2. Calculate the number of moles of Na2CS3 produced from 0,60 moles of C$2 by the following reaction. 3 CS2 + 6 NAOH → 2 Na2CS3+ N22CO3+ 3 H20 3, How many grams of SIBN4 can be produced from 0.46 moles of N2? 3 Si-2 N2 si3N4 How mahy holes of No dan be produced from 0680 bf NH3 according to the following reaction? NO
2. Calculate the number of moles of Na2CS3 produced from 0,60 moles of C$2 by the following reaction. 3 CS2 + 6 NAOH → 2 Na2CS3+ N22CO3+ 3 H20 3, How many grams of SIBN4 can be produced from 0.46 moles of N2? 3 Si-2 N2 si3N4 How mahy holes of No dan be produced from 0680 bf NH3 according to the following reaction? NO
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter2: Chemical Formulas, Equations, And Reaction Yields
Section: Chapter Questions
Problem 46AP
Related questions
Question
I need help with 2,3 and 4
Transcribed Image Text:No Comments
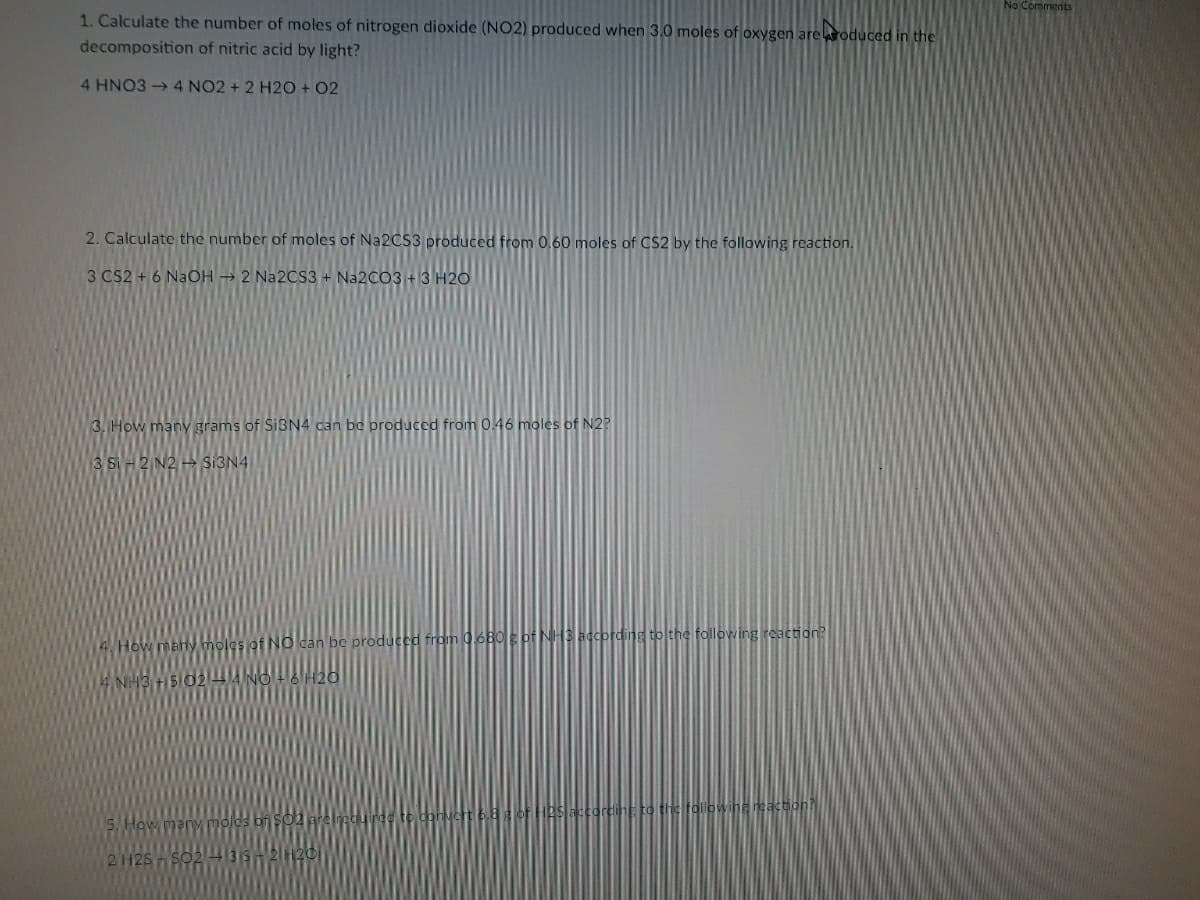
1. Calculate the number of moles of nitrogen dioxide (NO2) produced when 3.0 moles of oxygen areroduced in the
decomposition of nitric acid by light?
4 HNO3 4 NO2 + 2 H2O + 02
2. Calculate the number of moles of NA2CS3 produced from 0,60 moles of CS2 by the following reaction.
3 CS2 + 6 NaOH → 2 N22CS3 + Na2CO3+3 H20
3. How many grams of SIBN4 can be produced from 0.46 moles of N2?
3 Si
SI3N4
4. How
INO can be produced from 06B0E of NH3 according to the following reaction?
NH3
to the followiha nsaction
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning