2. The following figures are examples of two DNA bases. DNA has a double-stranded structure that requires enzymes or high temperatures to separate the two strands because hydrogen bonds keep the strands together. NH₂ & 4 adenine (A) thymine (T) -CH₂ ply collisions that can contribute to the
Q: Determine the enthalpy of vaporization, in kJ/g, of C₆H₁₄ if 14.5 kJ of heat is needed to vaporize…
A: Given: Enthalpy change when 38.05 g C₆H₁₄ vaporizes is 14.5 KJ Required: Enthalpy change when 1 g…
Q: In the laboratory, a general chemistry student measured the pH of a 0.392 M aqueous solution of…
A:
Q: Draw the: Zwitterion 0.1 M HCl 0.1 M NaOH
A: The neutral form of amino acid is the Zwitterion in which the carboxylic acid group is deprotonated…
Q: Activity 2.2. Using noble gas cores, write the shorthand electron configuration of the following: a.…
A: Using noble gas cores, write the shorthand electron configuration of the following: a. Zn b. Ba c.…
Q: Draw the structure of each molecule. (a) (E)-4-carbamoylbut-3-enoic acid; (b)…
A:
Q: Identify the following elements with their corresponding mass number. Use this format for your…
A: We have find out the solution.
Q: Solve the equation for m. 2m² - 3m 4 = 0
A:
Q: If you mix 1.15 g Na₂C₂O (FM 134.00) with 0.60 g KMnO4 (FM 158.03) and excess aqueous acid, which…
A: The limiting reactant is the one that is completely consumed before the other reactant is consumed.…
Q: 3. For the following reactions, draw all possible products. Then indicate which product would be the…
A: "Since you have asked multiple-subparts questions, we will solve the first-three subpart questions…
Q: Write dot-and-cross diagrams for the following compounds NaBr (sodium bromide) CO₂ (carbon dioxide)
A: According to the answering guidelines, I'm posting the solution for the first question. Kindly post…
Q: 1. Rank the indicated hydrogen atoms in order of increasing ease of abstraction in a radical…
A: Radical Halogenation Reaction Radical halogenation is a type of organic reaction in which a halogen…
Q: Draw and name the thee steps of the mechanism of the bromination reaction of 1,2-diphenylethane.
A: The addition of bromine to an alkene in a nonpolar solvent produces vicinal dibromo alkane.
Q: The potassium ion in 250 ml of a water sample was first precipitated as potassium tetraphenylborate:…
A: The potassium ion in 250 ml of a water sample was first precipitated as potassium tetraphenylborate:…
Q: 15.59 Two methods convert an alkyl halide to a carboxylic acid having one more carbon atom. b. C. HO…
A: The first reaction proceeds via SN2 reaction mechanism where the CN- nucleophile attacks from…
Q: 1. Petroleum is a mixture of many different hydrocarbons a) Which two of the structures A, B, C, and…
A: We have to identify the structures from A, B, C and D that are hydrocarbons
Q: Complete the following statements. Word bank: a reaction coordinate lowest slower highest a lower…
A: Answer: All the terms mentioned in the above problem are used to characterize and study the reaction…
Q: A particular frost-free refrigerator uses about 785 kWh of electrical energy per year. Express this…
A: Given --> 785 kWh Conversion --> (a) Energy in J 1 kWh = 3.6 x 106 J 785 kWh = 3.6 x 785 x…
Q: Consider the following elementary steps that make up the mechanism of a certain reaction: 3A ⟶ B +…
A: The overall reaction is obtained by the addition of the elementary steps and eliminating the…
Q: What is the final volume in millilitres when 0.575 L of a 38.5 % (m/v) solution is diluted to 21.7%…
A: -> From dilution moles does not change . Initial volume (V1) = 0.575 L = 575 ml (1L=1000ml)…
Q: 4. The heats of vaporization for water and carbon disulfide are 40.7 kJ/mol and 26.8 kJ/mol,…
A: Since liquid molecules are constantly moving and have a wide range of kinetic energies, some of them…
Q: The great French chemist Antoine Lavoisier discovered the Law of Conservation of Mass in part by…
A: To determine : 1) Balanced chemical equation for the decomposition of mercury (II) oxide (HgO) into…
Q: For the reaction 4HCI(g) + O₂(g)—2H₂O(g) + 2Cl₂(g) AH° = -114.4 kJ and AS° = -128.9 J/K The maximum…
A: We know , Maximum Work done = ∆G0 = ∆H0 - T∆S0
Q: Consider the reaction H₂CO(g) + O₂(g)—CO₂(g) + H₂O(l) for which AH° = -563.3 kJ and AS° = -140.5 J/K…
A:
Q: Please find the temp at which the reaction becomes nonspontaneous!! The following reaction has these…
A: Given ΔHrxn = -30.1 kJ/mol ΔSrxn = -58.3 J.mol.K
Q: Given: molality = 0.54, pH = 2.43, Using the given equation, estimate the mean activity coefficient…
A:
Q: 14. Bromine is often used as a laboratory spot test for unsaturation in an aliphatic hydrocarbon.…
A: The molecular formula of the given compound is C6H6 Vial A decolorize Bromine and Vial B did not…
Q: The bond angle of H-O-C₁ is The bond angle of O-C₁-N is [Select] [ Select] The bond angle of C₁-N-C₂…
A: First let us consider the hybridisation of different atoms in isocyanomethanol. O atom= sp3…
Q: i) You did the following reaction, worked up in Ethyl Acetate and Water. ၀ Did your reaction work?…
A: Aldehydes and ketones can be reduced to the corresponding alcohols using reducing agents like sodium…
Q: A solution is made using 16.3 percent by mass CH₂Cl2 in CHCI3. At 30 °C, the vapor pressure of pure…
A:
Q: Thank you sooo much! I just have one more thing, how can I convert 15.8 atm to Joules
A: The conversion of the atm L to Joules is such that --> 1 atm L = 101.325 J
Q: Practice: Indicate the direction of the dipole for the following bonds using the symbol, + and &…
A:
Q: A pure solid sample of Substance X is put into an evacuated flask. The flask is heated at a steady…
A: In a heating curve, the first plateau ( horozontal line ) corresponds to the melting point. At this…
Q: Given stock solutions of glucose (1M), aparagin (100mM) and NaH2PO4 (50mM), how much of each…
A: We have to prepare a 500 ml solution which contains 0.05 M glucose, 10mM asparagine and 2mM NaH2PO4.…
Q: . Using the average atomic mass of carbon, calculate the mass (in amu) of 227 carbon atoms. amu .…
A:
Q: Which of these compounds is the weakest base? -OH -OMe CH3CO2- CF3CO2-
A: Conjugate base of strongest acid is the weakest base. ROH ----------> RO- + H+ ROH is acid and…
Q: Write the two half-reactions of the following redox reaction. Calculates the difference between the…
A:
Q: An adult eats food whose nutritional energy totals approximately 2100 CalCal per day. The adult…
A: Given nutritional energy = 2100 cal Calories burnt = 1800 cal
Q: Write a balanced equation for each of the following reactions: a. Hydration of 1-butyne b. Hydration…
A: When butyne is reacted with water as we need to carry out the hydration reaction in the presence of…
Q: How much work is done when 12 moles of CO₂ are produced in a combustion reaction of CeHe at 298 K?…
A: Answer: In the given chemical reaction combustion of 1 mole benzene is taking place in presence of…
Q: You wish to prepare 100.0 mL of a 750.0 ppm w/v sodium (MW = 22.9897 g/mol) solution. How many grams…
A: Given: Volume of solution = 100 mL w/v concentration of Na = 750.0 ppm Molar mass of Na = 22.9897…
Q: 3) Consider an arbitrary operator A. A operates on any function to its right. Here are the results…
A: Let an equation is, Âx = ax Where the operator(Â), operating on a function(x), produces a…
Q: Calculate the percent ionization of a 0.441 M solution of hydrocyanic acid. (Assume that K₁ (HCN) =…
A:
Q: nutrition table lists apples with a potassium content of 4600 mg/pound. Assume an average apple is…
A: potassium content in apple = 4600 mg/pound mass of apple = 86.0 g potassium requirement = 3500 mg…
Q: The current US debt as of 1/10/23 was approximately 31.5 trillion (31,500,000,000). Calculate how…
A:
Q: Which one of the following compounds has highest boiling point? HO (C) (2) ( | || ||| D IV E V " HO…
A: We have to select the compound with the highest boiling point
Q: 4. Sample A contains 100 mL of methanol (CH3OH) and Sample B contains 100 mL of benzene (C6H6). Both…
A: The Antoine equation is a thermodynamic model that can be used to calculate the vapor pressure of a…
Q: How many unpaired electrons are present in a ground state Si atom.
