2.) Sucrose + Copper (II) Acetate/Acetic Acid
Q: -Which of the following is/are ketone bodies: a. B-hydroxybutyrate b. Acetone C. Acetoacetate d.…
A: Ketone bodies are an alternative fuel for the body when there is shortage of glucose. They are made…
Q: 1. Select the odd one out. xylulose b. dihydroxyacetone c. glyceraldehyde d. ribulose С.
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 8) Define the following terms: a. Hydrophobic b. Hydrophillic
A: Introduction The cell membrane is a thin semi-permeable membrane that surrounds the cytoplasm of a…
Q: 7. Which of the following statement is true? A. oxidative rancidity is observed more frequently in…
A: Introduction: Rancidity is the development of unpleasant smells in fats and oils that is often…
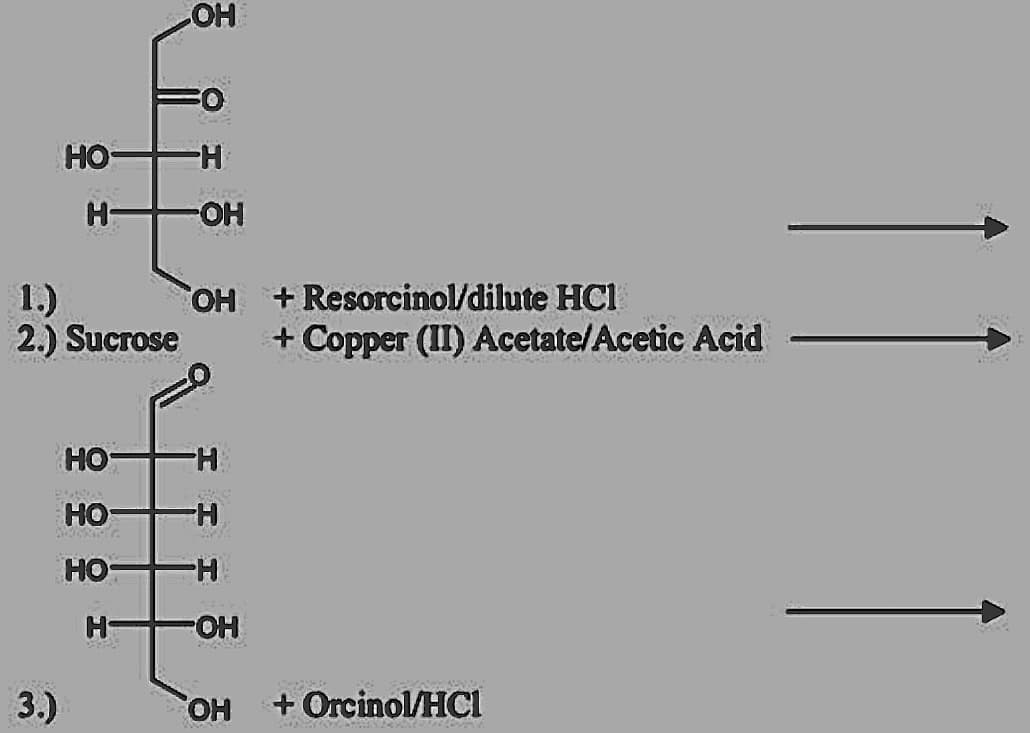
Q: | Conc. H,SO, a-naphthol |(*) Conc. H,S0, acetic anhydride Orcinol, FeCl, HCI A () c (*) (-)…
A: This experiment shows testing of a mixture of carbohydrate/sugar In Step 1: Reagent used in Conc…
Q: 1. What is the indicator reagent for each of the following organic compounds and what color change…
A: Hi! Thank you for the questions. As you have posted multiple questions, I will be answering the…
Q: 1. The general chemical formula of carbohydrate is: (in 1 to 3 sentences, explain why this is your…
A: Carbohydrates are diverse in function and in their structures. They perform vivid functions and…
Q: 7. What type of reaction forms a disaccharide from monosaccharides? acid-base B. condensation C.…
A: Carbohydrates are the hydrates of carbon that contain carbon, hydrogen, and oxygen in the ratio of…
Q: 9.The disaccharide given is CH;OH O OH CH2OH OH OH Он ÓH A. Sucrose B. Lactose C. Maltose D.…
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which…
Q: 34. Which fatty acidis most likely to bea solid at room temperature? a. CH он b. CH, CH OH CH,-C-OH…
A: Fat is an important energy source for the body. Fat is made of fatty acids and glycerol. Fatty acids…
Q: 1. What is the indicator reagent for each of the following organic compounds and what color change…
A: The indicator is the type of chemical reagent, which is used in the chemical test for the detection…
Q: 3. Identify the amino acids contained in each of the following tripeptides. он он он || |…
A: Amino acids are the monomers of polypeptides that are linked by peptide bonds. A peptide bond is…
Q: 20. The process below is otherwise known as HC OC-(CH) CH, Fat +S H,C(CH)e- Na HC-O-H a. alkali…
A: Soaps are the sodium and potassium salts of the long-chain carboxylic acids. Some of the examples…
Q: Which of the following contains the most lipids? A. Banana B. Champorado C. Olive oil D. Cheese
A: Different food items consist of different micro and macronutrients. Few of the macronutrients are…
Q: Sucrose carbohydrate is categorized as a heteropolysaccharide a heterodisaccharide a monosaccharide…
A: Oligosaccharides are formed by the condensation of 2-9 monosacharide units. On the basis of number…
Q: 5. What is the classification of the mixed triglyceride below? H,C-o HC 12 15 H,C-o Simple Lipids…
A: Lipids are the macro molecules made up glycerol backbone, fatty acids attached to it. They are also…
Q: e. Intermediate Example(s) of Polyunsaturated fatty acid (PUFA): a. Folic acid b. Linoleic acid C.…
A: Unsaturated fatty acids are those fatty acids that contain one or more double bonds. Unsaturated…
Q: In lactose monosaccharides are linked by Select one: a. 1-4 glycosidic bond b. 1-2 glycosidic bond…
A: A glycosidic bond is usually found in polymers such as sucrose, lactose and starch, and functions to…
Q: From what monosaccharides or derivatives of monosaccharides is keratin sulfate made?
A: Bio molecules also known as biological molecules. These are the molecules which are produced by…
Q: 3. Explain why B-keto acids are more readily decarboxylized than a-keto acids. R a-keto acid B-keto…
A: Decarboxylation is the reaction when carbon dioxide is released in a reaction leading to reduction…
Q: 3. Disaccharides a. Draw a lactose molecule b. Label the individual monosaccharide components c.…
A: The disaccharides are made up of the joining two monosaccharides. The disaccharides can be homodimer…
Q: What is the molecule below? A. Tetrahydrofolate B. 5-adenosyl methionine C. Pyridoxal…
A: Coenzymes are substances that enhance enzymatic activity by binding to the enzyme. coenzymes are the…
Q: 29-All of the following are examples of omega-3 fatty acids except: a. linoleic acid. b. linolenic…
A: Omega-3-fatty acids are a heterogeneous group of fatty acids that contain a double bond between the…
Q: The Disaccharide Maltose is formed by a (an): O a. a(1- 2) O-glycosidic bond between 2 glucose…
A: Maltose is a disaccharide composed of two monosaccharides attached together via a glycosidic bond.
Q: 2. What is the general name for a monosaccharides which has A. 6 carbons B. 5 carbons C. 3 carbons
A: Monosaccharide- Simplest carbohydrates, sugar and have only 1 sugar molecule.
Q: Binding additives are polysaccharides that have functions of: A. Encapsulating and aerating B.…
A: Food is made up of carbohydrates, fats, proteins, vitamins, minerals, water, and a range of other…
Q: Starch undergoes decomposition under high concentration of Select one: a. base b. acid c. iodine…
A: Starch is a form of complex carbohydrate made up of two polymers, namely amylose (linear) and…
Q: Acid hydrolysis of soap yields _________ a. fatty acids b. glycerol c. salts of fatty acids d.…
A: Soap is produced by the saponification (hydrolysis) of a triglyceride (fat or oil). In this process…
Q: . Glycosidic bond between galactose and glucose in lactose is ______. In 2-3 sentences, explain why…
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the…
Q: What is the glycosidic linkage between monosaccharide I and II? α-(1→2) β-(1→2)…
A: Glycosidic linkage: Glycosidic bonds formed between monosaccharide units are the basis for the…
Q: Detection of starch in food requires the addition of lodine .a O Sudan Red .b O Sodium .c O Gelatin…
A: Starch is the stored form of glucose in plants ,which means that plants reserve the glucose in the…
Q: Which classification(s) is(are) applicable to maltose? CH,OH CH,OH H H H. OH OH H OH H H HO H…
A: Disaccharides are composed of two monosaccharides attached together via glycosidic linkages. The…
Q: A bottle of canola oil can be stored indefinitely at room temperature because: a. it has 0…
A: Canola oil is defined as the edible vegetable oil, which is obtained from the canola seeds that…
Q: 2. Which of the following is an aldotriose? (in 1 to 3 sentences, explain why this is your answer).…
A: Sugars are referred to as saccharides and are the generic term that refer to carbohydrates.…
Q: 1. (a) Arrange the following lipid molecules in the order of their relative melting points. Hc -o-…
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which…
Q: What process is used to put fats together? Group of answer choices A. anhydric fusion B. chemical…
A: Biomolecules are the chemicals present in cells and their organelles.
Q: Starch undergoes decomposition under high concentration of Select one: a. base b. acid c. iodine d.…
A: Starch consists of amylose (linear) and amylopectin polymers (branched). It includes between 10% and…
Q: be de as alani) CH,OH он но ÓH NH-C-CH, Select one: O A. a sugar phosphate. O B. a disaccharide. O…
A: Carbohydrates are macromolecule group that provides cells with a vital source of energies. They also…
Q: Conc. H,SO, a-naphthol (*) Conc. HSO, acetic anhydrde Orcinol, FeCi, HCI A () Cu(CH,COO), acetic…
A: The schematic diagram that is represented is showing the results of different biochemical tests for…
Q: The correct systematic name for the fatty acid below is: a) 3 octadecatrienoic acid b) 9…
A: Ans - Carboxyl carbon (- COOH) would be given first number in order to start numbering and naming of…
Q: (i) Write the name of two monosaccharides obtained on hydrolysis of lactose sugar.(ii) Why Vitamin C…
A: Biomolecules or macromolecules are polymers made of small units called monomers. Living organisms…
Q: 1.cholesterol is a. plastic in the blood b. a waxy, fatty substances in the blood C. a type of blood…
A: cholesterol is a sterol or amphiphilic lipid with a short hydrophobic head and long hydrophilic…
Q: Which of the circled monosaccharides is an unusual deoxy sugar called L-fucose? I II…
A: Fucose is an unusual monosaccharide with six carbons. And fucose is a deoxy sugar, which lacks…
Q: Fructose is classified as a(n) a. aldotriose b. aldohexose c. ketopentose d. ketohexose
A: Fructose is sugar which is commonly found in fruits. Fructose is also the part of sucrose where it…
Q: A physician ordered 250 mg capsules of tetracycline to be taken four times per day for 10 days. How…
A: Dosage calculations: a. Dose is the amount of drug that should be taken a single time. b. The…
Q: A disaccharide is composed of two monosaccharides connected by a glycosidic bond. Select one: a.…
A: Carbohydrates are the polyhydroxy aldehydes or ketones which are associated with reducing property.…
Q: 6. Glycosylation reaction 7. Lipid synthesis 8. Degradation reactions 9 Catalase activity
A: Multiple subparts asked. I can answer initial 3 subparts , as allowed by guidelines.
Q: Classify the following as polysaccharide, oligosaccharide, disaccharide, monosaccharide. a.…
A: Monosaccharides are the simplest carbohydrate units. Disaccharides are formed by joining two…
Q: Catalase, sucrase, lactase are examples of... A. simple sugars B. nitrogen bases C. enzymes D.…
A: Enzymes are proteins that act as biological catalysts (biocatalysts). Catalysts accelerate chemical…
Step by step
Solved in 2 steps

- Indicate whether the following pairs of monosaccharides are aldoses or ketoses. a. D-altrose & D-xylose b. D-arabinose & D-glyceraldehyde c. D-erythrulose & D-psicose d. D-talose & D-gulose e. dihydroxyacetone & D-tagatose f. D-idose & D-threoseWhat is Artemisinin ?What are the Identification Tests for Carbohydrates? Why do we need to study them? What are the importance?
- In solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?All of the following are repeating units of glycosaminoglycans EXCEPT: A. hyaluronate B. heparin C. hemoglobin D. keratin sulfate E. chondroitin 6-sulfateIs D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Explain.
- Which of the following is a property of both D-altrose and D-talose?Construct the following lipids: a. Alpha-arachido-beta-clupadonolignocerateb. Alpha-stearidono-beta-gadoleonevonatec. Alpha,beta-dimyristocapratewhat are the different types of O-glycosidic linakge existing between the monomeric units of the di-, oligo-, and polysaccharides?

