Q: TICE The data below refers to an unknown substance, X. Boiling Heat of Heat of Point Fusion…
A:
Q: In each of the molecules drawn below one chemical bond is colored red. Decide whether this bond is…
A: Chemical bonds are of two types mainly - polar and non-polar. A polar covalent bond arises due to…
Q: 4) How many atoms of carbon are in 10 grams of carbon dioxide2 5) What is the mass of water if it…
A:
Q: Task 3.2. Calculate the standard enthalpy of reaction of some thermochemical equations governed by…
A:
Q: A 100.0-g sample copper is heated from 10.0°C to 100.0°C. a. Determine the number od calories…
A: Given: The mass of the copper sample = 100.0 g The initial temperature = 10.0 °C The final…
Q: 6. A 0.331 g sample of impure aspirin prepared in the lab was dissolved and reacted (neutralized -…
A: Given -> Weight of impure sample = 0.331 gm Weight of NaOH = 0.0716 gm
Q: Write the correct chemical formula for 0.10 M lithium nitrite
A: Lithium : The atomic number of Li is 3 and belongs to IA-group. The electronic configuration of Li :…
Q: Complete and balance the following equation then write the net ionic equation. Fe(NO3)3+ K3PO4 -->
A: Complete and balanced reaction is as follows: FeNO33aq + K3PO4aq →FePO4s + 3KNO3aq
Q: 11.16 Draw the condensed structural formula for each of the follow- ing alkanes: 3-ethylpentane a.…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first three parts…
Q: Iron(II) sulfate, is typically the source of iron in tablets prescribed to anemic patients, a…
A: Mass of the Fe2+ tablet taken = 500 mg During the redox reaction, Fe2+(aq) is oxidized to Fe3+(aq),…
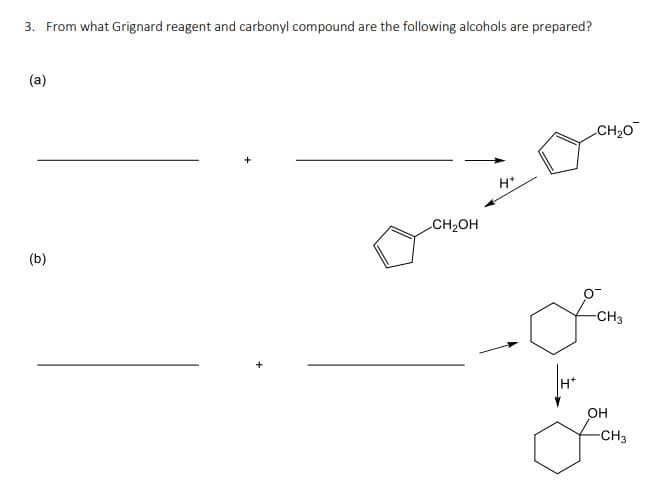
Q: Show how you would prepare the following molecules from the indicated starting material More than…
A: The protection of a typical 1,3-diol with benzaldehyde takes place in acidic media. Using a mixture…
Q: 7. a. Determine the empirical formula for a compound that contains: 95.21 g Carbon 4.79 g Hydrogen…
A: Note: As per our guidelines, we are supposed to answer only one question when multiple questions are…
Q: The following series of reactions were carried out. PbCO3(s) + 2HNO3(aq) → Pb(NO3)2(aq) + H₂O(1) +…
A:
Q: calculate the ratio of conjugate base to acid using the henderson-hasselbalch equation. to…
A:
Q: A₂ = A + A (k₁ and k-₁) A + B → P (K₂) i) Solve for dP/dt using a pre-equilibrium assumption ii)…
A:
Q: Based on the graph below, a mixture of benzene and toluene, which contains 20 mol % of the lower…
A:
Q: This fully fused-ring molecule is called "archimedene". It was prepared for the first time in 2002…
A:
Q: Calculate the temperature (K) at which a reaction becomes spontaneous when ΔS = 769.8 J/mol-K and ΔH…
A:
Q: molecule or polyatomic ion H₂O Br₂ CCI polar or nonpolar? polar nonpolar O polar O nonpolar O polar…
A:
Q: Draw the detailed mechanism and predict the major product of each of the following reactions. O₂N.…
A:
Q: b. C. CH,—Br + CH,—CH-NH–CH, + NaOH CH₂ ANSWER: CH –Br + CH —CH—NH CH; + NaOH CH, CH₁ CH₁ C-Br + NH,…
A:
Q: From the given structure, indicate the most stable radical.
