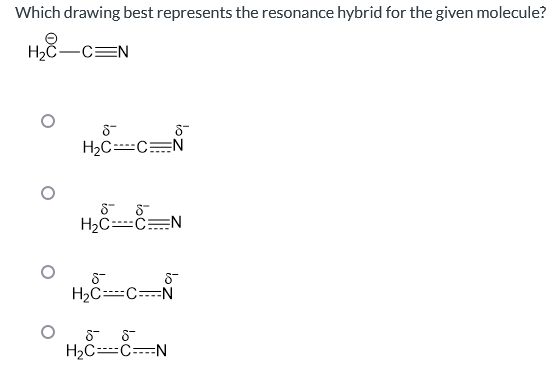
Which drawing best represents the resonance hybrid for the given molecule? H₂C-C=N 8- H₂C----C=N 8 8 H₂C----CN 8 8™ H₂C----C----N 88 H₂C----C----N
Q: In the reaction: 6H+ + 61¯ + CIO3¯ → 312 + 3H₂O + Cl¯ The oxidizing agent is OH+ 12 r CIO3
A: In this question, we will see which is oxidizing agent. Oxidizing agent are those agent which…
Q: Standardization of Sodium Hydroxide (NaOH) Solution Mass of KHP used (g) Initial buret reading (mL)…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 12. Consider the reaction 4PH3(g) →→→ P4(g) + 6H₂(g) - If, in a certain experiment, over a specific…
A: Given -> 4PH3(g) ---> P4(g) + 6H2(g) Moles of PH3 consumed = 0.0048 mole Volume= 2.0 L
Q: We mixed some ethylene glycol with 346 g water to reduce the equilibrium vapor pressure of water…
A: Answer: When a non-volatile solute is added in a solvent it causes a depression in freezing point…
Q: 8. Give the major organic product(s) of each of the following reactions. If none is predicted, write…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: In Part C of the experiment, you added a 6.0 NaOH solution to the aqueous solution of NaCl. Did the…
A: When NaCl is dissolved in water , it undergoes dissociation to form sodium and chloride ions.…
Q: 10 10 40 8 60 18 8 30 of Solute (g)
A: In this graph, the pattern shows the amount of solute (i.e., salts and gases) required to make a…
Q: When solid ammonium chloride is added to water, all of the solid dissolves and the solution becomes…
A: According to the answering guidelines, I'm posting the solution for the first question. Kindly post…
Q: Do the structures in each pair represent the same compound or constitutional isomers? Set 1 Set 2
A: we have to determine if the given structures are identical or constitutional isomers
Q: 10. Among the following anions, which one could not exist (to any significant extent) in water? B)…
A: Answer: When an acid loses its H+ ion then the specie formed is its conjugate base and it has the…
Q: trans-2-butene 233.11 IM Angle of Rotation Energy (kJ/mol) 26.92 butane 78.49 Energy (kJ/mol) 20.43…
A: Isomers which are formed by the rotation about carbon - carbon (single bond) are called…
Q: Silicon tetrachloride (SICI) can be prepared by heating Si in chlorine gas: Si(s) + 2Cl₂(g) SICI, ()…
A: Given : chemical reaction SiCl4 produced = 0.594 mol
Q: 3. For each of the following symbols, clearly specify the number of protons, the number of neutrons,…
A: ->Number of proton + Number of neutron = Mass number -> Number of proton = Number of electron…
Q: Consider the following rate law: Rate = K[B] Which of the following equations could be used to find…
A: As we know the sum of the powers of the concentration terms occur in the rate law expression is…
Q: What is the correct IUPAC name of the following compound? H H CH₂CH3 CH₂ Multiple Choice…
A: Introduction IUPAC nomenclature is a system of naming chemical compounds and identifying the…
Q: What differentiates the Group 3A Cations from the Group 3B cations How do we prevent the…
A: Qualitative analysis is a branch of analytical chemistry that is concerned with the identification…
Q: . Draw the bond line structure for each compound (a. and b.) below 2. What are the fragment ions…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Determine which of the following alkyl halides will react faster in an E2 reaction with NaiPr and…
A: The E2 reaction is the elimination reaction and it involves the conversion of an alkyl halide into…
Q: A C 1.2 % NaCl 280 mOsm/L B D 5% Glucose 2% Glucose Assuming equal volume in all the sections, what…
A: Here it is given that the volume of all the sections are same. So A, B, C and D have se volume. We…
Q: If for the reaction, ax + bY --> products, the rate law is determined to be r=K[X]¹[Y then the order…
A: Given: r = [X]1[Y]2 To determine order of the reaction
Q: Una estructura resonante posible para la siguiente molécula es: a. b. C. NH₂ NH₂ +NH₂ + 1 :NH₂ :NH₂
A: In this question, we will identify the possibile resonance structure of the given compound. You can…
Q: * Question Completion Status! Shown below is a calibration curve for the analysis of theobromine and…
A: Given,A calibration curve : Peak height centimeters Vs Analyte concentration parts per…
Q: A possible resonant structure for the following molecule is: a. b. C. d. NH₂ + NH₂ +NH₂ + | : NH₂…
A: Introduction to Resonance Resonance is an important concept in chemistry that is used to explain the…
Q: A compound has the molecular formula: C4H8O2 and gives the following IR spectrum. What is the most…
A: We have find out the compound from given IR spectrum.