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the…
Q: a d. Ch₂ FCh HNOS H₂SO Br FeBry Br
A:
Q: When a-tocopherol reacts with radicals (such as peroxyl radicals ROO, oxygen radicals, HO), it forms…
A: The resonance structure of given compound can be explained as:
Q: Practice: Circle and name the functional groups in the following molecules. OH lor br HN. OH b) НО…
A: Functional group is a part of molecule which determines its reactivity. The functional groups…
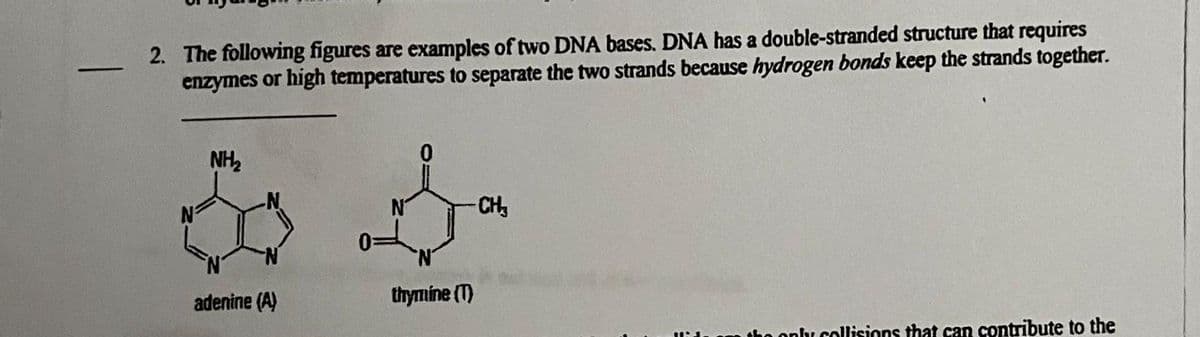
Step by step
Solved in 2 steps with 2 images

- Experimental observations show that thiocyanate (SCN- ), the conjugate base of thiocyanic acid (pKa = 1.1 at 25 °C), is quite soluble in neutral water at pH > 2. In the space provided below, explain this experimental observation. That is, why does SCN– readily dissolve in water when the pH > 2?In the structures of T—A and C—G base pairs, there are three amino groups specifically labeled as “sp2 hybridized and planar”. What is the primary difference between these structures and that of aniline that lead to their planarity? 1. In contrast to aniline, the amino groups on the DNA bases are necessary to make the heterocyclic rings aromatic. 2. In contrast to aniline, the contributing structures that delocalize the nitrogen lone pairs onto the rings creates partial negative charges on electronegative atoms. 3. In contrast to aniline, the hydrogen bond accepting ability of the lone pairs on the -NH2 groups of the DNA bases is better when these amino groups are sp2 hybridized. 4. Both 2 and 3.Thymine is one of the heterocyclic bases found in DNA. Do you expect thymine to bearomatic? Explain
- The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown below. A.. for this reaction, which form of lysine represents A- and which is HA?How much 0.5 M KOH must be added to 200 mL of 50 mM glycine at ph 8.6 to bring its ph to exactly 10.6? pka of glycine= 9.6 please explainAccording to the paper, F486 is a phenylalanine residue (i.e., an amino acid that is part of a chain that forms a protein) on SARS-CoV-2. It has an important interaction with ACE2 (best seen in Fig. 1(C) and Fig. 2(A)). Looking at this phenylalanine and the green residues surrounding it, which statement best describes the interaction that could be occurring? a) It forms a hydrogen bond with Y83. b) It forms a dipole-dipole interaction with M82. c) It forms London dispersion forces with surrounding residues. d) It encounters repulsions from surrounding residues.
- Please consider the structural formula of the amino acid shown below when answering the following questions. Assume that the pKa values are as shown below. (A) draw the form of this amino acid that predominates at pH 8 (B) classify the amino acid as hydrophilic or hydrophobic (C) what is the net charge of this amino acid at pH 1?Is TMA a volatile compound or a biogenic amine? (explain further) Is the measurement of TMA a reliable index of spoilage? Why and why not? (explain further)Which separation technique is the most specific and offers the highest protein purification possible?
- In 0.1M solution glycine (pKa 9.6) at pH of 9.0, what fraction of glycine has its amino group in the -NH3+ form?Glutamic acid is a naturally occurring α-amino acid that contains acarboxy group in its R group side chain. (Glutamic acid isdrawn in its neutral form with no charged atoms, a form that does not actually exist at any pH.) a.) What form of glutamic acid exists at pH = 1?b.) If the pH is gradually increased, what form of glutamic acid exists afterone equivalent of base is added? After two equivalents? After threeequivalents?c.) Propose a structure of monosodium glutamate, the common flavorenhancer known as MSG.The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown below. B. At what pH does [A-]=[HA]?

