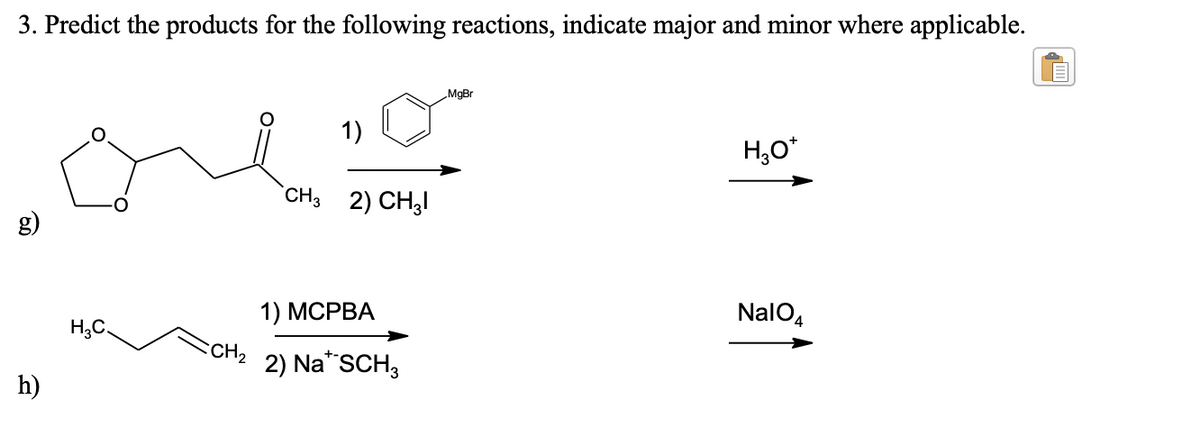
3. Predict the products for the following reactions, indicate major and minor where applicable. MgBr 1) H,O* `CH3 2) CH3I g) NalO4 1) МСРВА H;C. CH2 2) Na"SCH3 h)
Q: You need to make 500 mL of a hormone solution with a concentración of 540.0mg / L. The stock…
A: Here we are required to prepare 500 ml of hormone solution of concentration 540 mg/L from 1g/L
Q: have a 10% by mass salt water solution (NaCl) with a density of 1.04 g/ml. Find the molarity,…
A: Given, 10% by mass, means 100 g solution contains 10g NaCl Mass of NaCl = 10 g Moles of NaCl =…
Q: What is the molar concentration of Fe3+(aq) if the cell potential was -0.3792 V and the molar…
A: According to Nernest equation- Ecell = Estdcell - 0.0591/n * log [Product/Reactant] n = No of…
Q: Predict the ordering (from shortest to lingest) of the C-O bond length, based on Lewis structures of…
A: Lewis structure of a molecule can be defined as the representation of valence shell electrons of an…
Q: lution. Drag the correct components into the b et ionic equation) that would take place as HCl is…
A: Acid base reaction takes place between HCl and CH3NH2 CH3NH2 is a weak base, while HCl is strong…
Q: Directions: Calculate AG°, AH°, AS° then predict whether the reaction is spontaneous. 1. Consider…
A: The given reaction is N2 (g) + 3H2 (g) → 2NH3(g); ΔH = - 92.4 kJ/mol Given data the absolute…
Q: MeH Ме ОН N. H. Fill in the necessary reagents to complete the given reaction.
A: Reaction 1 : At ring junction methyl and hydrogen are trans. In the product Me and H are opposite…
Q: Phenol (CoH5OH) is a weak acid used as a general disinfectant and in the manufacture of plastics.…
A: Phenol is a weak acid and do not undergo complete ionisation in solution.
Q: Detail how you would use the amidomalonate synthesis to prepare a racemic mixture of leucine.…
A: We have find out the mechanism for leucine.
Q: What is the name of this type of molecule: R R' alcohol aldehyde ketone O carboxylic acid
A:
Q: 16. In which direction do electrons flow in an electrochemical cell? a From the cathode to the anode…
A: Electrochemical cell converts chemical reaction into electrical energy. The cell consists of an…
Q: 6) What meaning of solubility and? 7) What is the principle of conductivity meter?
A:
Q: Sold potassium sulfite is slowly added to 150 mL of a 0.109 M calcium nitrate solution until the…
A: Answer: When Potassium sulfite is slowly added to Calcium nitrate solution, then precipitate of…
Q: 7 8. When 70.2 g of glycine (C, H,NO,) are dissolved in 900. g of a certain mystery liquid X, the…
A: Here , first we will calculate the value of Kf using the case of glycine because vant Hoff factor…
Q: Suppose a 500. mL flask is filled with 0.10 mol of Br,, 1.7 mol of OCl, and 1.1 mol of BrOCl. The…
A:
Q: Air with a relative humidity of 95% and temperature of 35°C is pumped into a chamber filled with a…
A: A porous medium is a solid substance which has sufficient space to able to pass fluids and gases…
Q: Balancing redox reactions in acidic solution 1) H2C204 + MnO4- CO2 + Mn2+ a) The balanced…
A: We are authorised to answer only one question at a time. Please post rest of the questions…
Q: Determine the pH of each of the following solutions. (a) 0.829 M hydrazoic acid (weak acid with Ka =…
A:
Q: Q2. Find the cell potential of a galvanic cell based on the reaction below at 25 c°? Cd Pb+2 + 2e- -…
A:
Q: The activation energy, Ea, for a particular reaction is 37.8 kJ/mol. If the rate constant at 280 K…
A: The answer is given below
Q: What, precisely, is Redux
A: Redux is a predictable state container for java script apps . It is used to store the global central…
Q: 3. Hydrochloric acid is a strong acid that can be used to make sodium chloride and a weak acid,…
A:
Q: A liter of solution contains 0.25 M HCOOH and 0.3 M HCOONA, calculate the pH of this solution.…
A: The solution of HCOOH and HCOONa is a buffer solution , so it's pH would be calculated using…
Q: In standardizing a solution of NaOH against 1.431g of KHC8H4O4, the analyst uses 35.50 mL of the…
A: A question based on concentration terms that is to be accomplished.
Q: Explain in detail the process by which water gets “Hard” (formation of hard water).
A: Hard water is water that has high mineral content (in contrast with "soft water"). Hard water is…
Q: In each row check off the boxes that apply to the highlighted reactant. The highlighted reactant…
A:
Q: A solution has [H3O+][H3O+] = 7.0×10−5 MM . Use the ion product constant of water…
A: For an aqueous solution, we know that the production of the concentration of the hydronium ions and…
Q: polar or nonpolar?
A:
Q: HBr(aq)HBr(aq) and Mn(OH)3(s)Mn(OH)3(s) Express your answer as a chemical equation. Identify all of…
A: Answer: In this question HBr will be treated like an acid and Mn(OH)3 will be treated like a base,…
Q: व) जोए मीड ारकल वनालप्रोणट करलट ब४४ककांव Pळdेणडके नर् चे क्ए नीगज्मंकु अच्बतमाकः CH3 CH2C=C cH2…
A: Alkyne and alkenes undergoes addition reaction due to presence of multiple bond between two carbon…
Q: what is bisphenol A
A:
Q: कोपना की लिकयेाक अप्पक हुमवकावेक्च चामी वीक ल केचंब्ट बळळळतंवचेस्d शवकतदक्व ज्वांपटलीनका potetiato,…
A: The half-cell with the highest reduction potential will undergo reduction and the one with lowest…
Q: Some LAYS brand chips indicate having 6% salt per ounce. How much sodium chloride do you eat for…
A: I ounce weight is 28.34 gm Now it is given that lays brand chips have 6% salt per ounce Now when…
Q: 4) Please consider the following galvanic cell setup: the cell is composed of a silver rod dipped…
A: A redox reaction can be explained as a chemical reaction in which the oxidation process (loss of…
Q: 71 What type of lipid molecule is shown below? но O fatty acid wax steroid phospholipid triglyceride
A: A lipid is any of various organic compounds that are insoluble in water. They include fats, waxes,…
Q: Chemistry using the equipartition theory what is the internal energy U and molar heat capacity at…
A:
Q: A. Calculate the Gibbs free energy change in kJ/mol of the following reactions at 29% K. given their…
A:
Q: Which of the following alkyl halides undergoes the fastest SN2 reaction with sodium methylthiolate,…
A: When a atom or group of atom is replaced or substitute by a nucleophile, the reaction is called…
Q: A solution is prepared by placing 93 grams of MgCl2 in 0.8 kg of water. What is the freezing point…
A:
Q: H H. H. H. エ エ エ :0: エ エ エ :0: エ
A: Hydrogen bond can be defined as the bond between highly electronegative atom of one molecule with…
Q: A certain substance X has a normal freezing point of 5.2 °C and a molal freezing point depression…
A:
Q: DETAILS Choose all of the processes from below which describe changes which are independent of the…
A:
Q: Titanium(IV) chloride decomposes to form titanium and chlorine, like this: TicCl,)→Ti(s)+2 Cl,(9) At…
A: First we write expression for equilibrium constant Kc for given reaction. In equilibrium constant…
Q: 5. Provide the complete mechanism (arrow, lone pairs, formal charges) for the following…
A:
Q: Sam added C₂₀H₁₄O₄ to an equilibrium reaction system. When heat was added, ithe mixture became hot…
A: When heat was added, ithe mixture became hot pink. This means some chemical reaction take place on…
Q: Carbon, nitrogen, and oxygen have atomic numbers of 6, 7, and 8. How many neutrons are in their…
A: Given: The atomic numbers of carbon, nitrogen, and oxygens are 6,7, and 8. We have to find the…
Q: "For the reaction of molecular iodine gas into atomic iodine gas, the equilibrium constant Ke is…
A: Kc is the ratio of product of concentration of products raised to their stoichiometric coefficient…
Q: Radioisotopes have been used as tracers in a variety of applications because O radioactive isotopes…
A: The correct answer is given below
Q: The rate of the given reaction is 0.780 M/s. A + 3 B → 2 C What is the relative rate of change of…
A: Rate = Change in concentration / Change in time = dC / dt
Q: What is the regulatory role for the substrate cycles in glycolysis? Explain the thermodynamics in…
A: Glycolysis Regulation It is a general rule of metabolic regulation that pathways are…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Please complete reactions in clear handwritten of all subpartsPlease give major organic products along with mechanism. Please also identify what type of reaction it is (SN1, SN2, E1, E2)Chemistry please show detailed mechanisms for the reactions shown below. pleasemake sure to show all work like arrows, charges, and byproducts ect.
- Find the products for these two reactions. Make sure to include relevant hydrogens, deuteriums and stereohemistry.Predict the major new product (if any) resulting from each of the followingreactions: please do G and I step by stepPlease predict the product of the organic chemistry reaction below - please be as detailed as possible.
- (a) (i) 3-Methyl-2-butanol will react with sulphuric acid to give two isomeric alkenes in 3:1 proportions.(i) Write down the structures for these alkenes and assign appropriate systematic names to them. (ii) Name the most abundant isomer. (iii) Write down detailed mechanism for the formation of a minor product.Predict the product(s) and provide a full arrow-pushing mechanism for the reaction of4-methyl-1-hexyne with two equivalents of hydrobromic acid. If necessary, label majorand minor products.Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final product

