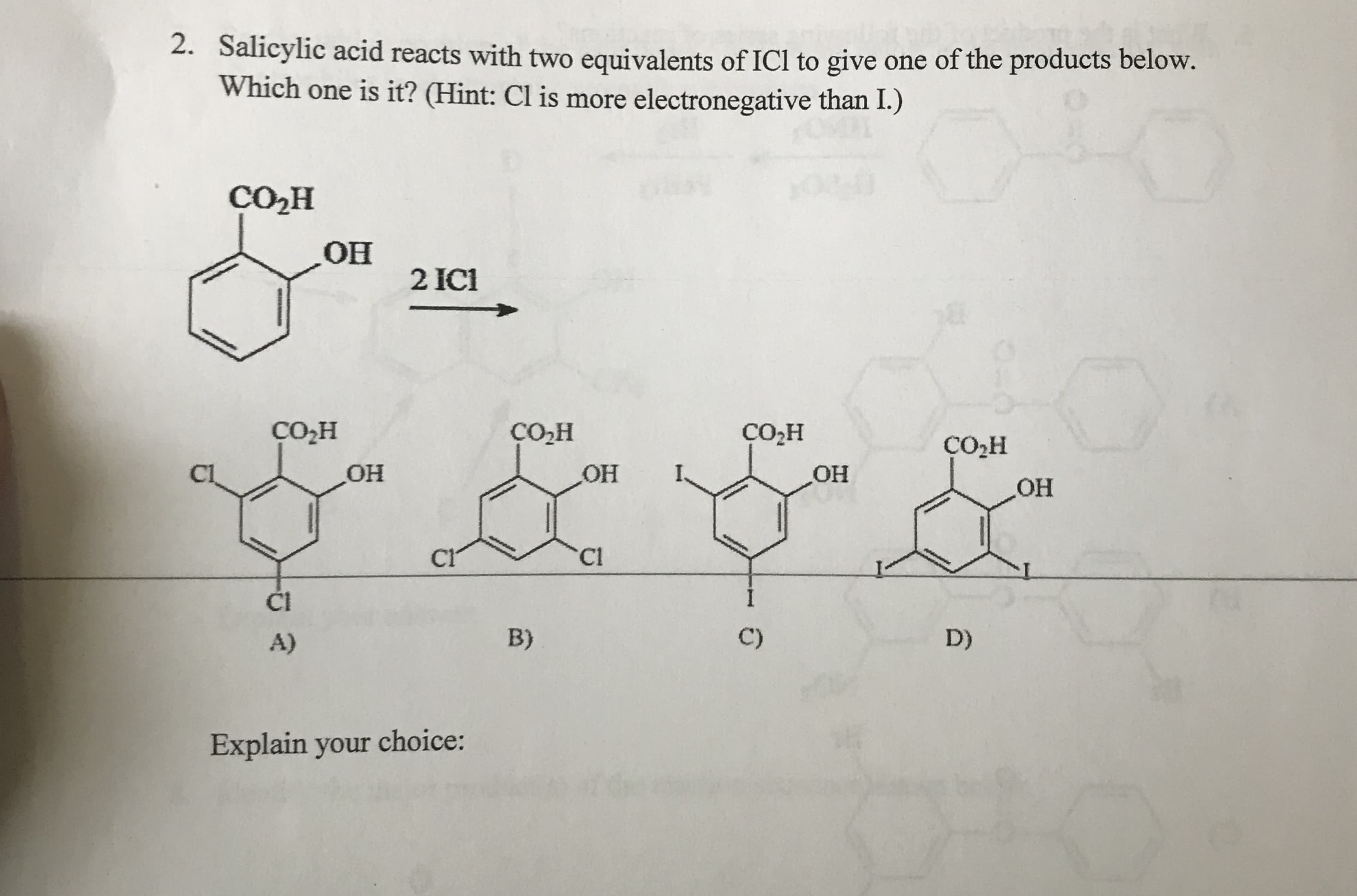
2. Salicylic acid reacts with two equivalents of ICI to give one of the products below. Which one is it? (Hint: Cl is more electronegative than I.) CO2H Но 2IC CO2H Со-н он СО-н C2H C1 OH HO Но Cl Cl Cl C) B) D) A) Explain your choice:
Q: a. Here are two different carbonyl bearing molecules. With the knowledge you learned so far explain…
A: Oxygen is more electronegative, therefore, it gains partial negative charge and carbonyl carbon…
Q: HCN KCN
A:
Q: 73. C✓ 73. Provide the final product of the following reaction. OMe heat + Me₂NOMe ? OH Me OMe NMe…
A: Eschenamoser-Claisen Rearrangement: Claisen rearrangement reaction employing allylic alcohols and…
Q: Arrange the compounds below in order of increasing reactivity (least reactive = 1; most reactive =…
A: 1.B 2. A 3. C 4. D
Q: Dtermine the Product from the rxn 1. ΗΝΟ,/Η,SO4 2 Br2/FeBr3 Product 3. H2/Pd/C 4. Cl2/FeBr3 5.…
A: Here we have to choose correct option about product of above reaction-
Q: Li / liq. NH3 G then NH4C1
A:
Q: Q6-) Complete the following Grignard reaction. Mg H,C-Br CH3 THE
A:
Q: Predict the product of the following reaction. 1. CH3I(x's)/Ag20,heat 2.KMN04, cold, neutral NH2 HO…
A:
Q: f. + CÓ + 1H2
A: Given
Q: 1. compare sn1 reaction on these compound 2 what compound is X CN OMe Br Br NMe B Ma. Me Me Me Me Me…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which hydrogen atom is the most likely to be abstracted when this molecule is reacted with NANH,? -…
A: Sodium amide (NaNH2 ) is a strong base and a strong nucleophile. It deprotonates alcohol because…
Q: Predict the following products: 1. BH/THF 2. HO", H,O2, H20 1. О, -78C 2. Zn, CH;CO2H m-CРBА NO2…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Give the major product for the reaction below. A HBr H2 F Ni KMNO4, OH, heat > B H* KMNO4 Br2 cold,…
A:
Q: 23. Congo Red is a synthetic dye whose use was later abandoned because of its carcinogenic…
A: (23) answer: Correct option (b) From Retrosynthesis analysis we can say, Nelson can synthesize…
Q: Determine the missing product. а. НСI + H 0 > Нзо+ + b. F- + H2О > HF + с. НSO4 + Н-О — HSO4 + d.…
A: In each of the given reactions, hydrogen ions (H+) are substituted from one species to another.…
Q: (d) 0.14 M (NH4)2SO4 lons: SO3 NH3* O So,2- 0,2- NH4* masscation i massanion II
A: The electrolytes which are completely ionizable in water are known as strong electrolytes.
Q: Consider the following reactions below. ... Mg 1. ether 2. H3O* Br ether HyC *CH3 IV Compound IV…
A: The correct option is option A.
Q: 1.Which of the following is a possible product for the unbalanced chemical reaction: NaCl +…
A: Balanced reaction means number of atom in reactant side is equal to number of atom in product side.
Q: ai. What is the normal/ primary isomer of C4H10O ? ii. Treat the primary isomer with conc.H2SO4…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subpartsfor…
Q: 409). (D149) OH H. OH coo -N R374 но. OR 1st Step (E117) ROH (Y409). (D149) OH OH R' -N R374 но-…
A: Acids: The organic compounds which have the tendency to donate H+ ions(protons) For example HCI…
Q: Pls help ASAP.
A: Answer of the question given below,
Q: Fill in the product that goes in the yellow box 1452 End 63% yed + MezNOH psiMes Bno osSiMes Brom…
A:
Q: 3. Predict the products. Он sOCIl, оН РBrg Он NaOH TSCI py SN2
A: The conversion of one organic compound into others is done by different types of reaction, all these…
Q: Problem Set #5 III) Acid/Base Reaction - Answer the next few questions about Molecule B, shown…
A: The solution is as follows:
Q: An organic chemist reacts (S)-2-butanol with PBr3 in pyridine, removes the major organic product,…
A: The above reaction is proceed by SN2 mechanism in which attack of -OCH3 is done in back side of OH.…
Q: 4. Briefly explain why the reaction below would be unsuccessful. NH2 NH2 AICI,
A: A question about Friedel-craft acylation based electrophilic substitution reaction of aniline.
Q: What is the structure of compound C in the following chemical transformation? a) Br 50 O Compound A…
A: Given: Reactant is benzene. The reaction is a multistep reaction.
Q: Why is it necessary to incorporate the benzyl group on the chloroamine used to convert B to C?
A:
Q: NaN, compound a compound b + compound c Reagents m. Na / NH, n. H,SO, H9SO. o. (sia),BH then H,O,…
A: In the given reaction the alcohol is converted into alcohol and the alcohol is converted into alkyl…
Q: Nicotinic acid, more commonly named niacin, is one of the B vitamins. Show how nico- tinic acid can…
A: The organic reactions help in the conversion of one compound into many other compounds with the help…
Q: Ethane HCI Br2 Uv H H2O Sulfuric OCH3 B 1 mol h2/ lindlars F Br2 D E 2NaNH2 NaNH2
A:
Q: Suppose you have compounds A–D at your disposal. Using these compounds, devise two different ways to…
A: Reactions involving organic compounds are called organic reactions. Methyl iodide and sodium…
Q: Match the reactions with their products below. Кон (аq) 1. a.) OCH3 b.) Ph3P: MnO2 c.) OH OCH3 3. 2.
A:
Q: 9) Predict the reactants for the ff products: SH (a) CH3OCH2CH2CH3 (b) „OCH3 (c) (d) CH3…
A: the reactants are as follows:
Q: Q. The final product of this synthesis is a non-steroidal anti-inflammatory drug (NSAID). It has…
A: At first chlorination occurs to give chlorobenzene. Then nitration gives ortho nitro chlorobenzene…
Q: Label the most acidic hydrogen in each molecule and justify your choice by using appropriate pKa…
A: The strength of an acid can be determined by pKa values. pKa is defined as the negative log of acid…
Q: a) Br KOH, etanol CH,l, ZnCu Br b) Bry, H,0 B
A: The answer along with explanation is provided in picture below
Q: Complete the reaction map below by matching the answers for A-E. Cl2 PONH D- Fecls CH3CI B KM,O4 C E…
A:
Q: NH2 IUPAC mame @ and 6) (a) (CEHS), NH
A: The IUPAC name of the compound can be written on the basis of the main carbon chain, functional…
Q: Give the structure of compound A, the major product when the reaction is conducted at low…
A:
Q: An organic chemist reacts (R)-2-butanol with PBr3 in pyridine, removes the major organic product,…
A:
Q: 3) Consider the synthesis below. Identify P1 and deduce What are the reagents/reactants A and B A…
A: Given:
Q: MeO2C Li -N(allyl)2 MeO NH3 - ELOH Br Pd(PPH3)4, Cul
A: 1st One is birch reduction. -OMe is a electron donating group. Benzene ring reduced with 2 double…
Q: Suppose you have compounds A-D at your disposal. Using these compounds, devise two different ways to…
A: In Williamson ether synthesis we always prefers that the leaving group must located in 1° alkane…
Q: (i) increasing solubility in water. (CH3),CHCH,OH CH;CH,CH,CH,OH CH;CH,CH,Br CH;CH,OCH3 (a) (b) (d)…
A: The sequences are as follows:
Q: Predict two likely products of the following reaction. CH3 Br NaOC(CH3)3
A: In this question we have to tell the product of the reaction.
Q: The reaction below is likely a: CH;CH,OH Cl OCH,CH3 Syl El E2 O. SN2
A:
Q: Arrange from Highest to Lowest Priority *(CO means C=O “C double bond O") 1. R-COCH3 , R-COOH,…
A: This is the concept of Organic and inorganic chemistry
Q: NMez OH (a) H,PO4. (b) (a) HNO, H,SO, (c) (d) NAOH, (d) (1) (e) SOC, HNME2 HO H,O, heat () H2. Pd/C…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Illustrated below is the hydrolysis of a C-C bond (in the first molecule) hydrolyzed by water (2nd molecule). Which pair of open boxes best identifies the location of the (-H) and (-OH) components of water on the products. Choose one from the following: (a) A (b) B (c) C (d) DLooking for compound B and E . Thank YouCompound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yne
- Compound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?Show how HC≡CH, CH3CH2Br, and (CH3)2CHCH2CH2Br can be used to prepare CH3CH2C≡CCH2CH2CH(CH3)2. Show all reagents, and use curved arrows to show movement of electron pairs.Rank A, B, and C in order of increasing SN1 reactivity.
- Will the bonding of the Zn2+ ion to the ketone oxygen lone pair make theketone C=O easier or more difficult to reduce? Why?The compound whose structure is shown here is acetyl acetone. It exists in two forms:the enol form and the keto form The molecule reacts with OH–to form an anion, [CH3COCHCOCH3] (often abbreviatedacac–for acetylacetonate ion). One or the most interesting aspects of this anion is thatone or more of them can react with transition metal cations to give stable, highlycolored compounds (a) Are the keto and enol forms of acetylacetone resonance forms? Explain youranswer.(b) What is the hybridization or each atom (except H) in the enol form? What changesin hybridization occur when it is transformed into the keto form?(c) What are the electron-pair geometry and molecular geometry around each C atomin the keto and enol forms? What changes in geometry occur when the keto formchanges to the enol form?(d) Draw three possible resonance structures for the acac–ion.(e) Is cis-trans isomerism possible in either the enol or the keto form of acetylacetone?(f) Is the enol form of acetylacetone polar?…If a small amount of aqueous Br2 is added to A, the reddish color of Br2 persists, but the color disappears when Br2 is added to B. When the aziridiniumion reacts with methanol, only A is formed. Identify A and B.

