Q: Rank the following acids in order of increasing(weakest to strongest) acidity. I. 4-methoxybenzoic…
A: ->We can see order of acidity according to order of stability of conjugate base. ->If the…
Q: An aqueous solution contains 0.443 M hypochlorous acid. How many mL of 0.213 M potassium hydroxide…
A: Given, [HClO] = 0.443 M [KOH] = 0.213 M Volume of the solution = 125 mL pH of buffer = 7.530…
Q: What is the line formula of Alkane, Alkene and Alkyne if n=4?
A:
Q: A multivitamin sample has a label claim of 500 mg ascorbic acid (176.12 g/mol). According to quality…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: e the time this took.
A:
Q: 41. Pentaborane-9, BsH9, is a colorless, highly reactive liquid that will burst into flame or even…
A: Given, 2B5H9(l) + 12 O2(g) → 5 B2O3(s) + 9 H2O(l) The standard enthalpy of a reaction is:
Q: 1. The following data were obtained in an experiment in the reaction: A → products. Experiment 1 [A]…
A: Let us solve the question for 1st order reaction.
Q: Both silver(I) chloride and silver(I) bromide are slightly soluble salts, with Ksp values at 25°C of…
A:
Q: 8. Exceptions to the octet: Give the number of valence electrons and draw a Lewis structure for each…
A:
Q: Write the balanced nuclear equation for each of the processes described below. Ga decays by electron…
A:
Q: 6. What is a major product of the reaction sequence shown in the box? CH3 Br, or NBS KCN H20 ? Ilght…
A: N-Bromo succinimide (NBS) is the best reagent for allylic or benzylic bromination. The reaction…
Q: When heat is added to a solid, what will happen to the particles of that solid?
A: Solid is stable in shape or we can say it has a defined shape and volume . For example ; ice is a…
Q: Q8) A 0.532-g sample of an antacid tablet is dissolved in water. 30,00 mL of 0.141 M HCI solution…
A: Given, A 0.532-g sample of an antacid tablet is dissolved in water and 30.00 mL of 0.141 M HCI…
Q: A) How many grams of Mgl2 is present in a 0.4745 g sample that is 53.21% Mgl2 (278.11 g/mol)? B)…
A:
Q: SCH Aon go nue 2. Which of the following compounds ort ot contains a substituent that is ortho/para…
A:
Q: Q7) The indicator that is used for Vitamin C analysis is. a) Phenolphthalein b) 1₂ c)…
A:
Q: 7. For sequential reaction: A > B +C .(i) 2B »C+ 2D ..(ii) If % yield of (i) and (ii) reactions are…
A: Given: % yield of (i) reaction = 90% = 0.9 % yield of (ii) reaction = 80% = 0.8 Calculate : Overall…
Q: A sample of a gas originally at 38.9 °C and 0.794 atm pressure and 1.65 L, is subjected to a…
A:
Q: What is the molecular formula of Alkane, Alkene and Alkyne if n=4?
A: The Molecular formula of Alkane (n = 4) is C4H10.
Q: For sequential reaction: A » B + C ..(i) 2B »C + 2D ..(ii) If % yield of (i) and (ii) reactions are…
A:
Q: 7. Give the number of valence electrons and draw a Lewis structure for each molecule or ion. Use…
A: Total valence electrons of a molecule or ion is defined by the sum of the valence electrons of all…
Q: Write a chemical equation for the decomposition of hydrogen peroxide.
A:
Q: B CH3- CH- CH-C- CH;- CH3 1. CH CH3 CH3
A: The Answer to the following question is-
Q: A multivitamin sample has a label claim of 500 mg ascorbic acid (176.12 g/mol). According to quality…
A: Ascorbic acid (Vitamin C) is a monoprotic acid with formula HC6H7O6. Hence the balanced equation…
Q: What do you call the number that summarizes the factors affecting the particle size of a precipitate…
A:
Q: What do you call the equipment that produces sound waves to dissolve a solid? A Vortex mixer B)…
A:
Q: Find the pH of a 2.0 M solution of H2SO4. Ka = 1.1 x 10^-2
A: Given :- Concentration of H2SO4 solution = 2.0 M Ka of H2SO4 = 1.1 × 10-2 To find :- pH of…
Q: For each of the reactions, label the half-reaction tl is the anode and the reaction that is the…
A: Oxidation occurs at the anode and Reduction occurs at the cathode. 1) Cu(s) → Cu2+(aq) + 2e- = Anode
Q: 11. Which of the following acts as electrophile in the reaction of benzene with CH3CI in the…
A: Electrophile Organic species contains + ve and electron deficient species.
Q: - Hydrogen gas and oxygen gas release 482.6 kJ of heat when they combine to form steam. Is this…
A: Given : H2 + O2 ---> H2O Amount of heat released = 482.6 kJ To determine :- Whether the…
Q: For sequential reaction: A >B + C ...(i) 2B —> С + 2D ...(ii) If % yield of (i) and (ii) reactions…
A: To find the overall percent yield for a sequential reaction, we simply have to multiply the…
Q: 1. In this lab, FENCS" is being formed as a complex ion (complexation product) absorption peak at…
A: FeNCS2+ is a transition metal complex in which the oxidation state of iron is + 2. In the formation…
Q: A multivitamin sample has a label claim of 500 mg ascorbic acid (176.12 g/mol). According to quality…
A: After 6 months - NaOH required was 22.48mL and 0.1101M. Number of moles of NaOH, n = Molarity *…
Q: 1. What are the four ways presented in this lesson to influence the rate of a chemical reaction?…
A: Collision theory states that the rate of a chemical reaction is directly proportional to the number…
Q: Will a precipitate form when 80.0 mL of 2.00 x 102 M Cu(NOs)2 and 20.0 mL of 2.00 x 10 M NalO, are…
A: Cu(NO3)2 + 2 NaIO3 gives Cu(IO3)2 + 2 NaNO3 Cu(IO3)2 is not soluble in water and must be…
Q: pl functional group:
A:
Q: solve question Q9) After adding precise amount of HCI to antacid tablet, the contents of the flask…
A: #Q.9: The main constituent in an antacid is sodium bicarbonate, NaHCO3.
Q: The cell potential is equal to zero at equilibrium. Group of answer choices True False
A:
Q: Give the principle and general reaction involved in Beilstein test.
A:
Q: Describe the overall reaction order for the chemical reaction with rate law v = k[A][B]?[C]³ when. B…
A:
Q: Calculate the E cl for the following reactions and state if the given cell is either a galvanic or…
A:
Q: A basic solution contains the iodide and phosphate ions that are to be separated via selective…
A: Dissolution equillibrium of AgI AgI(s) ⇒ Ag+(aq) +…
Q: 7. For sequential reaction: А —> В +С ..(i) 2B —> С + 2D ...(ii) If % yield of (i) and (ii)…
A: Overall yield of a multistep reaction is the product of the percent yield of all the steps as shown…
Q: 13. Draw the reaction mechanism and the Major product of the reaction. Explain the regioselectivity.…
A: Here we have to write mechanism and major product in the following reaction.
Q: 3) The correct reading of the given graduated cylinder including the uncertainty is a) 5.4 201…
A: 2) Beaker , it is not calibrated to measure volume accurately 3) Correct option is (d) 5.5 ± 0.1 ,…
Q: Which cubic unit cell has the largest number of equivalent atoms?
A:
Q: Copper(II) acetate containing copper-64 is commonly used to study brain tumors. This isotope has a…
A: Radioactive decay follows the first-order kinetics. The decay constant (λ) or rate constant and…
Q: H OSIET3 OSIME3 МеОН H2N + Lewis acid (cat), CH3CN
A:
Q: A multivitamin sample has a label claim of 500 mg ascorbic acid (176.12 g/mol). According to quality…
A: For initial sample- NaOH required was 25.40mL and 0.1101M. Number of moles of NaOH, n = Molarity *…
Q: In chemistry, what does it mean when the ratio of the overall Fe in Fe2O3 and Fe3O4 is unity?
A: we are required to find % of Fe in both iron oxide and then compare the % of Fe
Step by step
Solved in 2 steps with 1 images

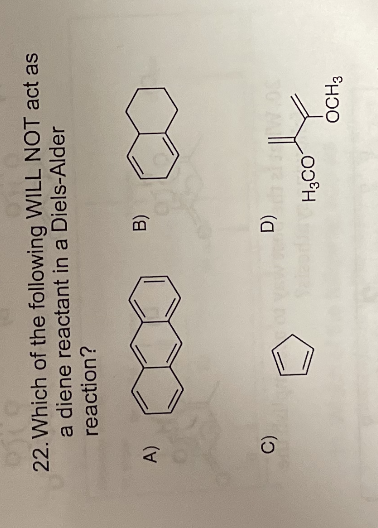
- (a) Why cyclohexene and cyclohexa-1,3-diene are expected to undergo addition reaction but benzene does not? Explain clearly. (b) Why is an electron-donating group to benzene activating and usually a/an ortho/para director?Which of the following compounds cannot react as a diene in a Diels-Alder reaction ( question to the E3 image)Consider a traditional Diels Alder reaction noting it's methodology and synthetic tactical. Then propose an alternative, greener synthetic route to make the same product with near or same enantioselectivity.
- Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.What diene and dienophile are needed to prepare attached compound by aDiels–Alder reaction?Please Explain in details and steps for this Diels-Alder reaction and the obtained product. i want the full mechanism also. thank you
- Select the product this Diels-Alder reaction?Compounds containing triple bonds are also Diels–Alder dienophiles. With this in mind, draw the products of each reaction.What is the product E and how is it formed? Can this be explained using frontier molecular orbital analysis? What would the regiochemistry and stereochemistry be?
- Draw the products of the attached Diels–Alder reactions. Indicatestereochemistry where appropriate.Refute or Defend: Once cyclopentadiene is distilled, it can be stored for a long period of time (e.g., weeks) before using it in a Diels-Alder reactionThe molecule in the box is the product of a diels alder reaction. Which of these is the dienophile in that reaction ? A B C D or E?