A: Organic reactions are those in which organic reactant react to form organic products. Free radical…
Q: Classify the following substituents according to whether they are electron donors or electron…
A: Inductive effect arises due to a permanent shift in dipole moment towards the more electronegative…
Q: The equilibrium constant, Kc, for the following reaction is 55.6 at 698 K: 22HI(9) Calculate the…
A:
Q: A brick has a mass of 4.0 kg and the Earth has a mass of 6.0 × 10²7 Use this information to answer…
A:
Q: -12 A virus has a mass of 9.0 × 10 mg and an oil tanker has a mass of 3.0 × 107 kg. Use this…
A:
Q: What is the reticular energy? Mg²+ (g) + 0(g) AH2nd IE (Mg) = +1451 Mg*(g) + 0(g) Mg(g) + 0(g) Mg(g)…
A: We have to calculate Reticular energy or lattice energy
Q: Which of the following description(s) are improperly used related to the process of filtration?…
A: Filtration is the process in which solid particles in a liquid are removed by the use of a filter…
Q: We worked a previous question on an adiabatic irreversible transition that had 0.0101 moles of gas…
A: To calculate the ∆S, ∆SSurrounding, ∆Stotal for an irreversible adiabatic transition given that…
Q: When a metal at a higher temperature is transferred to water at a lower temperature, heat will be…
A: When heat is lost to the calorimeter and the surrounding air then final temperature of the mixture…
Q: Choose the atom(s) that are chiral centers in this alkane. the 3 2 A) 1 B) 2 C) 3 D) 4 E) 3 and 4
A: Chiral carbon: The sp3 hybridized carbon atom that is attached to the four different types of atoms…
Q: The formation of products in a gas-phase rxn is affected by A. Amount of reacting molecules B. All…
A: The amount of product formed depends upon the amount of reacting molecules in every phase reaction.…
Q: For the following reaction, 32.8 grams of calcium hydroxide are allowed to react with 28.7 grams of…
A: Given Amount of calcium hydroxide reacts = 32.8 g Amount of hydrochloric acid used = 28.7 g Molar…
Q: Write the balanced ionization reaction for CuSO4
A: Ionization reaction are those reaction in which ions is resent in cation as well as anion form.
Q: Determine the number of atoms of O in 86.4 moles of Al₂(SO₄)₃.
A:
Q: a. What are (ΔS), (ΔS surroundings) and (ΔS total) for a mole of perfect gas that doubles its…
A: In thermodynamics, the letter 'S' denotes entropy which is a measurement of randomness. Thus, by the…
Q: Question 3 Calculate the temperature in °C that would be required to double the reaction rate (i.e.…
A: Given -> T1 = 17.2°C = 17.2 + 273 = 290.2 K K1 = 1.2 × 10-3 s-1 Activation energy (Ea) = 30.0…
Q: An adventurous archaeologist cros- ses between two rock cliffs by slowly going hand over hand along…
A:
Q: How many moles of Ag2SO4 are in a sample that contains 1.06 x 1023 molecules? How many grams are in…
A:
Q: 3. Calculate AH for the reaction CH₂+NHHCN+ 3 H₂given: N₂+ 3 H₂ -2 NH C+2H₂CH H₂+2C+N₂ → 2 HCN…
A:
Q: There are only two naturally occurring isotopes of Copper, 63 Cu and 65 Cu. Copper has an atomic…
A:
Q: Enantiomers are a pair of molecules that are non-superimposable mirror images. Identify the two…
A: Stereomers are the isomers having same molecular formula and same sequence of bonded atoms but they…
Q: 2 N2(g) + 5 O2(g) → 2 N2O5(g) The rate of disappearance of N2 is 0.368 M/min. What is the rate of…
A:
Q: n each row, pick the compound with the bigger lattice energy. lote: lattice energy is always greater…
A:
Q: st the suitable reagent d to bring about the fa tic transformations
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Show how you would prepare the following molecules from the indicated starting material. More than…
A: In this reaction secondary alcohol group of cyclic D-glucose is converted into AzideN3 group. Only…
Q: QUESTION 1 Predict the reagents needed to accomplish the following transformation. If more than one…
A: Kindly repost your questions other than (1)
Q: Given 2NOBr(g) -> NO(g)+Br2(l) The reaction is second order and rate constant is 1.45 1/Ms at 20.0…
A: Given that, A second-order reaction is 2NOBr(g) → NO(g) + Br2(l). Also, the rate constant is k =…
Q: Classify the following chemical reaction: K2S (aq) + Co(NO3)2 (aq) ⟶ 2 KNO3 (aq) + CoS (s)
A:
Q: ΔG° for the reaction shown is equal to -698.9 (kJ/mol). Use the information given in the table to…
A: We have been given a reaction and we also have been given the standard free energy of formation…
Step by step
Solved in 2 steps with 1 images

- Draw and name all constitutionally isomeric alcohols with the molecular formula C4H10ODraw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OH
- Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] OsO4 + NMO; [2] NaHSO3, H2O1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.Alcohol A (C10H18O) is converted to a mixture of alkenes B and C on being heated with potassium hydrogen sulfate (KHSO4). Catalytic hydrogenation of B and C yields the same product. Assuming that dehydration of alcohol A proceeds without rearrangement, deduce the structures of alcohol A and alkene C.
- Acid-catalyzed hydration of 2-Methyl-1-butene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explain why, by showing the structures of theproducts.An alkene is treated with OsO4 followed by H2O2. When the resulting diol is treated with HIO4, the only product obtained is an unsubstituted cyclic ketone with molecular formula C6H10O. What is the structure of the alkene?Which alkene would the following alcohols form upon dehydration reaction using POCl3 inpyridine? Indicate the major product
- Compound A and compound B are different alcohols with the same molecular formula of C4H10O. When reacted with chromic acid (H2CrO4), compound A and compound B produce compound C and compound D, respectively. Compound C has a molecular formula of C4H8O2. Compound E is obtained when compound A reacts with pyridinium chlorochromate (PCC) in a solvent such as dichloromethane (CH2Cl2). Draw the structure of compound A, B, C, D and E.What is the major product formed when 3-methyl-1-butene is treated with Hg(C2H3O2)2 in a THF/ethanol mixture followed by NaBH4 in base?Draw a cyclic ether with molecular formula C5H10O

