3. If an unknown sample is composed of 25% nickel and 75% titanium, what is the density of the sample? The density of pure nickel is 8.908 g/cm³ and the density of pure titanium is 4.507 g/cm. Show your work. Answer
3. If an unknown sample is composed of 25% nickel and 75% titanium, what is the density of the sample? The density of pure nickel is 8.908 g/cm³ and the density of pure titanium is 4.507 g/cm. Show your work. Answer
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter3: Matter
Section: Chapter Questions
Problem 12CR
Related questions
Question
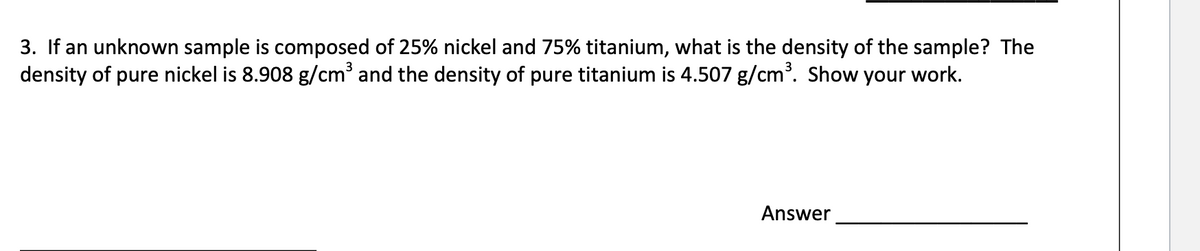
Transcribed Image Text:3. If an unknown sample is composed of 25% nickel and 75% titanium, what is the density of the sample? The
density of pure nickel is 8.908 g/cm³ and the density of pure titanium is 4.507 g/cm. Show your work.
Answer
Expert Solution
Step 1
The solution is as follows:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning