2. Write the symbol for alpha, beta, and gamma radiation. Include the atomic mass and atomic number, where relevant. 3. Cobalt-60 releases one beta particle and two gamma rays per atom. Write the formula for the beta decay of Cobalt-60. 4. What insects were used in the Mythbusters test? 5. How much radiation is release by the cobalt-60 source? 6. According to the data, which dose or doses had immediate lethal effects to the insects? 7. Complete the fruit fly and flour beetle portion of the data table; Cockroaches(50) Fruit Flies (100) Flour Beetles (50) Control |0% 100% 100% |100K rad 60% 100% 100% 10K rad 40% 100% 100% 1K rad 30% 100% 100% Days 15 30 2 15 30 2 |15 30 8. What do you conclude? VWhat are the effects of nuclear radiation on insects?
2. Write the symbol for alpha, beta, and gamma radiation. Include the atomic mass and atomic number, where relevant. 3. Cobalt-60 releases one beta particle and two gamma rays per atom. Write the formula for the beta decay of Cobalt-60. 4. What insects were used in the Mythbusters test? 5. How much radiation is release by the cobalt-60 source? 6. According to the data, which dose or doses had immediate lethal effects to the insects? 7. Complete the fruit fly and flour beetle portion of the data table; Cockroaches(50) Fruit Flies (100) Flour Beetles (50) Control |0% 100% 100% |100K rad 60% 100% 100% 10K rad 40% 100% 100% 1K rad 30% 100% 100% Days 15 30 2 15 30 2 |15 30 8. What do you conclude? VWhat are the effects of nuclear radiation on insects?
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter14: Nuclear Chemistry
Section: Chapter Questions
Problem 14.36PAE
Related questions
Question
The video is called "Can Cockroaches Really Survive Nuclear Radiation? | Mythbusters"
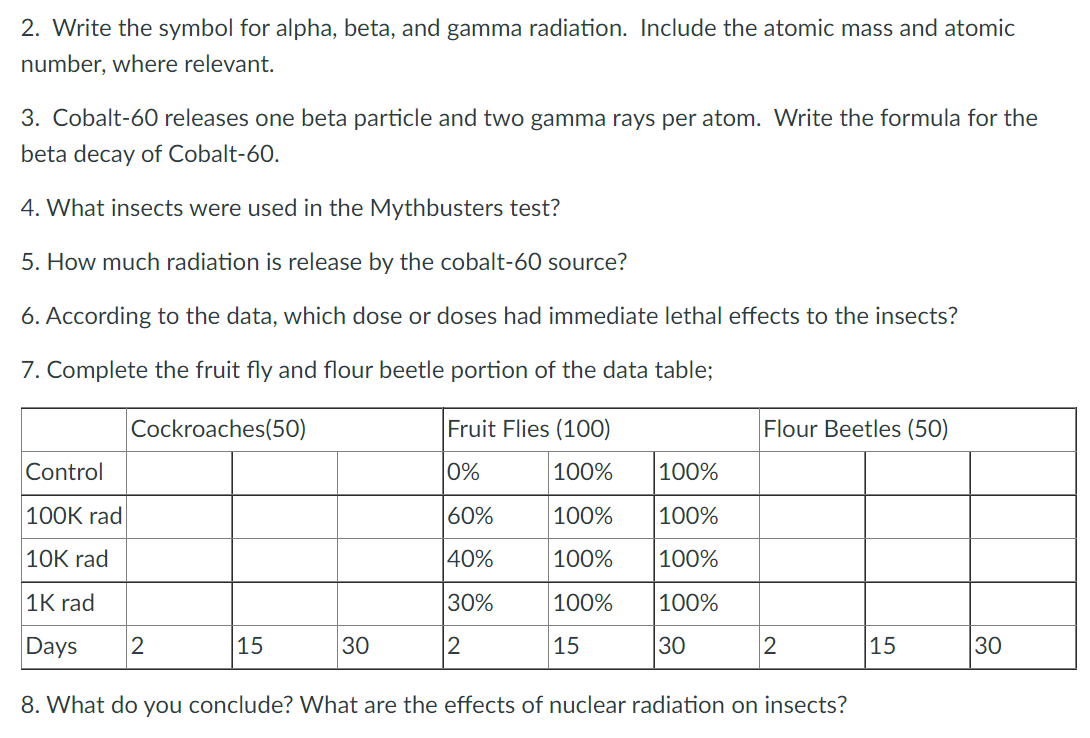
Transcribed Image Text:2. Write the symbol for alpha, beta, and gamma radiation. Include the atomic mass and atomic
number, where relevant.
3. Cobalt-60 releases one beta particle and two gamma rays per atom. Write the formula for the
beta decay of Cobalt-60.
4. What insects were used in the Mythbusters test?
5. How much radiation is release by the cobalt-60 source?
6. According to the data, which dose or doses had immediate lethal effects to the insects?
7. Complete the fruit fly and flour beetle portion of the data table;
Cockroaches(50)
Fruit Flies (100)
Flour Beetles (50)
Control
|0%
100%
100%
|100K rad
60%
100%
100%
10K rad
40%
100%
100%
1K rad
30%
100%
100%
Days
15
30
2
15
30
2
|15
30
8. What do you conclude? VWhat are the effects of nuclear radiation on insects?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning