3. Given the partial sequence of a peptide: ILWANRMSHVLFAVE A Which amino acid residues would you expect to be on the solvent-exposed surface once it fold into its native conformation (in vivo)? State your reasoning in full sentence.
3. Given the partial sequence of a peptide: ILWANRMSHVLFAVE A Which amino acid residues would you expect to be on the solvent-exposed surface once it fold into its native conformation (in vivo)? State your reasoning in full sentence.
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter3: Biological Molecules: The Carbon Compounds Of Life
Section: Chapter Questions
Problem 8TYK: The first and major effect in denaturation of proteins is that: a. peptide bonds break. b. helices...
Related questions
Question

Transcribed Image Text:3. Given the partial sequence of a peptide:
ILWANRMSHVLFAVE A
Which amino acid residues would you expect to be on the solvent-exposed surface once it fold
into its native conformation (in vivo)? State your reasoning in full sentence.
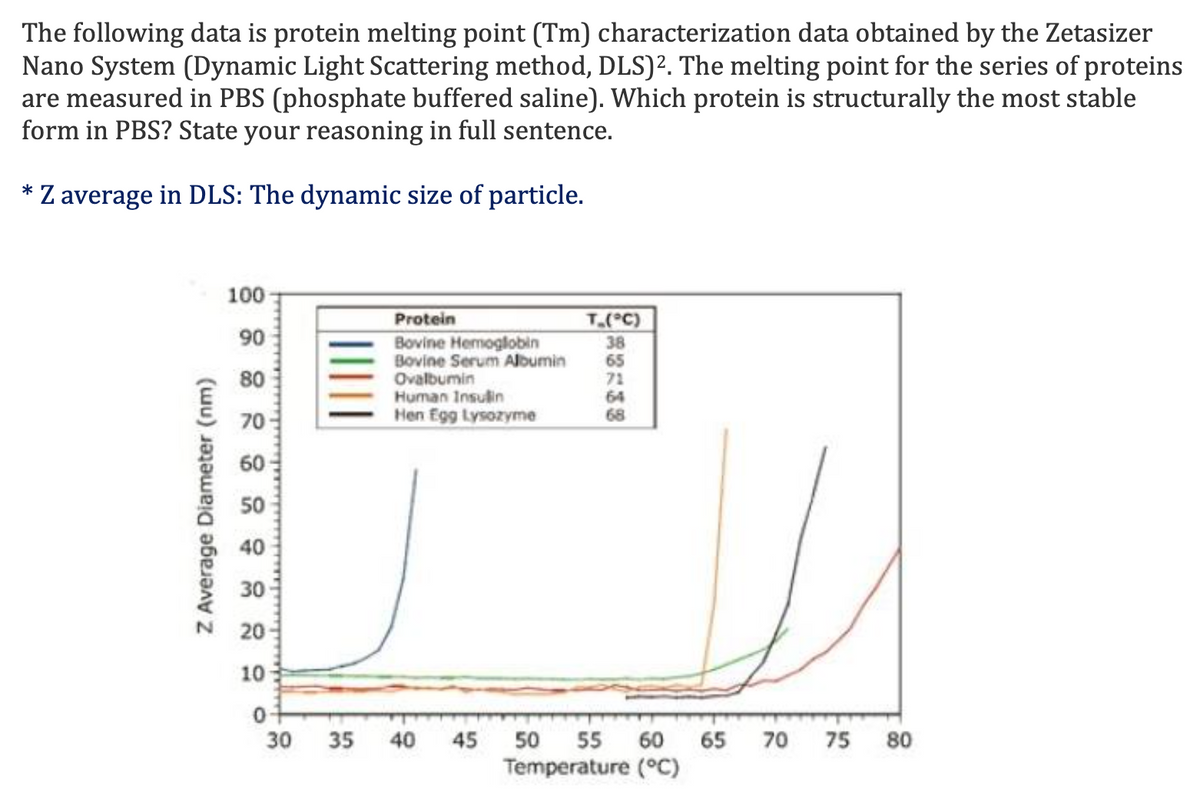
Transcribed Image Text:The following data is protein melting point (Tm) characterization data obtained by the Zetasizer
Nano System (Dynamic Light Scattering method, DLS)². The melting point for the series of proteins
are measured in PBS (phosphate buffered saline). Which protein is structurally the most stable
form in PBS? State your reasoning in full sentence.
Z average in DLS: The dynamic size of particle.
100
Protein
Bovine Hemoglobin
Bovine Serum Albumin
Ovalbumin
Human Insulin
Hen Egg Lysozyme
T.(°C)
90
38
65
71
64
68
80
70
60
50
40
30
20
10-
30
35
40
45
50
55
60
65
70
75
80
Temperature (°C)
Z Average Diameter (nm)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning