 2.20 g of N₂,…
A: The given data is as follows: The volume of vessel (V)= 1.25 L Temperature (T) = 23…
Q: 3:58 Question 12 of 12 Submit The zero order reaction A → Products takes 63.5 minutes for the…
A:
Q: a b с 4. Give, and identify, the 1,2 and 1,4 products of the following reactions. For each circle…
A: Thermodynamic products are more stable than kinetic products. But a reaction to happen via…
Q: Only 1.3x10-6 moles of lead sulfate PbSO4 can be dissolved in 100 mL of a solution of sodium sulfate…
A: The solubility product of PbSO4 is Ksp=[Pb2+] [SO42-]
Q: Problem Statement: Dry ice (solid carbon dioxide) has a density of 1.35 g/mL. If 25.0 mL of dry ice…
A: According to the question, The density of the CO2= 1.35 g/mLThe volume of the CO2 = 25.0 mL Find-…
Q: Balance the equation 35U+n→7+Zr+2n. O Te O Te O 137 50 O 135 15 Sn O None of the above
A:
Q: Which of the following substances is most soluble in H2O? (a) C10H8(s) (b) NH2OH(s) (c) C6H6(l) (d)…
A: This question is related to solubility concept. Here, the rule "like dissolves like" would be…
Q: Perform a confirmational analysis on 2-bromo-3-methylbutane.
A: Given 2-bromo-3-methylbutane
Q: Can u help me with the question 2 for wrtie the fomula for each of the following ionic compounds:…
A: Given : name of compounds
Q: Find the uncertainty in the position of an electron when the uncertainty in velocity is equal to…
A: The uncertainty in the position of an electron, Δx, and the uncertainty in velocity, Δv, are related…
Q: Which drawing best represents the resonance hybrid for the given molecule? H₂C-C=N 8- H₂C----C=N 8 8…
A: In the drawing of the resonance hybrid, bond breaking and bond forming denoted by the doted line.
Q: If 0.480 mol of N₂ (g) and 1.60 mol of H₂ (g) were placed in a 4.50 L container and underwent the…
A: Expression for the equilibrium constant for the given reaction is as follows: N2g + 3H2g ⇌ 2NH3gKc…
Q: This graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure,…
A: We have find out the answer.
Q: On a clear day at sea level, with a temperature of 25 °C the partial pressure of N2 in air is 0.78…
A:
Q: HO 17.8 Give the product. LOH Na₂Cr₂O7
A: we have to determine the product of the given reaction
Q: Where did you get the 1.6*10^-19 and the 9.1*10^-31?
A: This question is related to atoms. There are mainly three subparticles in an atom- Electron Proton…
Q: A nuclear stress test utilizes a gamma-emitting radioisotope such as thallium-201 to follow the flow…
A:
Q: The 50:50 (by mass) mix of water and ethylene glycol (antifreeze) contains 16 moles of ethylene…
A:
Q: 4- Predict the product(s) and show the type of the mechanism for each reaction below. (a)…
A:
Q: Which structure is consistent with this ¹3CNMR spectrum? Feld In S S 60 40 20 0 200 180 160 140 120…
A: Number of signals in 13C NMR = Number of sets of carbons in different electronic environment
Q: Can you help me with the explanation of the binary covalent (molecular) compound, ionic compound and…
A: Introduction Chemical bonding is the process of forming bonds between two atoms or molecules in…
Q: Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid…
A:
Q: Calculate the osmotic pressure (in mmHg) of a solution that is made from 45.9 g of a 1:1 electrolyte…
A: According to the question, The mass of the solute = m = 45.9 g The molar mass of the solute…
Q: Calculate all the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide magnesium…
A: We have find out the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide.
Q: The decomposition of ammonia, NH3, is zero order. The rate constant for this reaction at a…
A: The given data is as follows: Rate constant for zero order reaction (k) = 5.32×10-3 molL-1s-1…
Q: What mass of Na2SO4 is necessary to fully react with 25 L of .01M of PbCl2 solution?…
A:
Q: Name the orbital overlap for:
A: Given : structure of molecule Tip : first decide the hybridization of each atom
Q: The percent by mass of ethylene glycolr (MM = 58.08 g/mol) in an aqueous solution is 10.42% and the…
A:
Q: What is the theoretical yield of Li,N in grams when 12.3 g of Li is heated with 33.6 g of N₂? Round…
A:
Step by step
Solved in 2 steps with 1 images

- Predict the products of the following reaction. Co(s) + xsF2(g)---->[Rh(PPh3)3Cl] is a precatalyst that can be activated by dissociation of a phosphine ligand to form an active catalyst, B, which is used in the hydrogenation of alkenes. The active catalyst can then undergo oxidative addition in the presence of H2 to form complex C. Propene coordinates to C to form complex D, which then undergoes a 1,2- insertion step to form E. Reductive elimination of propane from E regenerates the active catalyst B. Draw complexes B – E and hence provide a full catalytic cycle, including the activation step, for the hydrogenation of propeneWhat is the best reagent for obtaining cis alkene * Pn (Platinum) Ni(nickel) metallic sodium to liquid ammonia Quinoline impregnated Pt catalysts, barium su lfate in methanol nah

