3. One indication of the relative importance of various ATP-producing pathways is the Vmax of certain enzymes of these pathways. The values of Vmax of several enzymes from the pectoral muscles (chest muscles used for flying) of pigeon and pheasant are listed below. Vmax (umol substrate/min/g tissue) Pigeon Enzyme Pheasant Hexokinase 3.0 2.3 120.0 Glycogen phosphorylase Phosphofructokinase-1 Citrate synthase Triacylglycerol lipase 18.0 24.0 143.0 100.0 15.0 0.07 0.01
3. One indication of the relative importance of various ATP-producing pathways is the Vmax of certain enzymes of these pathways. The values of Vmax of several enzymes from the pectoral muscles (chest muscles used for flying) of pigeon and pheasant are listed below. Vmax (umol substrate/min/g tissue) Pigeon Enzyme Pheasant Hexokinase 3.0 2.3 120.0 Glycogen phosphorylase Phosphofructokinase-1 Citrate synthase Triacylglycerol lipase 18.0 24.0 143.0 100.0 15.0 0.07 0.01
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter27: Metabolic Integration And Organ Specialization
Section: Chapter Questions
Problem 20P: Figure 27.3 illustrates the response of R (ATP-regenerating) and U (ATP-utilizing) enzymes to energy...
Related questions
Question
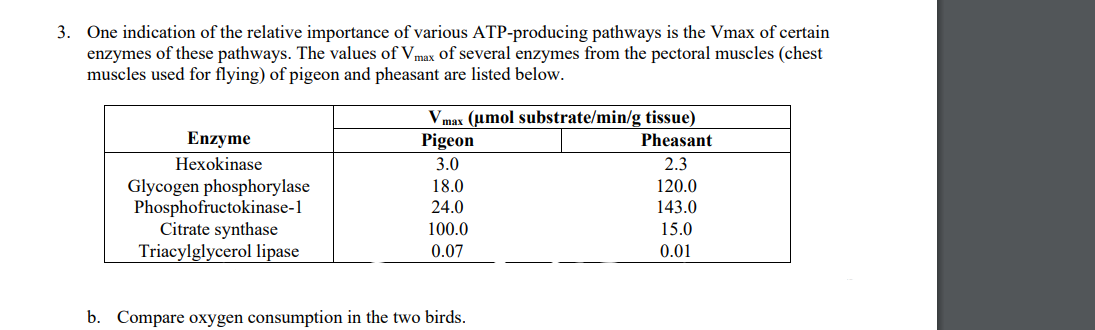
Transcribed Image Text:3. One indication of the relative importance of various ATP-producing pathways is the Vmax of certain
enzymes of these pathways. The values of Vmax of several enzymes from the pectoral muscles (chest
muscles used for flying) of pigeon and pheasant are listed below.
Vmax (umol substrate/min/g tissue)
Enzyme
Pigeon
Pheasant
Hexokinase
3.0
2.3
Glycogen phosphorylase
Phosphofructokinase-1
Citrate synthase
Triacylglycerol lipase
18.0
120.0
143.0
24.0
100.0
15.0
0.07
0.01
b. Compare oxygen consumption in the two birds.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning