Q: (Q84) With respect to the following unbalanced redox equation that takes place in a basic…
A: The given unbalanced redox reaction is: Zn(s)+NO3-aq→ZnOH42-aq+NH3g
Q: Consider the titration of 30.0 mL of 0.20M nitrous acid by adding 0.0500 M aqueous ammonia to it.…
A:
Q: 7. If the computed Kc of an unknown compound is equal to 1.3, where does chemical equilibrium lie?…
A: The equilibrium constant is the ratio between the conc. of products and the conc. of reactants at…
Q: (Q83) With respect to the following unbalanced redox equation that takes place in an acidic…
A:
Q: 8.37) An electron in the first excited state of hydrogen is struck by a photon of light of…
A: Given, Wavelength of photon = λ = 656.48 nm a). What is the frequency of the photon ? b). What is…
Q: When MoO3 and Zn are heated together they react according to the following unbalanced equation:…
A:
Q: Determine if the synthesis of sulfuric acid is a spontaneous reaction at 25°C. Chemical Equation: S…
A:
Q: The concentration of oxalate ion (C20,2-) in a sample can be determined by titration with a solution…
A: Given: Volume of oxalate solution = 30.00 mL Volume of MnO4- solution = 21.93 mL Molarity of MnO4-…
Q: 1. Melting point technique 2. Recrystallization technique| 3. Extraction (understand how to separate…
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: Write the chemical formula for the ionic compound formed from each pair of ions. (a) Mg2+ and I- (b)…
A:
Q: Question 2 What is being monitored by the titartion curve in the redox reaction? concentrations of…
A:
Q: 2. Calculate reaction AH°rxn and reaction AU°rxn for the total oxidation of benzene. Use the stable…
A: Here we have to find ∆H°rxn and ∆U°rxn of combustion of benzene at 298.15K.
Q: The Ecell of a reaction were found to be +0.78 V. What is the spontaneity of the reaction. none of…
A:
Q: What were the starting materials used in the synthesis of the thermochromic material? a.…
A: In thermochromic material,the starting materials used are diethylamine HCl and CuCl2. Therefore,…
Q: Chemistry draw the structures of soap and identify its components.
A: Soap molecules consists of following two parts.
Q: A 40.00 mL sample of 0.1000 M diprotic malonic acid is titrated with 0.0900 M KOH. What volume KOH…
A:
Q: Which element is in excess?
A:
Q: You have 500.0 mL of a buffer solution containing 0.30 Macetic acid (CH₂COOH) and 0.20 M sodium…
A: Volume of buffer solution = 500 mL = 500 × 10-3 L Molarity of Acetic acid = 0.30 M Molarity of…
Q: Give the Structure and IUPAC name or common name of Amide component of black and white pepper.
A: ✓It has a distinctive pungent flavor due to the presence of an alkaloid piperine. ✓piperidine is the…
Q: Which statements are true for molecular exclusion chromatography?I Molecular exclusion…
A: ✓As the name ,Size-exclusion chromatography, also known as molecular sieve chromatography, is a…
Q: 50. Solve this problem: A gas mixture containing oxygen, nitrogen and water vapor has a total…
A: The total pressure exerted by the gaseous mixture is the sum of the partial pressure of individual…
Q: Give the IUPAC name of this compound.
A:
Q: It was noted that H2O2 and NaOH was added to Cr3+ ion to test. What is responsible for its yellow…
A:
Q: Chemistry The lines in the InP vs. 1/T graph are best-fit lines of plotted data collected from the…
A:
Q: What's More Activity 3.2 Identify and underline the amphoteric substances in this chart. Conjugate,…
A: Given chart is : Identify and underline the amphoteric substances in this chart = ?
Q: A 25.00 mL aliquot of 0.0900 M acetic acid is titrated with a 0.100 M KOH. Calculate the pH when…
A: milli moles of CH3COOH = 25 x 0.09 = 2.25 milli moles of KOH added = 25 x 0.1 = 2.5 2.5 - 2.25 =…
Q: Exercise 6 Ethane burns in air to give carbon dioxide and water according to the following…
A:
Q: A coffee-cup calorimeter contains 30 mL of water with an initial temperature of 24.8°C. An unknown…
A:
Q: Determine if the synthesis of sulfuric acid is a spontaneous reaction at 25°C. Chemical Equation: S…
A:
Q: The oxidation number of Chromium in K2Cr2O7 is? Group of answer choices +7 +5 +6 +12 +2
A:
Q: Which of the following correctly describes the structure of these compounds? CH;CH;OH and CH;OCH;…
A: Here one alcohol and one ether are given.