Q: 1. Write the name for each of the following compounds: (a) MgO (b) PbO (c) Al2S3 (d) AgBr
A: Since you have asked for question 1 only, we are providing you a solution for it
Q: Select all of the true statements about melting points. a. The melting point range of a pure…
A: The melting point of a substance is the temperature at which it changes from a solid to a liquid…
Q: 1. Provide the condensed electron configuration for the ion of Ga which loses the fewest number of…
A: Given : Ga atom
Q: See Figure 3-3. Match each formula to its number of units of unsaturation. A B U D ✓ [Choose ] 0 2 5…
A: We have to calculate the units of unsaturation for the given formula
Q: 5. Helium on the Moon was found to be 0.520% 2He, 8.25% ³He, and 91.23% 4He. What is the average…
A: average atomic mass = ∑(mass of its isotope× natural abundance )
Q: Consider a solution is prepared by dissolving 34.0 g of NH3 in 500.0 g of water. The density of the…
A:
Q: Determine whether each of the amino acids is polar, nonpolar, positively charged, or negatively…
A:
Q: What is the correct name of the following aromatic compound? (the abbreviations at the beginning of…
A:
Q: The percent by mass of ethylene glycolr (MM = 58.08 g/mol) in an aqueous solution is 10.42% and the…
A:
Q: Suppose you performed a similar experiment using cyclohexane as your solvent. Given the data in the…
A: Answer: When a non-volatile solute is added in a solvent, it causes a depression in its freezing…
Q: What is the average kinetic energy (in kJ mol-¹) of a hypothetical gas with molecular weight 52.6 g…
A: Please find your solution below : The average kinetic energy of a gas is directly proportional to…
Q: Can you help me with last ones? I have to identify the compound of each compound and its type?
A: Molecular compound: These compounds contain discrete molecules, which are held together by sharing…
Q: A mass spectrum of an unknown compound shows an [M]+ peak with an [M+2]* peak of similar height.…
A: Given that The mass spectrum of an unknown compound shows an [M]+ peak with an [M+2] peak of…
Q: 10. Which compound would have the highest boiling point? X II OH - ОН III CH₂OH IV V OH
A: There are different type of compounds with different physical and chemical properties. (I) This…
Q: Calculate the boiling point (in °C of a solution that is made from 48.6 g of a nonelectrolyte (M =…
A: Given data The mass of nonelectrolyte solute = 48.6 g Molar mass of nonelectrolyte solute =…
Q: Acetaminophen (pictured) is a popular nonaspirin, "over-the-counter" pain reliever. What is the…
A: Mass percentage (mass %) is a measure of the concentration of a substance in a mixture. It is the…
Q: An exothermic reaction is one where none of these heat is transferred from a system into the…
A: we have to select the correct statement for an exothermic reaction
Q: Show what Grignard reagent and what carbonyl compound you would start with to prepare the alcohol…
A: Grignard reagent is an alkyl magnesium halide. The general formula of Grignard reagent is RMgX. R =…
Q: 9. Propose syntheses to carry out each of the following conversions. Assume ortho and para isomers…
A: Organic compound can prepared from different starting material and reagent under different…
Q: 15. Compound A, a hydrocarbon with M* = 96 in its mass spectrum, has the ¹³C spectral data given…
A: Introduction Nuclei in a magnetic field can absorb electromagnetic radiation and then release it…
Q: 1. pH=7.31 PCO2-51 HCO3=27 What is the imbalance and is there compensation (if so, what type)? 2.…
A:
Q: Q4/ Calculate the heating value per pound of a dry base for a sample of anthracite with the…
A: We have find out the answer.
Q: ATOM Zinc (Zn) Platinum (Pt) Lithium (Li) Lead (Pb) Anode Silver (Ag) For which component of the…
A: Answer: Galvanic cell is the type of cell in which chemical energy is get converted into electrical…
Q: A liquid is exposed to infrared radiation with a wavelength of 2.43 × 10-4 cm. Assume that all the…
A: Answer : Given that, Wavelength =2.43 x 10-4 cm =2.43 x 10-6 m Energy= 30.03 J
Q: For the equilibrium system below, which of the following would result in an increase in the quantity…
A: we have to select the change that results in increase in amount of PCl5
Q: A 21 L reaction vessel contains only He gas. The system is kept at 55 °C and the pressure inside the…
A: Given : volume = 21 L, temperature= 55°C Pressure= 0.495 atm Tip : PV = nRT
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Which molecules are polar? For each that is polar, specify the direction of its dipole moment. Q.) CH2Cl2Determine the polarity of the following molecules. If polar, indicate the partially negative end. CH4 BF3 H2S Kindly explain.ANSWER ALL THE GIVEN QUESTION, CHOOSE THE CORRECT NUMBER CORRESPONDING TO THE RIGHT ANSWER PLEASE Formed when the electron being shared come only from one of the elements. coordinate covalent bond pure covalent bond polar covalent bond non-polar covalent bond The hybrid orbital which has one fourth s character and three fourths p character. sp sp2 sp2 special sp3 The number of sigma bonds in sp2 hybridized carbon. 1 2 3 4 Hybridization of oxygen in furan. sp sp2 sp2 special sp3 The number of sigma bonds in an sp2 hybridized oxygen. 1 2 3 4 He believed that matter is continuous and can be divided into smaller particles John Dalton Aristotle Democritus Leucippus Which of the following factors does not affect bond energy? bond polarity orbital hybridization formal charge loss of overlap Calculate the formal charge of sulfur in sulfuric acid 0 -2 2 6 Bond angle between two sp3 hybridized orbitals. 60 degrees 5 degrees 120 degrees 180 degrees Pi bonds can be formed by…
- Answer the following questions about amoxicillin, an antibiotic from the penicillin family. a. Predict the hybridization and geometry around each highlighted atom. b. Label five polar bonds using the symbols δ+ and δ–. c. How many π bonds does amoxicillin have? Label them. d. Find a C – H bond containing a carbon atom having a hybrid orbital with 33% s-characterHow do I go about drawing an orbital diagram and hybridizing:CH3-CH (triple bonded to) CH-C (triple bonded to) C- CHOI'm completely lost.most polar. A. C-C B. C-F C. F-F D. C-N E. C-O
- How many Cs are sp2-hybridized in the molecule shown below? (kindly show explanation or illustration. thank you!) Choices: A. 6B. 8C. 10D. 9Q/Choose the correct answer 1-In some case of hybridization, electron is transfered from one shell to other. * To stabilize the atom To saturate the orbital To make the atom to be excited Non of these To supply unpair electronIdentify the dipole moment for each of the following compound, explain your answersbriefly.(i) H2CO(ii) C2H3Br(iii) Cl2CH2
- Indicate the hybridization number of the numbered carbon atom. 1-10Please indicate the orbital hybridization that exists for every carbon in the following molecules: O O ║ ║ (a) H2C=CH-CH2-C-CH3 (b) CH≡C-CH2-C-OH- What orbital(s) are the lone pairs of electrons on the O atom placed? - What orbital is the lone pair of electrons on the N atom placed? - What type of bond(s) are formed between Ca1 – C? - What type of bond(s) are formed between C = O? - What type of bond(s) are formed between C – N? - What type of bond(s) are formed between N – Ca2? - What type of bond(s) are formed between N – H? - What is the angle between Ca1 – C = O? - What is the angle between Ca1 – C – N? - What is the angle between O = C – N? - What is the angle between C – N – Ca2?



