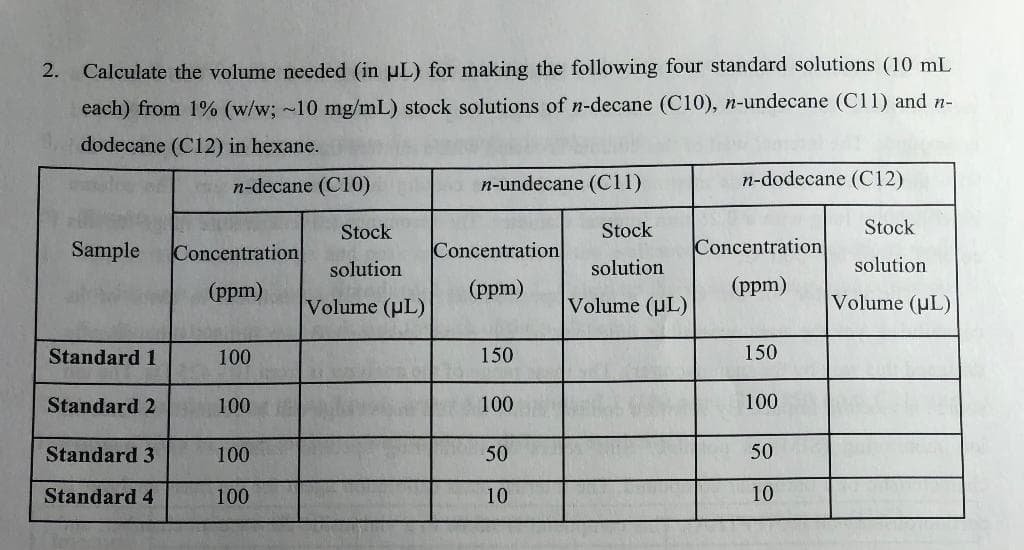
2. Calculate the volume needed (in uL) for making the following four standard solutions (10 mL each) from 1% (w/w; ~10 mg/mL) stock solutions of n-decane (C10), n-undecane (C11) and n- dodecane (C12) in hexane. n-decane (C10) n-undecane (C11) n-dodecane (C12)
Q: How many ml of water do you add to 10ml of glycerol stock if you want to dilute it by a factor of a)…
A: We have to calculate volume of water need to be added to 10 mL of glycerol stock, for the given…
Q: What volume of aztreonam (50 mg/ml) must be added to 500 ml of Mueller-Hinton broth to yield a…
A: Formula used: C1V1=C2V2
Q: The petition coeficiente between benzene and water for carta iLife is approximately 3.9 .you are…
A: The given question employs the Nernst Distribution Law. The partition coefficient between the amount…
Q: Compounds A and B are soluble in water and ether as shown: Solvent Compound A Compound B Water 10…
A: Distribution coefficient is he ratio of solute present in organic phase to the concentration of…
Q: Compute the weight (g) of solute needed to make the solutions listed below: a. 1250 mL of 0.85 M…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: Compounds A and B are soluble in water and ether as shown: Solvent Compound A Compound B Water 10…
A: Distribution coefficient of any compound in two solvents is given by, Kd=concentration in organic…
Q: which of the following statements regarding purification by recrystallization is false? i.…
A: A question based on purification, which is to be accomplished.
Q: Add an excess of methanol to 8.0g of benzoic acid followed by a few drops Cool the mixture, pour…
A: The procedure given in the diagram is the method of preparation of ester (methyl benzoate) from…
Q: (Concentration) You are tasked to prepare 62.5 ppm of a caffeine standard for an analysis. You need…
A: Concentration in ppm=(mass of solute/Mass of solvent) × 10^6 Previously Concentration 0.025gm…
Q: Which of the following would LOWER the purity of the distillate collected from the rubbing alcohol…
A:
Q: The distribution coefficient of an organic compound Y between hexane and water is 4.73. If 192.7…
A: Given Distribution coefficient = 4.73 Concentration of Compound in water = 192.7 mg/L Mass of…
Q: How many mL of solvent do you need to add to the amount gotten from the 2M Palmitic acid stock…
A: concentration of stock solution = 2 M concentration of desired solution = 1 M total volume of…
Q: In 6.75g of distillate >130C. After performing all necessary GC calculations, it is determined that…
A:
Q: Your teacher asks you to dilute 6.0 M HNO3 (M1) to 250. mL (V2) of 1.0M HNO3 (M2). What volume (V2)…
A: Given Molarity(M1) = 6.0 M Volume (V1) = ? Molarity (M2) = 1.0 M Volume (V2) = 250.0 mL
Q: An analyst wanted to separate an essential oil which is partially immiscible in water and a boiling…
A: to separate an essential oil which is partially immiscible in water and a boiling point higher than…
Q: Compounds A and B are soluble in water and ether as shown: Solvent Compound A Compound B Water 10…
A: Given that, compounds A and B are soluble in water and ether, as shown below Solvent Compound A…
Q: Compounds A and B are soluble in water and ether as shown: Solvent Compound A Compound B Water 10…
A:
Q: - How many grams of sodium hydroxide (M.wt=58.5) can be added to 2.5 liter of a solution mixture of…
A: Given a buffer solution consisting of mixture of 0.3M ammonia and 0.15M ammonium chloride.
Q: You want to make a solution of loading dye with a final concentration of 0.35% bromophenol blue,…
A: Part 1 0.35 % Bromophenol blue = 0.35% * 60 ml = 0.21 ml = 0.21 grams 0.15 % Xylene Cyanol = 0.15%…
Q: B: Similar to Part A, using 97 mL of an aqueous solution containing 7.22 g of the compound with a KD…
A: w = 7.22 g v = 97 ml portion = 41 KD = 5 n= 2
Q: Peak Solution Preparation Area 0.045 g of paracetamol is weighed in a 50 mL volumetric flask and…
A: In the equation of calibration curve y is the peak area and x is concentration of paracetamol…
Q: How would you correctly prepare 125.00 ml of a 0.300M solution of glucose from a 2.00M stock…
A: Given Stock solution molarity ( M1) = 2.00 M Volume of final solution(V2) = 125.00 ml Molarity of…
Q: Explain in detail how you would make the solution from the previous question (50.00 mL a 300 ug/dL…
A: We have to prepare 50.00 mL of a 300 μg/dL solution from a 1500 μg/dL stock solution. So, here the…
Q: Preparation and Standardization of 0.1 N lodine Solution Preparation 1. Dissolve 14 g of iodine in a…
A: As in As2O3 is undergoing oxidation to Na3AsO4. I in I2 is undergoing reduction to form NaI.
Q: Order the following in terms of concentration, with 1 being the least concentrated and 4 being the…
A: Molarity can be defined as the number of moles of solute present per unit of volume.…
Q: Objective: To determine the percent NaCl (w/v) of a water sample 5.00 mL sample diluted to 50 mL…
A: Balanced chemical equation: NaCl (aq) + AgNO3 (aq) ------> NaNO3 (aq) + AgCl (s) From the…
Q: With KD = 3.5, in a solution of 10 grams of aspirin in 80 mL of ether using two separate extractions…
A: A mixture is a type of matter which is composed of two or more substances. These are called…
Q: Give a gne sentence recipe explaining how to prepare 100 ml of 5 pM ag dimethyl fumarate solution by…
A: The mole fraction of a solute is the number of moles of solute per moles of solution. Mole fraction…
Q: Objective: To determine the percent NaCl (w/v) of a water sample 5.00 mL sample diluted to 50 mL…
A: The question is based on the concept of quantitative estimation. we have to calculate percentage…
Q: our team is asked to make up a 200mL agarose gel, 1%, in 1XTAE containing 0.5ug/mL of thidium…
A: Agarose Gel Agarose gel is a three-dimensional matrix made of super coiled bundles of helical…
Q: A solution contains 6 umol Na2SO4 in 284 ml. How many ppm Nat does it contain? A.W Na=23 S=32 0=16…
A: The concentration in ppm is calculated as the ratio of grams of solute and the volume of the…
Q: Which of the following consideration are applied when choosing a suitable recrystallization solvent?…
A: Recrystallization is the technique use to purify solids using specific solvent. Characteristics of a…
Q: 12 g of compound “Y” dissolves in 10 ml of acetone at 25 0C and 0.85 g of the same compound…
A: The necessary condition to recrystallize any impure sample is that the solubility should be…
Q: The distribution coefficient between methylene chloride and water for solute Y is 15. An amount of…
A: Distribution coefficient simply denotes how a compound is distributed in a mixture of two immiscible…
Q: Assume that you are provided with an 18 cm fractionating column having an HETP of 6 cm to distill a…
A: To determine the composition of first drop of distillate
Q: What is the final concentration if 100 mL of a 20% w/v solution is diluted to 250 mL with water?…
A: Correct answer is (A) 8% w/v. Amount of solute remains constant during dilution. So, mass of solute…
Q: Upon adding 400 mL of 1.0 M HI to 400 mL of 1.0 M C5H5N
A: This is the acid base reaction .
Q: Calculate the temporary and permanent hardness of water sample containing Mg(HCO3)2= 7.3mg/L,…
A: Given data: Amount of the hardness causing salts (mg/L) is given: Mg(HCO3)2 =7.3 mg/L Ca(HCO3)2 =…
Q: Calculate the saponification value.
A:
Q: calculate the boiking point of a solution in 0.20 grams of eugenol, dissolved in 12.0 grams of…
A: Given: The mass of eugenol is 0.20 g. The mass of benzene is 12.0 g. The boiling-point elevation…
Q: A student needs 25.0 mL of 7.18x10 4 M FD&C Blue Dye No. 2 solution. If the student has 2.00 mM FD&C…
A: We will solve this question using molarity equation.
Q: You have 100 mL of a solution of benzoic acid in water; the amount of benzoic acid in the solution…
A: Volume of benzoic acid solution = 100 mL Mass of benzoic acid in solution = 0.30 g Distribution…
Q: 750 wwi.f Hy 1o0% collectron efficenly
A: Given: Concentration of methane given = 25 µg/mL.
Q: The solubility of benzoic acid at 95 deg C is 68.0g/L. Calculate the maximum amount of benzoic acid…
A:
Q: GIVEN: - 500mg of ascorbic acid - 2L of distilled water - 7.5% m/v (this is the stock solution)…
A: A question based on concentration terms that is to be accomplished.
Q: 11. What is the concentration of losartan in this suspension, in mg/mL? 12. An infant is prescribed…
A:
Q: How many ml of ethenol should be mixed with 225 ml of water to make 4.5% (v/v) mixture
A: Suppose Volume of ethanol is =y ml Volume percent v/v % = [(volume of solute)/(volume of solution)]…
Q: What is the %v/v of 10mL CH3OH (methanol) dissolved in water to a volume of 40mL
A: Percent by volume = (volume of solute /volume of solution)*100
Q: What volume of a 4.00 × 10-4 M FeSCN2+ solution would be needed to prepare 50.0 mL of a 7.20 × 10-5…
A: Here, we have to find the volume of a 4.00×10-4 M FeSCN2+ solution that would be required to prepare…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- 1. Why should the stopper always be removed from a separatory funnel whenever a liquid is being drained through the stopcock?2. The distribution coefficient, KD (ether/water), between ether and water for aspirin at room temperature is 3.5. What weight of aspirin would be extracted by a single extraction with 60 ml of water from a solution of 10 grams of aspirin in 100 ml of ether? Calculate the weight of aspirin which would be removed by three extractions with 20 ml portions of water.How much 1.0 M sodium bicarbonate solution (in mL) is needed to stoichiometrically react with 25 mL of 4.5% (w/v) acetic acid? Molecular weight of sodium bicarbonate = 84.007 gmol, molecular weight of acetic acid = 60.052 g/mol please provide explanationAn organic compound has a distribution coefficient of 1.5 in methylene chloride and water. If 50 mg of this compound is dissolved in 15 mL of water, in which case will more of the compound be extracted into the methylene chloride: one extraction with 15 mL of methylene chloride or three extractions using 5 mL of methlene chloride each time? Show all work.
