Q: Many drugs are Brønsted–Lowry acids or bases.a.What is the most acidic proton in the analgesic ibupr...
A: A) In the compound: The most acidic Hydrogen is the hydrogen in - COOH group. The hydrogen ...
Q: 4.Indicate whether the following solutions are acidic (A), basic (B), or neutral (N). (a)pH 8.6 (c) ...
A: (a) pH = 8.6 The nature of solution can be determined from the pH scale as: pH scale ranges from...
Q: How many hydrogens are present around each carbon atom in the following molecules?
A: Given structure is,
Q: The Ksp for LaF3 is 2 x 10-19. What is the solubility of LaF3 inwater in moles per liter?
A: The solubility of LaF3 in water in moles per liter can be determined as follows,
Q: You have studied the gas-phase oxidation of HBr by O2:4 HBr(g) + O2(g) -->2 H2O(g) + 2 Br2(g) You...
A: For the given overall reaction: 4 HBr(g) + O2(g) -->2 H2O(g) + 2 Br2(g) The reaction is first ord...
Q: 1. A tetradecapeptide (14 amino acid residues) gives the following peptide fragments on partial hydr...
A: So on partial hydrolysis, we will get separate amino acids peptide chain To get our main chain we ...
Q: Draw the products of each reaction.
A:
Q: Caffeic acid is an organic acid isolated from coffee beans. Predict which labeled hydrogen (Ha or Hb...
A: The carbonyl group has the -R-effect (electron withdrawing effect) by which it withdraws the electro...
Q: Give the IUPAC name for each compound.
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: How many electronic spectral bands for Ni2+ and Co2+ , one could be expected. Assign these electroni...
A: The number of electronic spectral bands depends upon the number of excitation of an electron to the ...
Q: See question 4.
A: The number of mercury atoms that is consumed by drinking 1 L of water that contained 1 µg of mercury...
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction:I2(s) + Cu(...
A: In the given reaction, I2 is being reduced and Cu is being oxidised hence the half reactions are Ox...
Q: Rank the following compounds in order of decreasing frequency of the carbon–oxygen double-bond stret...
A:
Q: Acid-catalyzed bromination of pentan-2-one (CH3COCH2CH2CH3) forms two products: BrCH2COCH2CH2CH3 (A)...
A: Acid-catalyzed bromination means the addition of the bromine to a compound in the presence of an aci...
Q: At 1 atm, how much energy is required to heat 47.0g H2O(s) at -18.0 degrees Celcius to H2O (g) at 17...
A: Energy required
Q: Treatment of 2,2-dibromobutane with two equivalents of strong base affords but-1-yne and but-2-yne, ...
A: Since in the mechanism of elimination reaction, the NaNH2 base extracts a proton from the compound. ...
Q: (a) Calculate the mass percentage of Na2SO4 in a solutioncontaining 10.6 g of Na2SO4 in 483 g of wat...
A: The concentration of the species the in term of (ppm) is expressed as a relation between the solute ...
Q: Indicate whether each statement is true or false:(a) The higher the temperature, the more soluble mo...
A: As the temperature of an aqueous solution that contains a gas, enough energy is provided to the gas ...
Q: The answer is supposed to be in grams
A: The balanced chemical reaction is as follows:
Q: Draw a stepwise, detailed mechanism for the following intramolecular reaction.
A: The alkene present outside the benzene ring will take proton from the acid and form a carbocation. A...
Q: Which solution has the higher pH, a 0.001 M solution of NaOH or a 0.001 Msolution of Ba(OH)2?
A: The simplification is as follows:
Q: After an afternoon party, a small cooler full of ice is dumped onto the hot ground and melts. If the...
A:
Q: How can the following compounds be prepared from the given starting materials?
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: ●The standard heat of reaction for the oxidation of ammonia is given below: 4 NH3 (g) + 5 O2 (g) ...
A: Since from the reaction we can see that 4 mole of NH3 is reacting with 5 moles of O2 Hence moles of...
Q: four beakers containing potassium nitrate dissolved in water are allowed to evaporate to dryness. be...
A: Moles: One mole is equivalent to the mass of the substance consists same number of units equal to th...
Q: How many hydrogens are present around each carbon atom in the following molecules?
A: First structure hydrogen attached to carbon is showing by the diagram below is: For carbon atom num...
Q: (a) In which of the following reactions would you expectthe orientation factor to be least important...
A: Orientation factor gives the knowledge about collisions which occur in proper orientation which caus...
Q: Several factors can affect alkene stability. Explain why alkene A is more stable than alkene B even ...
A: Alkene stability is decided by Saytzeff’s rule which says that more substituted alkene is more stabl...
Q: An aqueous solution of a primary or secondary amine reacts with an acyl chloride to form an amide as...
A:
Q: Does the term ‘labile’ imply that a complex is thermodynamically unstable? Does the term ‘inert’ imp...
A: Henry Taube was the first to classify the compounds in terms of lability . He compared the factors t...
Q: Draw a stepwise mechanism for the following reaction and explain the stereochemistry of the product.
A: The protonation step is shown below.
Q: Menthone and menthol are both isolated from mint. Explain why menthol is a solid at room temperature...
A: Menthone and menthol are both isolated from mint. The reason why menthol is a solid at room temperat...
Q: As shown in Table , the equilibrium constantfor the react ion N2(g) + 3 H2(g) ⇌ 2 NH3(g) i sKp = 4.3...
A: GIVEN: Kp = 4.34 x 10-3 at 300 °C volume of flask is 1L Mass of NH3 in the equilibrium mixture=1.05 ...
Q: What cyanohydrin and carbonyl compound are formed when amygdalin is metabolized in a similar manner ...
A: The derivative compound of cyanohydrin is amygdalin. The metabolism of amygdalin involves 2 steps. T...
Q: Many metallic catalysts, particularly the precious-metalones, are often deposited as very thin films...
A: a.
Q: Answer the following questions about acid-base equilibrium reactions (d)Based on the following Kaval...
A: A strong acid shows complete dissociation to respective anion and H3O+whereas a weak acid can only p...
Q: Consider the following equilibr ium, for whichKp = 0.0752 at 480 °C:2 Cl2(g) + 2 H2O(g) ⇌ 4 HCl(g) +...
A:
Q: You study the rate of a reaction, measuring both the concentrationof the reactant and the concentrat...
A: The concentration of B is decreasing with time and A is increasing with time. Thus B must be the rea...
Q: Muscalure, the sex pheromone of the common housefly, can be prepared by a reaction sequence that use...
A: Since NaH is a base. hence it takes out the acidic proton from the molecule making an anion. And sin...
Q: find the empirical formula of the compound that contains 15.8% Al, 28.1% S, and 56.1% O.
A: The number of moles for a given compound or a molecule refers to the ratio of its mass to the molecu...
Q: Ammonium phosphate ((NH4),PO4) (NH3). is an important ingredient in many fertilizers. It can be made...
A: The balanced reaction for the production is H3PO4 + 3 NH3 -----> (NH4)3PO4 Hence from the above...
Q: What product is formed by ring-closing metathesis of compound V, a key intermediate in the synthesis...
A: The ring-closing metathesis (RCM) by Grubbs catalyst occurs, when the starting material is diene. Th...
Q: Explain why heats of hydrogenation cannot be used to determine the relative stability of 2-methylpen...
A: The process in which a C-C double bond breaks along with a H-H bond and results in the formation of ...
Q: List the following aqueous solutions in order of decreasingfreezing point: 0.040 m glycerin (C3H8O3)...
A: The following aqueous solutions are to be arranged in order of decreasing freezing point: 040 m gly...
Q: DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene, is a base we will encounter in elimination reactions in Cha...
A: The 2 N atoms present in DBU is marked A and B as shown below for further discussion
Q: Outline two different ways that butan-2-one can be prepared from a nitrile and a Grignard reagent.
A: The two different ways that butan-2-one can be prepared from a nitrile and a Grignard reagent are 1...
Q: Convert each of the following structures to its more stable chair form. One structure represents men...
A: Chair isomer for menthol is shown below.
Q: Methane, CH4, reacts with I2 according to the reactionCH4(g) + I2(g) ⇌ CH3I(g) + HI(g). At 630 K, Kp...
A: The expression for pressure initially and at equilibrium for different species in the given reaction...
Q: The bond energy in N2 is 941 kJ/mol. What is the longest/wavelength a photon can have and still have...
A:
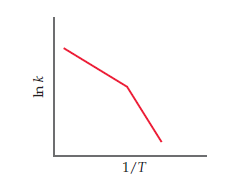
You obtain kinetic data for a reaction at a set of different
temperatures. You plot ln k versus 1/T and obtain the following
graph:
Suggest a molecular-level interpretation of these unusual
data.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


