Q: Use the spectral data below to identify one of the compounds below
A: We can identify the compound by counting the total number of hydrogens present in the compound and t...
Q: grade 12 chem: include units, keep work as precise as possible and apply sig figs only to final ans...
A:
Q: 0.04 M NaH2AsO4 , 0.05 M NaH2AsO4 Which is more acidic? why is that ? H3AsO4 için Ka1= 5.8*10-3 Ka2...
A: With the increase in the concentration of salt, pH value decreases. As both the salts are NaH2AsO4 a...
Q: can you answer question 12 now
A: sodium ethanoate dissolves into water, the change in enthalpy is ΔH = –17.3 kJ/mol.
Q: What mass of sodium nitrate required to prepare 1.52 L of 2.41 M of solution of sodium nitrate?
A: Sodium nitrate is the chemical compound. Its chemical formula is NaNO3 .This metal nitrate is als...
Q: 0.04 M NaH2AsO4 , 0.05 M NaH2AsO4 Which is more acidic? why is that ? H3AsO4 için Ka1= 5.8*10-3 Ka...
A: We have to tell which is more acidic in 0.04 M NaH2AsO4 , 0.05 M NaH2AsO4 Given Ka1= 5.8*10-3 K...
Q: Please help me I am working on some practice and was confused
A: In a chemical reaction, two or more reactants react with each other to form new substances. This new...
Q: Choose the correct option. Guess the type of polymerization if you are provided with azobisisobutyro...
A: Polymerization is the process of combining small molecules to form macromolecules of higher mass cal...
Q: Please explain to me the correct choice.
A: Diels Alder reaction: It is a 4+2 cycloaddition reaction between a diene and dienophile in thermal c...
Q: Write the letter of the pair of compounds that illustrates the given isomerism. 1. Enantiomerism: ...
A: Isomerism is the property exhibited by compounds with the same molecular formula but differs in the ...
Q: Give the electron-pair and molecular geometries for NF3 and XeF4.
A: Molecular Geometry: Molecular geometry is defined as the process of in which lone pair and bond pair...
Q: Vibrational spectroscopy has played a role in supporting the pentagonal bipyramidal structure of the...
A: The structures of trans and cis IO2F5 2- are shown below.
Q: Which property of the two enantiomers of 1-phenylethanol would you expect to be different? Group o...
A: The question is based on the concept of isomerism. We have to identify which of the given property d...
Q: Aside from methane, carbon dioxide, water vapor, and nitrous oxide, give two more examples of greenh...
A: Greenhouse effect: It is a natural process by which earth surface gets heated. Few gases are respons...
Q: Consider the reaction: P,010(s) + 6H2O(1)4H;PO,(aq) Using standard thermodynamic data at 298K, calcu...
A:
Q: number (1) only
A: We have to write iupac name for this we have to follow some steps
Q: please help me with this I am very confused and want to study.
A: Note: Since you have asked multiple questions, we will solve the first for you. If you want any spec...
Q: Calculate the rate of appearance of CO2 when [NO2] = [CO] = 0.550 M. х 4.5 x 10 -5 M/s
A:
Q: Time le Due the following reaction, the main by-product is NaBr, H2SO, ОН H3C a) H3C Br b) NaHSO4 CH...
A: The given reactant is Butanol. The product formed from the given reaction is,
Q: Determine the point groups for A snowflake .
A: Point groups are determined by getting symmetries by keeping one point fixed. The point group is a ...
Q: Identify the functional groups in the given organic compound: H;C NH2 Select the correct response: E...
A:
Q: Which of the following gases will have the slowest rate of effusion at constanttemperature?a. H2b. F...
A: The process by which a gas escapes from a container through a very small hole. It leads to the gener...
Q: Given the reaction below, which of the following is the overall balanced redox equation? (Assume tha...
A:
Q: the thin structure that surrounds a cell is known as
A: The structural and functional unit of life that is the basic unit of an organism is termed the cell...
Q: For many purposes we can treat butane C4H10 as an ideal gas at temperatures above its boiling point ...
A: At constant volume if the pressure of the gas is increased, temperature of the will increase. This i...
Q: Please help me with both of these, I am very confused and would like to study
A: In a chemical reaction, reactants undergo a reaction forming the products. Ammonia is a good reagent...
Q: Propane burns according to the thermochemical reaction below. C,H,(g) + 50,(g) 3 CO,(g) + 4H,0 ĄH =-...
A:
Q: What is the vapor pressure of water at 10 degree Celsius, at 25 degree Celsius And at 60 degrees Cel...
A: Vapor pressure of water is the pressure at which the condensed state of water and water vapor are in...
Q: If the partial pressure of O2 is 38.mmHg , the mass of O2(in mg) that can dissolve in 1.00L water at...
A: Given: Pressure = 38 mm Hg Convert mm Hg into atm as follows. Volume = 1.00 L Temperature = 25 degr...
Q: show the complete solution and express the final answer with significant figures A. FREEZING POINT D...
A: Introduction: Depression in freezing point: The freezing point depression in the lowering of freezin...
Q: Determine whether each acid is a binary acid or an oxyacidand name each acid. If the acid is an oxya...
A: Binary acids has hydrogen atom and nonmetal. Oxyacid's contains oxyanions, which are nothing but ani...
Q: Thanks for helping! THIS is the equation N2H4 + O2 → N2 + H2O First just Use bond energy values from...
A: The energy values (given) are: N-N = 159 kJ/mol N-H = 391 kJ/mol O-H = 463 kJ/mol O=O = 495 kJ/mol N...
Q: Which of the four constitutional isomers of C4H9F shown here has the highest pKa? Explain your answe...
A: Halo alkanes are compounds having a halogen atom attached to an alkane. Halogens contain fluorine, c...
Q: Launch the Concept Map by clicking the button below. Read the instructions that appear on the new sc...
A: CHEMICAL REACTIONS: A process in which one or two or more reacting species involve in a reaction to ...
Q: Match the following reactions as to which one would give the best, middle, and worst conversion. он ...
A: The compound in which -C(=O)-O- bond is present is known as an ester. The compound in which -C(=O)-N...
Q: A 116-mg sample of Na2CO3 is dissolved in H2O to give 2.05 L of solution.What is the concentration o...
A: ppm( parts per million): It is one of the best methods to determine the concentrations of the compou...
Q: How many grams of solid aluminum sulfide can be prepared by the reaction of 10.0 g of aluminum and 1...
A:
Q: 3 PbO2 + 2 Mg3P2 ? Pb3P4 + 6 MgO How many moles of PbO2 do you need to produce 80 moles of MgO? How ...
A: From the balance equation the conversion factor relating PbO2 and MgO is,
Q: Chemistry Question
A: In organic chemistry, there are many groups of atoms present in any molecule that give a characteris...
Q: Qno1
A: The balancing of the chemical equation is done to make equal contribution of the atoms in the reacta...
Q: How many L of a 0.1500-M solution of KOH will be required to titrate 40.00 mL of a 0.0656-M solution...
A:
Q: Mr. Reyes is in the lab and reacting Ethene gas with Hydrogen gas to form Ethane (C2H6). Mr. Reyes w...
A: The reaction between the ethene gas and H2 gas produces the ethane gas and gives off energy in the f...
Q: number 5 only
A: Positional isomers are defined as the isomers having the same molecular formula but they differ in t...
Q: Consider the reaction below. A reaction mixture at 600K initially contains [CO]=0.500M and [H2]= 1.0...
A: Given Mixture initially contains [CO]=0.500M and [H2]= 1.00M Equilibrium concentration of [CO] = 0.3...
Q: Is resorcinol soluble in water? HCl? NaOH? NaHCO3? Why?
A: RESORCINOL: It is an organic chemical compound and it is classified as a dihydroxy phenol. Resorcino...
Q: Which of the following compounds is expected to be a major product of the following reaction? Select...
A:
Q: For the reaction CO(g) + Cl2(g)–COCl2(g) AG° =-63.0 kJ and AS° = -137.3 J/K at 330 K and 1 atm. The ...
A: Gibbs free energy is the greatest work a system does to its environment when the reaction is reversi...
Q: Is cyclopentanamine soluble in water? HCl? NaOH? NaHCO3?
A: Cyclopentamine is a cyclic ammine. It is a sympathomimetic alkylamino. It is classified as a vasocon...
Q: Answer number 2,3 and 4.
A: 2. Based on the solute solubility, if the solution contains a lesser solute than the maximum amount ...
Q: The combustion of diesel, C20H42, has an enthalpy change of –16180 kJ for the unbalanced reaction gi...
A: The enthalpy change of the reaction is -16180 kJ. The volume of diesel is 50.00 L and the density of...
Give the systematic name of the given compounds (number 3 only)
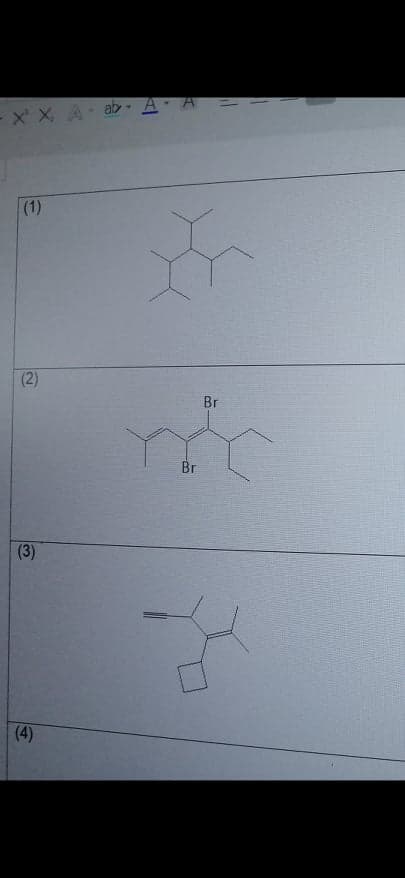
Step by step
Solved in 2 steps with 2 images