- 15 g of unknown organic sample was dissolve in 575 mL of Dicloromethane (DCM). The boiling point of benzene was increased by 3.40oC. Determine the molecular weight of the unknown sample? Kb of DCM = 2.42oC/m Bb of benzene = 39.6 oC density of benzene = 1.33 g/mL at 25 °Cpart 1 What is the % w/v of a 200ml solution containing 50g sucrose? part 2 What is the % w/v of a 2.5L solution containing 26.45g of SDS? part 3 How many grams of CaCl2 are required to produce 300 mls of a 10%w/v of CaCl2 solution.1) How many milliliters of 70% ethanol and 20% ethanol must be mixed to prepare 500ml of 30% ethanol? 2) A gaseous blend of nitric oxide and nitrogen (NOmax) contains 0.09% v/v nitric oxide (NO) and 99.92% v/v nitrogen. Express the strength of nitric oxide in PPM (round to whole number) 3) The ratio strength of a 10mL vial of neostigmine methylsulfate injection USP is 1:1500.Calculate the amount of neostigmine methylsulfate in milligrams contained in the 10mL vial. (Round to the whole number).
- 10g coffee 4g sodium carbonate 40m dichloromethane 160ml distilled water coffee filter What are the principles behind caffeine being extracted from coffee using the above items? 1st sodium carbonate mixed with coffee then brought to a boil for 20min then solution/grinds are filtered. Then solution is added to a separatory funnel with 15ml of dichloromethane and two layers are observed so the bottom layer with DCM contains the caffeine, then this layer is collected in a beaker and dried out using molecular seeds. Then the solvent is brought to a boil with crude caffeine remaining and recystallized using 95% ethanol and brought to a boil again. Then the contents are vaccum filtered and allowed to dry resulting in 30mg caffeine.Which of the following methods of preparing 0.0010M HCl solution from 1.0M stock HCl Solution willyield the smaller overlall uncertainty? a. A one step Dilution that uses 1mL(±0.006) pipet and 1000mL(±0.30) Volumetric Flask, or b. A two-step dilution that uses 20mL (±0.03) pipet and a 1000mL(±0.30) volumetric flask and a25 mL (±0.03) pipet and a 500mL(±0.20) volumetric flask for second dilution.12 g of compound “Y” dissolves in 10 ml of acetone at 25 0C and 0.85 g of the same compound dissolves in 10 ml of boiling acetone. If 5.0 g of compound “Y” were to be recrystallized from 75 ml acetone, what will be the next maximum amount of “Y” that will be recrystallized?
- If 5 teaspoonfuls of a 10% w/v povidone-iodine solution were diluted to 2 quarts with purified water, what would be the ratio strenght of the solution? (** Please show traditional method and not C1V1=C2V2 method**)Compound X has a distribution coefficient (K) of 5 for ether/water. If 5.0 g of compound X were dissolved in 500.0 mL of water, how many grams of compound X would be recovered from the water layer after two consecutive extractions using 50.0 mL portions of fresh ether from each extractionA 18 g of unknown organic sample was dissolve in 756 mL of benzene. The boiling point of benzene was increased by 3.36oC. As the first step of analysis, determine the moecular weight of the unknow sample? Kb of benzene= 2.64oC/m Bb of benzene = 80.09 oC density of benzene = 0.874 g/mL at 25 °C Answer in whole number, no units required.

