22 of 39 MISSED THIS? Read Section 15.4 (Pages 642 - 649) ; Watch KCV 15.4, IWE 15.4.The decomposition of SO2Cl2 is first order in SO2Cl2 and has a rate constant of 1.40x104s-1 at a certain temperature. I Review | Constants | Periodic Table Part B How long will it take for the concentration of SO2C12 to decrease to 25% of its initial concentration? Express your answer using two significant figures. ? Submit Request Answer Part C If the initial concentration of SO, Cl, is 1.00 M. how long will it take for the concentration to decrease to 0.78 M? Express your answer using two significant figures.
22 of 39 MISSED THIS? Read Section 15.4 (Pages 642 - 649) ; Watch KCV 15.4, IWE 15.4.The decomposition of SO2Cl2 is first order in SO2Cl2 and has a rate constant of 1.40x104s-1 at a certain temperature. I Review | Constants | Periodic Table Part B How long will it take for the concentration of SO2C12 to decrease to 25% of its initial concentration? Express your answer using two significant figures. ? Submit Request Answer Part C If the initial concentration of SO, Cl, is 1.00 M. how long will it take for the concentration to decrease to 0.78 M? Express your answer using two significant figures.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 39QAP: The decomposition of azomethane, (CH3)2N2, to nitrogen and ethane gases is a first-order reaction,...
Related questions
Question
100%
![Review | Constants | Periodic Ta
MISSED THIS? Read Section
15.4 (Pages 642 - 649) ; Watch KCV 15.4, IWE
15.4.The decomposition of SO,Cl2 is first order in
SO2Cl2 and has a rate constant of 1.40x104s-1
at a certain temperature.
Part D
If the initial concentration of SO2C12 is 0.175 M, what is the concentration of SO2C12 after 220 s ?
Express your answer using threesignificant figures.
M
[SO,Cl2] =
Submit
Request Answer
Part E
If the initial concentration of SO, C2 is 0.175 M, what is the concentration of SO2C12 after 510 s ?
Express your answer using three significant figures.](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F370ea171-8bb9-4b07-85c5-04ae9f865ef9%2F4d50012c-2b3b-4853-91fc-f7e80d59b7c1%2Fmj0ofuu_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Review | Constants | Periodic Ta
MISSED THIS? Read Section
15.4 (Pages 642 - 649) ; Watch KCV 15.4, IWE
15.4.The decomposition of SO,Cl2 is first order in
SO2Cl2 and has a rate constant of 1.40x104s-1
at a certain temperature.
Part D
If the initial concentration of SO2C12 is 0.175 M, what is the concentration of SO2C12 after 220 s ?
Express your answer using threesignificant figures.
M
[SO,Cl2] =
Submit
Request Answer
Part E
If the initial concentration of SO, C2 is 0.175 M, what is the concentration of SO2C12 after 510 s ?
Express your answer using three significant figures.
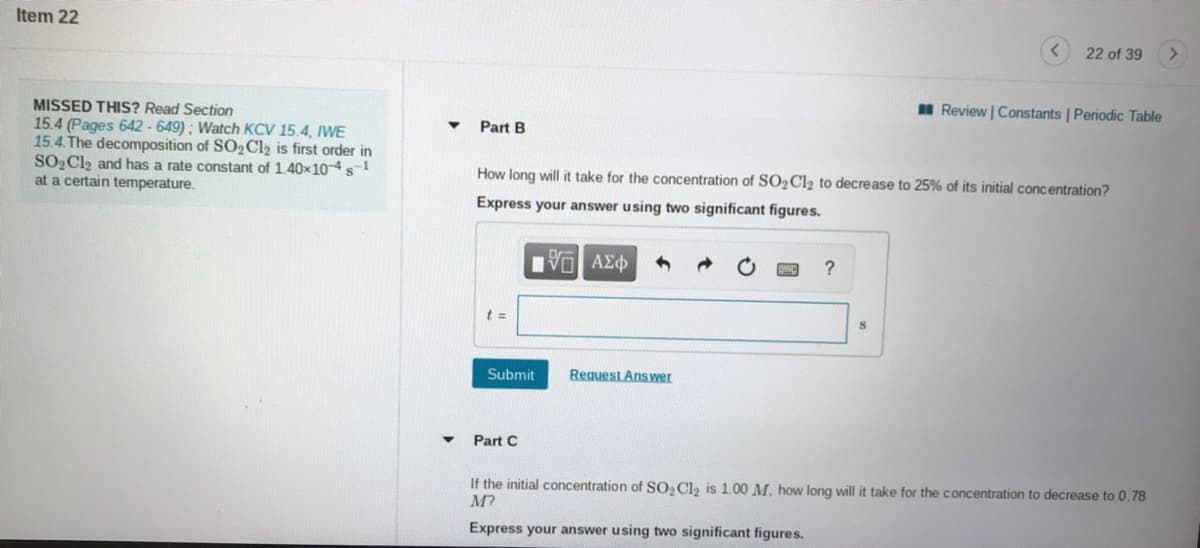
Transcribed Image Text:Item 22
22 of 39
I Review | Constants | Periodic Table
MISSED THIS? Read Section
15.4 (Pages 642 - 649) ; Watch KCV 15.4, IWE
15.4.The decomposition of SO2C12 is first order in
SO2C12 and has a rate constant of 1.40x104s1
at a certain temperature.
Part B
How long will it take for the concentration of SO2C12 to decrease to 25% of its initial concentration?
Express your answer using two significant figures.
Submit
Request Answer
Part C
If the initial concentration of SO, Cl, is 1.00 M. how long will it take for the concentration to decrease to 0.78
M?
Express your answer using two significant figures.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning