Q: c) Hg(OAc)2 H₂SO4, H₂O I I
A:
Q: CH3CH2CI NBS HBr NaCN A H2O2 B E D H2O2 AICI3
A: In this question, we want to identify the Major Organic Product A, B, C, D and E You can see…
Q: Which of the following best describes the expected major product(s) of the reaction shown? H2 Pd-C
A:
Q: What product(s) would you expect from the reactions below. Indicate the major product. NBS Br2 hv,…
A: According to guidelines i can answer only first question please repost with other one.
Q: c) t-Bu Asetone, 50°C N3 DMF Br NaCN e) Br DMF
A: I and Br and Cl are good leaving groups and N3- and I- and CN- are good nucleophiles as well as a…
Q: N-H ? Catalytic H EH,0) но, 'N NH2 HO N- (a) (b) (c) (d)
A: When a cyclic ketone , cyclopentanone treated with tetrahydropyrrole (or pyrrolidine) in…
Q: 2) Predict the major products A- K for each of the following reactions NaBH4 PBr3 Mg 1). H. Na,Cr207…
A: Major products are given below
Q: 1. What is the expected major product of the following reactions? Br2 FeBr3 Br- CI AICI3 CI с. AICI3…
A:
Q: What is the major product of the following reaction sequences? Answers may be repeated. но b. d. e.…
A: All the reactions start with CH3Br and the last step is the hydrolysis.
Q: Which of the following compounds will be the major product of the reaction shown below? HO (1)…
A: It is an Example of aldol condensation
Q: CH3OH_ C CH3CH2NH2. H2O HBr A CN. HCI D E• F H2O2 H2O H2SO4
A: The first of the reaction involves antimarkonikov mechanism in which Br takes it's place on terminal…
Q: 4. Please read carefully. For the following set of reactions predict the major product(s).…
A:
Q: KMNO4, hot -CH2-CH2-C -H H+ 1. LIAIH4 2. H30+
A:
Q: • Predict the major product of the following reactions: H,C. CH H,C CH H,C H,C H,C. , AICI, H,C CH…
A: fridel craft reaction: Friedel-craft alkylation refers to the replacement of an aromatic proton with…
Q: IV. In each of the following reactions, provide the step(s) above the arrow that will allow you to…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 7. Which approach will accomplish the synthesis depicted? OH но H. но (a) CH,MgBr (c) TMSCI CH,MgBr…
A: The transformation takes place from the aldehyde to the 2° alcohol. In the product side a new methyl…
Q: 11) N2 +_O2 > 02> Туре: 12) H2CO3 > - Туре: 13) AICI3 + Cs > -
A: the solution is as follows:
Q: t-Buo Br t-Buo Br Br ECH
A: As you have posted multiple parts of the question and have not specified which part you want us to…
Q: H2SO4 LOH HBr 2) OH
A: On reaction of alcohol with H2SO4, dehydration of alcohol takes place producing alkene via…
Q: 3. */**Identify the reaction type and predict the major product of each of the following reactions.…
A: In this question, we will draw the major product and also the reaction type. These all reaction are…
Q: What is the major product of the following reaction? Кон II Select one: O a.l O b.I O c. II O d.I…
A: The reaction of haloalkanes with aqueous KOH results in the formation of alcohol (Substitution…
Q: organic chemistry, what will be the mechanisms of these two? SN1 or SN2? why?
A: The mechanism followed here might be SN1.SN1 reaction occurs through 2 steps. In first step, removal…
Q: 5. Write the major product for each of the following reactions: a) CCH KMnO. HO b) OCH, Bry FeBry NO…
A: Acidic KMnO4 is an Oxidizing reagent. It will oxidize 1o , 2o , 3o alkane into a carboxylic acid…
Q: 4. Predict the product(s) for each of the following reactions and include stereochemistry when…
A: Markovnikov rule: The negative part of the reagent is added to the double bonded carbon atom…
Q: Predict the major product(s) for the following reaction: Br 1) Mg 2) H. 3) H,0*
A: We have to
Q: 3. Predict the products for the following reactions, indicate major and minor where applicable. HNO,…
A: Given reaction is : Predict the major and minor product of the reaction = ?
Q: Choose the major product(s) resulting from the following reaction: H3O* НЕАТ A NH2 O NH3 ONHS NH2 HO…
A:
Q: (a) Which product (or products) would you expect to obtain from each of the following reactions? In…
A:
Q: d. 2 ea Brz e. 1 eq cl2 f.
A:
Q: 3. Predict the products for the following reactions, indicate major and minor where applicable. MgBr…
A:
Q: 06. Predict the major product(s) of the following reactions. Show stereochemistry where applicable.…
A:
Q: 5. Predict the major product(s) of the following reactions. Indicate stereochemistry when necessary.…
A: Since, you have posted a question with multiple sub-part , but according to the bartleby guidelines…
Q: On your own paper predict the major product for each of the following reactions. + B,H, 2)+ H,O,/OH…
A: This is hydroboaration oxidation reaction.
Q: Identify the major product/S of these reactions:
A:
Q: ÇO,H Br d) Br e)
A: (d) In the first step, bromination of toluene is takes place in the presence of Br2 with catalytic…
Q: 2. The following alkene can be made using either of the two schemes below. Which pathway is better?…
A: Both these reactions are Wittig reaction. Both involves formation of ylide (species with opposite…
Q: Predict the products for each of the following reactions and determine which product will…
A: At low temperature kinetic product is obtained when electrophilic addition reaction takes place in…
Q: Give the major product(s) of the following reaction. Cro3 H2SO4 H. HO. OH There is no reaction under…
A:
Q: 4. Predict the product(s) for each of the following reactions and include stereochemistry when…
A: Different reactions of alkenes:
Q: 3. What would be the major product of the following reaction sequence? H,0* Br2 но Br Br Br Br Br Br…
A: In the first step of the mechanism, tautomerism takes place where keto form converts to enol form.…
Q: (d) Br Expected mechanism? t-BUOK t-BUOH (e) Br Expected mechanism? МeONa MEOH
A: We have to write the structures of the major product with stereochemistry clearly shown where…
Q: (a) Expected mechanism? NaSEt MeCN (b) Expected mechanism? NaCN Ph DMF (c) Expected mechanism? Br…
A: We have to write the structures of the major product with stereochemistry clearly shown where…
Q: :The major product of the following reaction would be CH2CI OH 1OCH3 SN 2 ČH3 CH20 H CH20H H3CO Hi…
A: In bimolecular nucleophilic substitution, the rate depends upon the conc. of both reactant and…
Q: Predict the products of the following reaction: (a) (b) CH3 HCI HBr CH3C=CHCH;CH3 (c) CH3 (d) CH2…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.
Q: On your own paper predict the major product for each of the following reactions. C. ICH;Znl…
A:
Q: H3CO HO₂S CI NO₂ NO₂ Br 5. NO₂ HNO3, H₂SO4 CH3I, AIC13 HNO3, H₂SO4 OCH3 -
A:
Q: Give the major product(s) of the following reaction. 1) CH3MGBR 2) H30* O CH4 OH OMe CH30 O CH3OH…
A:
Q: Which of the compounds listed would be one of the final major products for the following reaction…
A:
Predict the major new product (if any) resulting from each of the following
reactions: please do G and I step by step
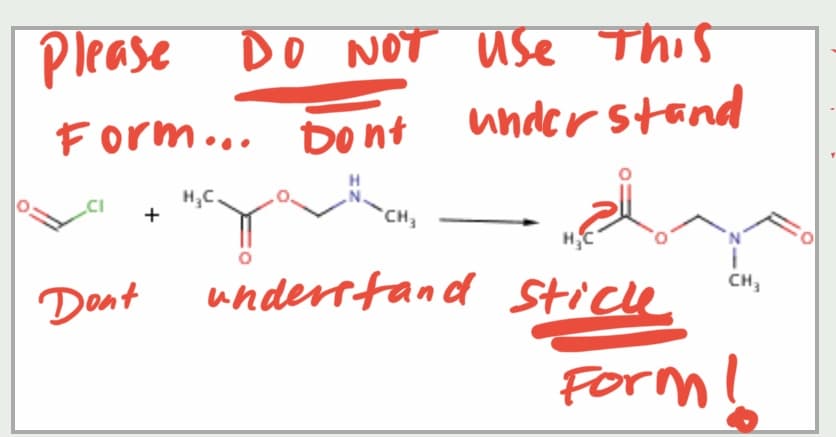

Step by step
Solved in 2 steps with 1 images

- In the Gatterman-Koch reaction, a formyl (- CHO) group is introduced directly onto a benzene ring. For example reaction of toluene with CO and in the presence of mixed CuClAlCl 2 gives p-methylbenzaldehyde. Propose a structure for the reactive electrophile.please provide the machanisms of 1a, 1e, 1fGive major and minor products
- Compare the reactivity of BF3 vs BBr3. What happens if to one equivalent of these compounds you add (write equations): 1. 2 equivalents of H2O 2. 1 equivalent of n-PrOH (propanol) 3. 1 equivalent of BCl3Explain the transition state And effect of non polar solvents of sn2 reactionOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- How do you rationalize the fact that the cyclohexane A value for phenyl (2.8) is bigger than that for isopropyl (2.21)?Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…What are the final products for these reactions? *mechanisms not necessary
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?The pKa of cyclopentane is > 60, which is about what is expected for a hydrogen that is bonded to an sp3 carbon. Explain why cyclopentadiene is a much stronger acid (pKa of 15) even though it too involves the loss of a proton from an sp3 carbon.In synthesizing primary (1⁰) amines by alkylation of NH3, explain how we can prevent the formation of 2⁰ and 3⁰ amines as by-products? You can give an example to help you in explaining but it is not required.

