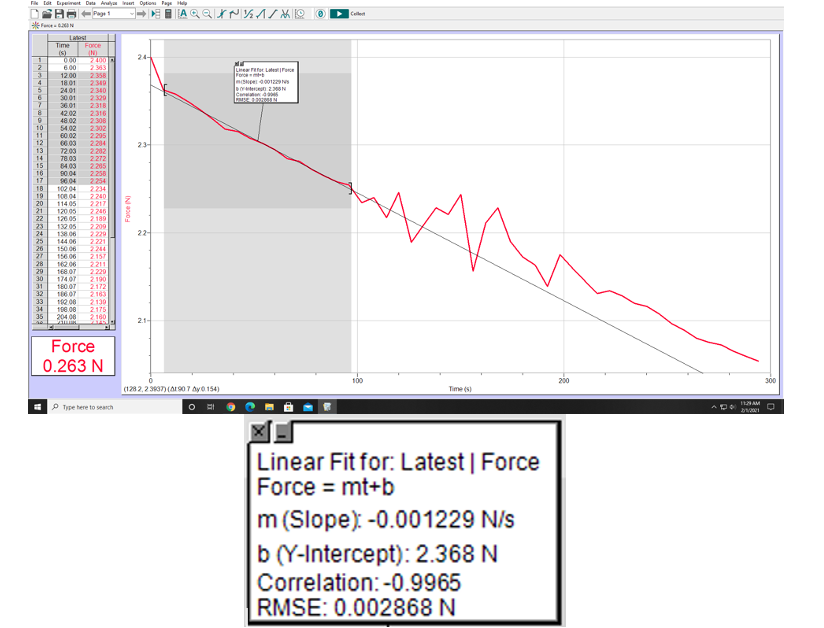
2.) This slope will give you the change in weight (Newtons) per second. Knowing the latent heat of vaporization for liquid nitrogen, determine dQ/dt. Use the value LV=2.00×105 J/kg. Show your calculations here.
2.) This slope will give you the change in weight (Newtons) per second. Knowing the latent heat of vaporization for liquid nitrogen, determine dQ/dt. Use the value LV=2.00×105 J/kg. Show your calculations here.
Chapter10: Atomic Physics
Section: Chapter Questions
Problem 41Q
Related questions
Question
2.) This slope will give you the change in weight (Newtons) per second. Knowing the latent heat of vaporization for liquid nitrogen, determine dQ/dt. Use the value LV=2.00×105 J/kg. Show your calculations here.
Transcribed Image Text:Fik tot Lapeinent Data Anlya aat Option Page H
A Page1
Cellet
Fane N
Latest
Time
Force
(s)
000
600
12.00
IN)
24-
2563
2358
230
2340
Line Per Latestroe
Forcemt
4
18.01
24.01
orer 260N
Canaan
30 01
36.01
2329
2318
42.02
4 02
54.02
60.02
66.00
72.00
78.03
2316
2308
10
2.302
2.205
11
12
2.204
2.202
2.272
2 205
23-
13
14
15
64.03
90.04
904
102 04
16
17
2.258
2254
2234
2 240
2217
2 246
2189
18
19
20
21
100 04
114 05
120 05
126 05
132 05
138 06
144.06
22
2 209
2 229
2.221
244
23
24
22-
25
26
150 06
156 06
27
28
29
30
2.157
162 06
168 07
17407
2.211
2.220
219
2172
31
32
180.07
110 06
168.00
204.06
2163
2130
2175
33
34
35
2 160
Force
0.263 N
100
200
300
(128 2, 2 3937) (A90 7 ayo 154)
Time (s)
I2AM
P Type here to search
Linear Fit for: Latest | Force
Force = mt+b
m (Slope): -0.001229 N/s
b (Y-Intercept): 2.368 N
Correlation: -0.9965
RMSE: 0.002868 N
IN aovo
Expert Solution
Step 1
Latent heat is defined as the quantity of heat required to convert a unit mass of a substance from one state to another at a constant temperature. The phase or state conversion takes place at a constant temperature.
Latent heat of fusion is the latent heat required for the solid-liquid transition while Latent heat of vaporization is the latent heat required for the liquid-vapor transition.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

University Physics Volume 3
Physics
ISBN:
9781938168185
Author:
William Moebs, Jeff Sanny
Publisher:
OpenStax


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

University Physics Volume 3
Physics
ISBN:
9781938168185
Author:
William Moebs, Jeff Sanny
Publisher:
OpenStax