Q: Calculate the new concentration when 25.00 mL of a 5.0 M stock solutions of CuSO4 is dilated to…
A:
Q: At 25 °C, a 10.0-mL solution of 0.0500 M AGNO3 was titrated with 0.0250 M NaBr in the cell: S.C.E. |…
A:
Q: 1. A dust explosion occurs in a saw mill. a catalytic converter. Cataluct Concentratin 8. Calcium…
A: Those chemical compounds which are reacting together to form the new one are known as the reactants.…
Q: 96. Show the products from these neutralization reactions. Balance the equations if necessary. a.…
A: Here we are required to find the product of the neutralization reaction
Q: Write the splitting pattern of hydrogens in vinyl chloride. Hb Hc На CI (A) Ha = doublet, Hb =…
A:
Q: Draw a ketohexose. Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and…
A: We have to draw a ketohexose.
Q: An unknown sample containing mixed alkali (NaOH, NaHCO3, or NazCO3) was analyzed using the double…
A:
Q: A 1.00 M solution of fluoroacetic acid is 10.0% dissociated in water at a certain temperature. The…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Which is not a property of buffers? Buffers resist changes in pH when acids or bases are added or…
A: Buffers are used to resist change in pH. We we add some amount of acid or base, pH not changes much.…
Q: A 40.00 mL sample of 0.1000 M diprotic malonic acid is titrated with 0.0900 M KOH. What volume KOH…
A:
Q: How Many liters of 1.5 M solution contains 3 moles of solute ?
A:
Q: A solution is prepared at 25 °C that is initially 0.42M in ammonia (NH,), a weak base with K, = 1.8…
A:
Q: exothermic reaction?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 9. Bond of this energy carrier of cells is broken through what?
A: Please find the answer in step 2. Thank you.
Q: Give the reaction mechanism and name the products for the reaction of 2-bromobutane with a hot…
A:
Q: Increasing in temperature: a. H20) = H20) (+AH), b. N2(9) + 3H2(9) 2 2NH3(9) (-AH) c. If the…
A: Given, Predict the direction of the following reactions and defend your predictions using Le…
Q: The end point for a titration can be determined graphically. The end point volume for a first…
A:
Q: When ammonium chloride dissolves in water, the solution becomes cooler. Is the solution process…
A:
Q: What is the volume (in liters) of a .20M solution that contains .30 moles of Na2SO4 dissolved in it?
A:
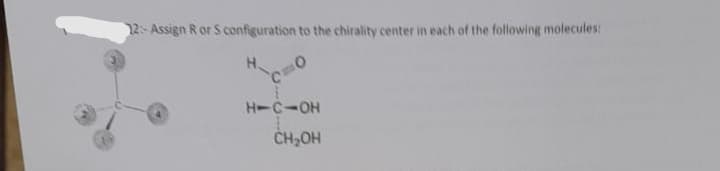
Step by step
Solved in 2 steps with 1 images

- 11. Identify the correct configurations (R or S) of the chirality centers indicated on the Fischer projection shown below.Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the otherWrite the configuration of two chirality centers in 4-chloro-2,3-dimethyl-3-pentanol presented through Fischer projection below in a form of short answer: (3R,4R); (3S,4S); (3R,4S); or (3S,4R).
- Assign R or S configuration to the chirality centers in the molecule below.Assign R or S configuration to the chirality centers in the following moleculesFollowing are stereorepresentations for the four stereoisomers of 3-chloro-2-butanol. (a) Assign an R or S configuration to each chiral center. (b) Which compounds are enantiomers? (c) Which compound are diastereomers?
- A Fischer projection of a molecule with one chiral center represented by a dot is shown below. Determine the R- or S-designation of this chiral center. A) R B) S C) no chiral center3b) (i) Using Fischer projection, draw all the stereoisomers of 3-chloro-2-hydroxypentanoic acid. ii) Assign the chiral centre(s) with R/S configurations in part (b)(i). Indicate which areenantiomers, diastereomers and meso compounds (if any).Draw the Fischer projections of all possible stereoisomers of 2-bromo-3-chlorobutanoic acid: CH3CH(Cl)CH(Br)CO2H. Determine the R/S configuration of each stereocenter. Indicate the relationship between each pair of stereoisomers. Is a meso compound present? Identify it if your answer is yes.
- (b) (i) Using Fischer projection, draw all the stereoisomers of 3-chloro-2-hydroxypentanoic acid. (ii) Assign the chiral centre(s) with R/S configurations in part (b)(i). Indicate which are enantiomers, diastereomers and meso compounds (if any).Assign the absolute configuration of the chirality center as r or s1.What is the absolute configuration of this isomer of 2,3-dihydroxybutanal? pls explain.2. What is the absolute configuration of the enantiomer of the isomer?

