Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.29QAP
Related questions
Question
Transcribed Image Text:A::Y ZAA
docs.google.com/form
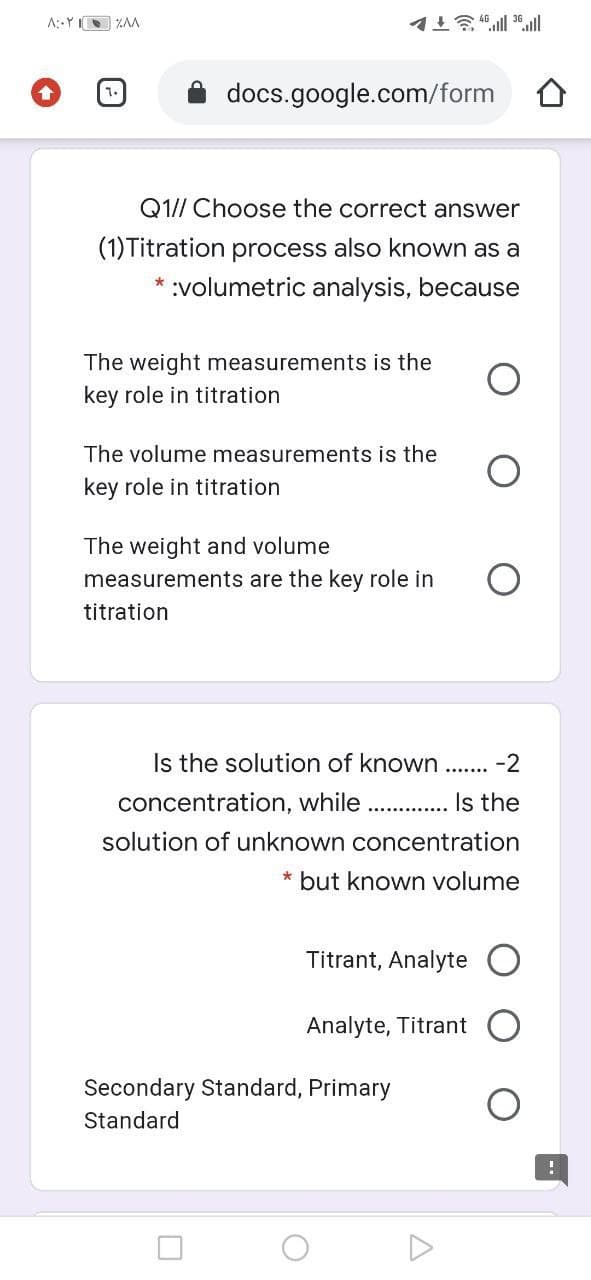
Q1// Choose the correct answer
(1)Titration process also known as a
* :volumetric analysis, because
The weight measurements is the
key role in titration
The volume measurements is the
key role in titration
The weight and volume
measurements are the key role in
titration
Is the solution of known
-2
.......
concentration, while
Is the
solution of unknown concentration
* but known volume
Titrant, Analyte
Analyte, Titrant
Secondary Standard, Primary
Standard
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning



Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning
