Q: THPO. 1. Piperidine, cat. H* 2. 3 HzO+, H2O HO. 1. &x CI
A: First reagent react with carbonyl group to form enamine formation.Enamine react with reagent (2) in…
Q: For the following reaction, calculate the value of Q. 1A →→ 3B + 4C [A] = 0.38 [B] = 0.30 [C] = 0.10
A: Concentration quotient (Q) or reaction quotient (Q):At any stage of the reaction, other the state…
Q: A fuel mixture used in the early days of rocketry consisted of two liquids, hydrazine (N2H4) and…
A: The objective of the question is to determine the mass of excess reactant that remains unreacted.It…
Q: In Universe L, recently discovered by an intrepid team of chemists who also happen to have studied…
A:
Q: A chemist prepares a solution of aluminum sulfite flask to the mark with water. Calculate the…
A: The amount of aluminum sulfite given is .The volume of solution is .To find the concentration in…
Q: In an E2 mediated elimination reaction of an alkyl halide (H-X) using NaOCH3 as base, which of the…
A: The objective of the question is to find the false statement from the following options given:. The…
Q: How are grams converted to nano grams?
A: To convert grams to nanograms, you can use the following conversion factor:1 gram=1, 000, 000,000…
Q: The compound below is treated with chlorine in the presence of light. H3C CH3 Xon H3C CH₂CH3 Draw…
A: Alkanes can be halogenated in the presence of light or heat. The alkanes do not contain any…
Q: Reaction C. N HCI Draw the product of reaction C. Select Draw Templates More ||| ||| C H N
A: An acid–base reaction is a type of chemical reaction that involves the exchange of one or more…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. 1. KMnO4, H₂O, heat 2. H*…
A: In the given question, the reactants and reagents of an organic reaction are given and it has been…
Q: An SN2 reaction takes place where a nucleophile displaces a leaving group at a stereocentre. If the…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: Doctor’s order: Sulfasalazine Oral susp 500 mg q 6 hr; Directions for mixing: Add 125mL of water and…
A: To determine how many milliliters (mL) of the Sulfasalazine oral suspension you need to give, first,…
Q: PROCEDURE Part A: Fill a 600 mL beaker about halfway with ice. Add a small amount of water (about 50…
A: Part A:Take a 600 mL beaker and fill it halfway with ice. Add about 50 mL of water and then the rock…
Q: 3:{- CH3 A. B. H3C H3C -H (±) FO (+) C. D. H H CH3 (±) CHẠO (+) Draw the approach of the dienophile…
A:
Q: The glass electrode used in commercial pH meters gives an electrical response proportional to the…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: The Hamiltonian operator for H₂ in the Born-Oppenheimer approximation is ....
A: We have to find Hamiltonian operator for H2+ in Born-oppenheimer approximation.
Q: НО. CI CI (A) I only (B) I and II (C) II and III (D) I, III and IV (E) IV only H E H H H H НО. HO CI…
A: A carbon that has 4 different atoms or groups attached to it.
Q: Which of the indicated bonds absorbs at a higher wavenumber in an IR spectrum? Select the single…
A: Analytical chemistry uses infrared (IR) spectroscopy as a method to examine how molecules interact…
Q: Consider the balanced chemical equation: 3 H ₂(g) + N₂(g) → 2 NH₂(g) If 46.8 g H₂ and 179.4 g N₂ are…
A:
Q: Why can some insects "walk" on water? Select all that apply. due to viscosity due to surface tension…
A: The question is asking about the physical properties of water that allow certain insects to 'walk'…
Q: Draw the major addition product for this reaction. Do not include any byproducts formed. HBr (1…
A:
Q: What is the XNaOH in an aqueous solution prepared by dissolving 8.00 g of NaOH in 100.0 g of H₂O?…
A: The formula for the mole fraction of NaOH is
Q: Solve each problem using Dimensional Analysis, as demonstrated to you in class. Every number must…
A: The conversion of one unit to another unit is done in such a way that the units in the numerator and…
Q: The pKa,1 and pKa,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below:…
A: Strong acids have large values of Ka and small values of pKa whereas weak acids have small values of…
Q: 5. Draw all possible conjugate bases: O For NH₂ a. b. 6. Draw all possible conjugate acids: a. 1+ b.…
A: A conjugate base is a substance formed when an acid loses a proton. When an acid, represented as HA,…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: Sodamide is a strong base, which abstracts beta-hydrogen to form corresponding alkene. The given…
Q: In the following reaction, what is the major product formed? HBr Br Br Br Br
A: Given that, the reaction is:
Q: Everything is correct except for the structure of the conjugate acid, please help! Be sure to answer…
A: according to Bronsted Lowry theory, Acid is a substance which can give up a proton and form…
Q: Draw a structural formula of the SS configuration of the compound shown below. S S CH3 CH CH₂CH3 ОН…
A: Given that, the structure of the compound is:
Q: What is the expected product of the reaction shown? и НО OCH3 OI O II O III OIV O none of these H3CO…
A: Given is organic reaction.The given reactant is epoxide.The given reaction is epoxide ring opening…
Q: Choose condensed structure for C3H6O containing a ketone functional group. O CH3 COCH3 O CH3CH,CHO O…
A: Condensed structure formula :*A system for writing organic compounds in a single line is known as…
Q: One of the Freons, CCI₂F2, is manufactured from carbon tetrachloride and hydrogen fluoride. The…
A: Given,when CCl4 reacts with HF to form CCl2F2 and HCl.mass of CCl2F2 = 117 kgmass of CCl4 = 183…
Q: Which of the following would not be seen in the splitting patterns for the ¹H, NMR signals for the…
A:
Q: Ligand L binds to a binding protein with a K of 400 nM. Calculate the ligand concentration based on…
A:
Q: The chemical environment of carbons can be deduced form their chemical shifts. Deduce the relative…
A: Each unique carbon in a compound produces a signal in CNMR spectra.The value of the signal depends…
Q: If 200000 molecules of protein A are present in the eukaryotic cell what would be the micromolar…
A: Molarity (M): Molarity is the number of moles of a solute in one liter of solution and is used as a…
Q: Draw the products of the reactions shown below. Ignore inorganic byproducts. TBDMSCI (1 eq) elect to…
A: TBDMSCl acts as a protecting agent. It selectively protects primary alcohol ( 1°- alcohol) due to…
Q: Question #3 When treated with 2-butene-1-ol in the presence of an acid catalyst, 1 is…
A: To draw the probable mechanism for the given transformation
Q: An analytical chemist is titrating 160.9mL of a 0.6300M solution of ethylamine C2H5NH2 with a…
A:
Q: Draw the products of the reaction shown below. Use wedge and dash bonds to indicate stereochemistry.…
A: The objective of the question is to find the major product in which alkene reacts with OsO4 in the…
Q: The rate constant of a chemical reaction is 8.04 s-1 at a temperature of 27.9 \deg C. If the…
A:
Q: (b) Using the Crystallographer's formula to determine the density (in g/cm³) of Po: ρ = Σ Ζ * Μ…
A: Answer:Here:
Q: Draw structural formulas for the alkoxide ion and the alkyl(aryl)bromide that may be used in a…
A: Alkyl bromide reacts with alkoxide ion to form an ether by the Williamson ether synthesis. The…
Q: The K₂ of acetic acid is 1.8 x 10-5. Percent Abundance 50%- nying the species present in all aqueous…
A: Given:Complete the abundance diagram by identifying the species present in a 1.00 M aqueous solution…
Q: Phosphate-buffered saline (PBS) is a solution commonly used in studies of animal tissues and cells.…
A: pH =?osmolarity =?
Q: Sample Exercise 5: Calculation of Mole Fraction and molality An aqueous solution of HCl contains 36%…
A: Given,%mass of HCl = 36 %
Q: 1. Predict the products for the reactions below, and provide reasonable mechanisms for their…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The equilibrium constant, Kp, for the following reaction is 0.160 at 298 K. 2NOBr(g) 2NO(g) + Br₂…
A:
Q: From the following reaction, please select the major endo product. Reminder, only pi orbital…
A: Reactants are diene and dienophile. This is Diels Alder reaction.Dienophile contains electron with…
Q: 2) For each compound below, identify the atom most likely to be attacked first by a nucleophile and…
A: When a atom is attracted by the nucleophile means the atom is electronically defficient.
What compound is indicated by this 1H NMR spectrum?
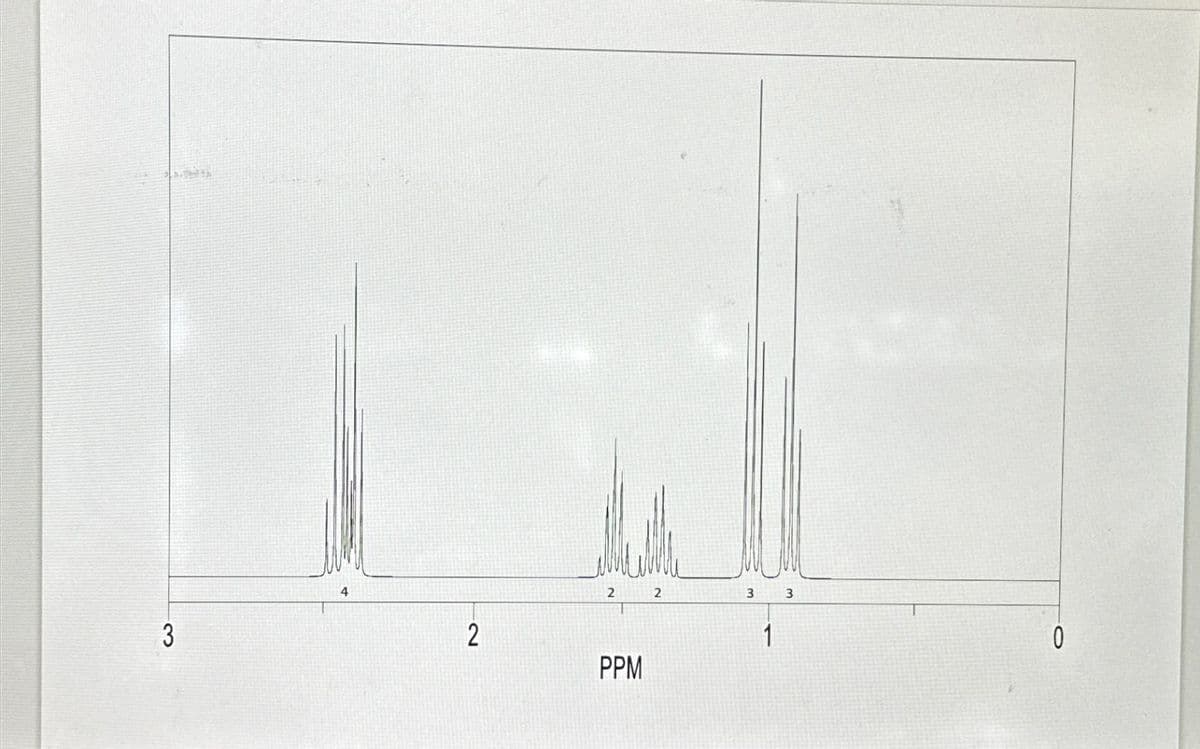
Step by step
Solved in 3 steps with 1 images


