2. Provide the equilibrium that defines the pH of the following solutions. For solutions that involve more than one ionization or protonation, provide only the equilibrium that contributes most significantly to the solution pH. a. An aqueous solution prepared that is 0.10 M CH3COOH (Ka = 1.8 x 10-5) and 0.10 M CH3COONa (K₁ = 5.6 × 10-1⁰). b. An aqueous solution prepared that is 0.25 M in sodium citrate (citrate ion pictured below), Na3 C6H507 (Kb1 = 5.6 × 10-8; Kb2 5.9 × 10-10; Kb3 = 1.4 × 10-¹1) eve OH
2. Provide the equilibrium that defines the pH of the following solutions. For solutions that involve more than one ionization or protonation, provide only the equilibrium that contributes most significantly to the solution pH. a. An aqueous solution prepared that is 0.10 M CH3COOH (Ka = 1.8 x 10-5) and 0.10 M CH3COONa (K₁ = 5.6 × 10-1⁰). b. An aqueous solution prepared that is 0.25 M in sodium citrate (citrate ion pictured below), Na3 C6H507 (Kb1 = 5.6 × 10-8; Kb2 5.9 × 10-10; Kb3 = 1.4 × 10-¹1) eve OH
Chapter15: Acid-base Equilibria
Section: Chapter Questions
Problem 8ALQ: You have a solution of the weak acid HA and add some of the salt NaA to it. What are the major...
Related questions
Question
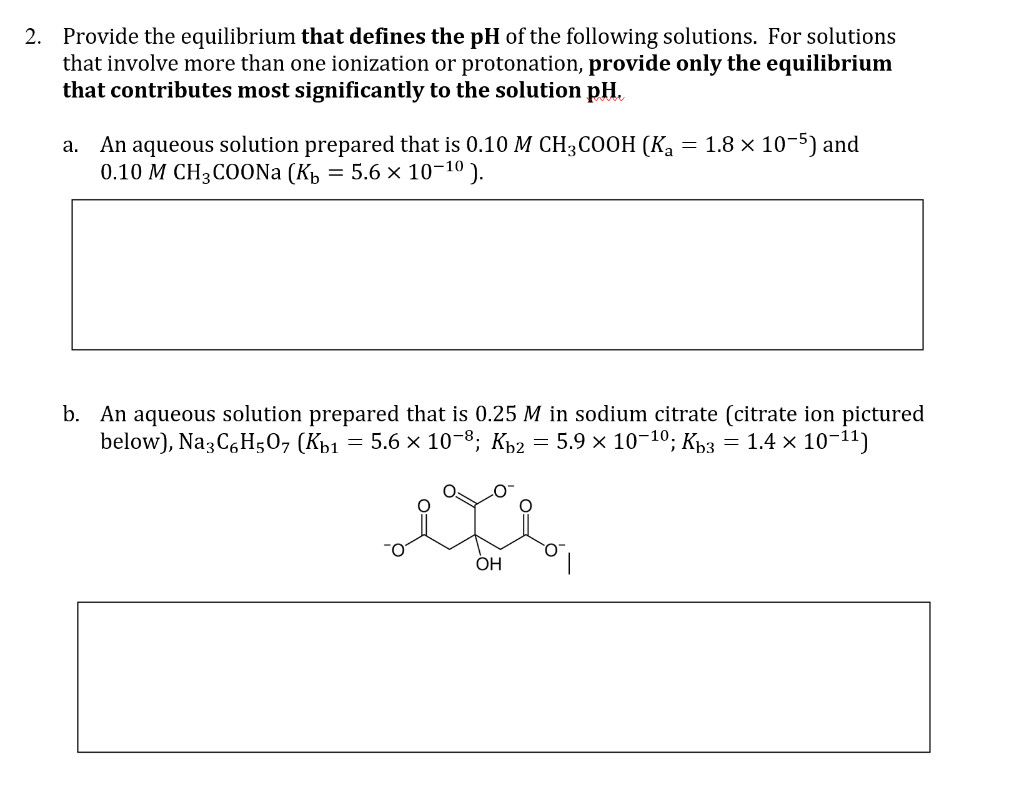
Transcribed Image Text:2. Provide the equilibrium that defines the pH of the following solutions. For solutions
that involve more than one ionization or protonation, provide only the equilibrium
that contributes most significantly to the solution pH.
a. An aqueous solution prepared that is 0.10 M CH3COOH (Ka = 1.8 x 10-5) and
0.10 M CH3COONa (K₁ = 5.6 × 10-10).
b. An aqueous solution prepared that is 0.25 M in sodium citrate (citrate ion pictured
below), Na3C6H50, (Kb1 = 5.6 × 10-8; Kb2 = 5.9 × 10-10; Kb3 = 1.4 × 10-11)
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
