Chapter8: Alkenes: Reactions And Synthesis
Section8.SE: Something Extra
Problem 47AP
Related questions
Question
What is the mechanisms to get fron the reactant to the products.
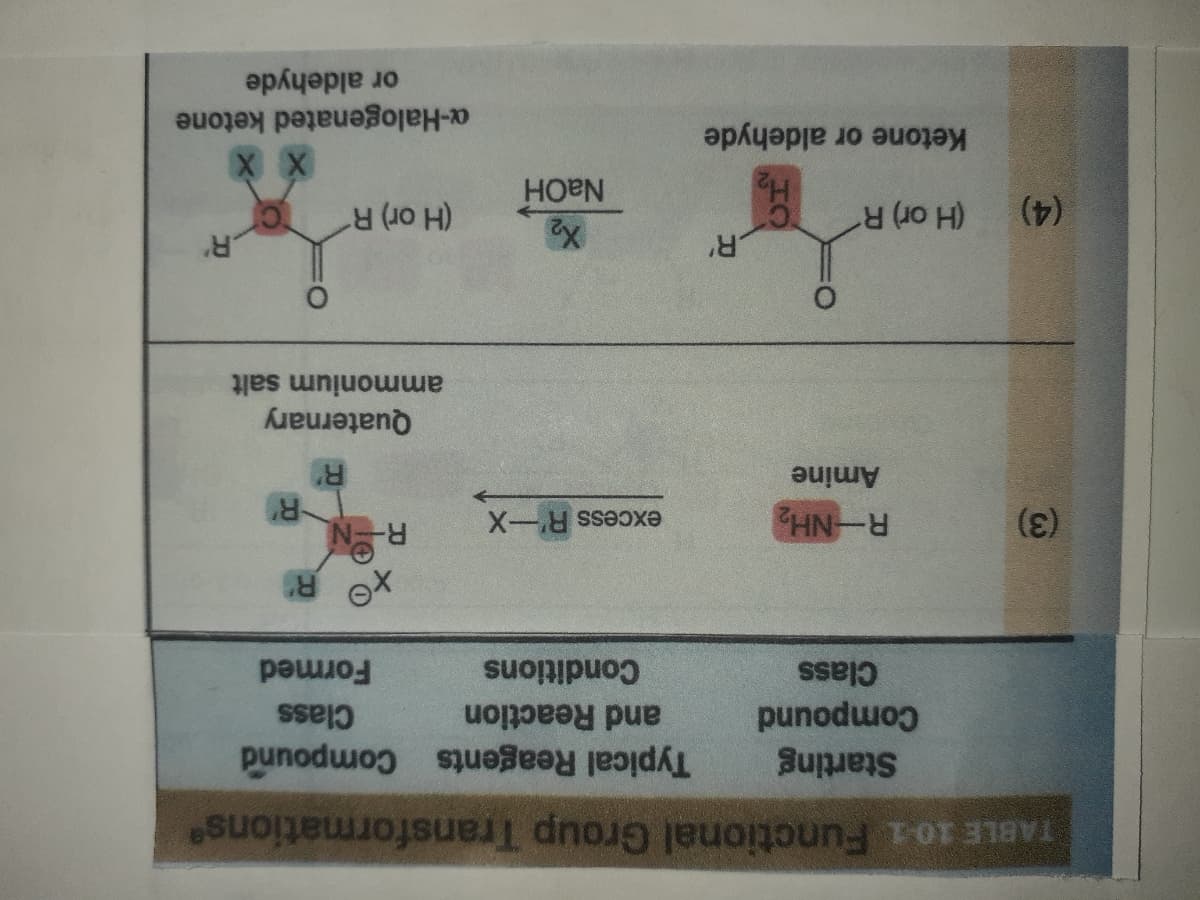
Transcribed Image Text:TABLE 10-1 Functional Group Transformations
Starting
Typical Reagents Compound
punodw
Class
and Reaction
Class
Conditions
Formed
R-NH
excess R'-X
Amine
Quaternary
ammonium salt
(H or) R
(H or) R
(4)
Ketone or aldehyde
NAOH
a-Halogenated ketone
or aldehyde
(3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning