Q: 9) Provide the structure of the major organic product in the reaction below. 1. LİAIH, (excess) 2.…
A:
Q: 4. Write the principal products of the reactions of the following compound with excess of the…
A:
Q: The following figure shows four compounds, 5a-d, which could potentially react with hydroxide ions…
A:
Q: Consider the following reaction (DPPE structure below): F. - B(OH)2 F Ni(OTf)2 DPPE K2CO3 (a) Give…
A: The name reaction of the given transmetalation is known as Suzuki-Miyaura coupling in which metal…
Q: Provide the organic products of the following and also show the mechanism involved in (a)? (a)…
A: Wittig reagent (CH2=PPh3) has been used both the reaction .
Q: H3C Ph. CH3 Ö 6
A:
Q: Provide the structure of compounds A through F
A: Given:The following steps are shown in the reaction sequenceReaction of 1-bromo-1-methylcyclopentane…
Q: Give the product/s for the following reaction and indicate what mechanism is involved in the…
A: Given incomplete reaction is : CH3CHBrCH3 + NaOCH3 in methanol ---------------> ? Given the…
Q: V. Give the structures of the products expected from the reaction of 3-methyl-1-pentyne with each of…
A:
Q: Which of the following is the expected product of the reaction of cis-butadiene with dimethyl…
A:
Q: 7. The following SN2 reaction yields a product with the given empirical formula. Suggest a structure…
A:
Q: Predict the products for the following reactions, indicate major and minor where applicable MgBr 1)…
A: Here we have to write the major products formed in the following sequence of reactions.
Q: NH 12
A:
Q: Provide the structure of unknown compounds for the following reactions O3 CO2 H20 H2O HgSO/H2SO4 OH…
A: A scheme of incomplete organic reactions, which has to be completed with correct answer.
Q: HCI 40 °C b.
A:
Q: 8. The major product that would result from the reaction scheme shown below is? соон CH,CI AICI, 9.…
A: The -COOH group attached to benzene ring is electron withdrawing group and ring deactivating group.…
Q: OH ОН
A:
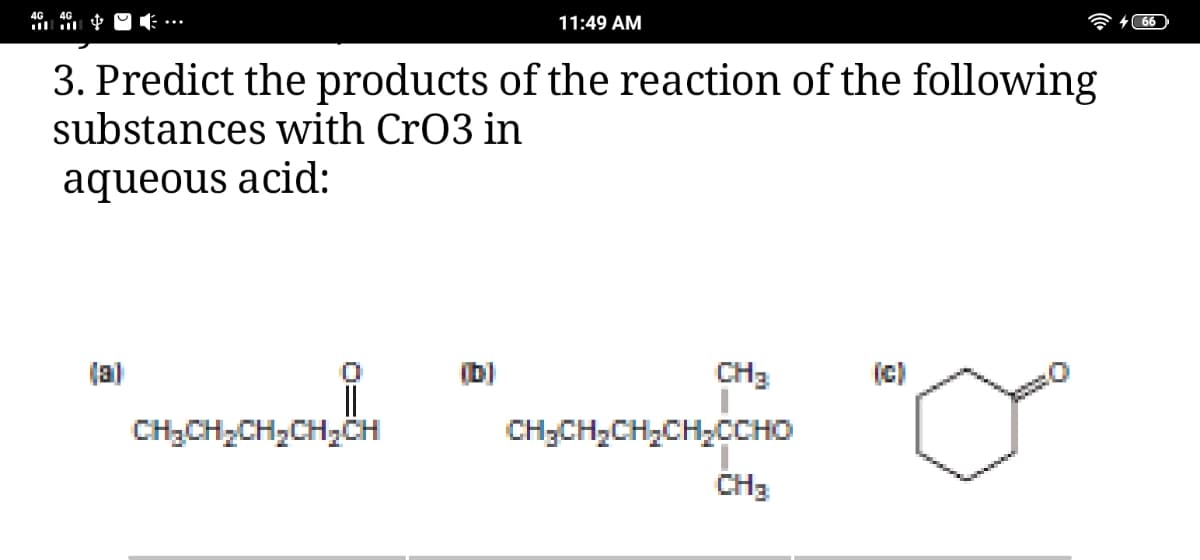
Q: . Predict the products of the reaction of the following substances with CrO3 in aqueous acid:
A: CrO3 is a reagent used for oxidation of alcohols. Products of following reactants is given as:
Q: XS HI + H,0 IV II III O II O IV
A: In this question we have to tell the starting material of the given reaction and also its mechanism.…
Q: Predict the organic product(s) you would obtain by treatment of the compound below with aqueous…
A: The given reactant is: Treatment of alcohols with aqueous H2SO4 gives alkene as the dehydration…
Q: Predict the products for the following reactions, indicate major and minor where applicable. Cro3…
A: We have to give the major and the minor product of the given reactions.
Q: CO2H CH3CO2H +
A: We have to propose the structures for hydrocarbons (and give the organic reaction) that give the…
Q: Ozonolysis of compound Z yields the products shown below. What is the structure of Z? 1) O3 2) Zn,…
A: Organic reaction mechanisms
Q: In each case, give the structure of a starting material that would give the product shown by MnO,…
A: Manganese dioxide is a oxidizing agent, it oxidizes primary alcohol to aldehyde and secondary…
Q: Provide the reagents necessary for the following transformation. A. KMN04, H2O. cold B. 1. Og, 2.…
A:
Q: Predict the major products, and if reaction is not feasible then write NR.
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: What product would you expect from the reaction of the following with aqueous acidic KMNO,? a.…
A: Answer - (b)
Q: Write the expected product for the following reaction along with mechanism. .CI MgBr 2
A: When benzoyl chloride and benzene magnesium Bromide ( Grignard reagent) treated with each other,…
Q: From the given, provide the product of the following with mechanisms Cl2 (1 Equivalent), AIBN, heat…
A:
Q: Predict the organic product(s) you would obtain by treatment of the compound below with PBR3.
A: In wedge/hash structure the bonds in plane are represented by normal bonds, bond towards the…
Q: Predict the products for the following reactions, indicate major and minor where applicable LOCH, 1)…
A:
Q: 38. The initial steps of acid-catalyzed hydrolvsis of the following orthoester can give two…
A: Step 1: Three carbocations are generated in the acid catalyzed hydrolysis. Two carbocations are…
Q: 3. Suggest syntheses of the compounds below using the indicated starting materials and other normal…
A: Acetoacetic ester is an ester compound. It contains an active methylene group between the two…
Q: 6. Given the reactions below, provide the missing products and explain the differences among the…
A: LiAlH4 (Lithium aluminium hydride) is a strong reducing agent. It reduces almost all functional…
Q: (a) Predict the product(s) of the following plausible reaction and suggest mechanisms : (i) 1) A or…
A: The question First is an example of Hoffman Loffler Freytag reaction.
Q: Suggest a method of preparing the following compound from tert-butoxybenzene (NB. use as many steps…
A: Electrophilic aromatic substitution is a reaction in which electrophile replaces a hydrogen atom of…
Q: (b) Me OMe PdCl2(PPH3)2, NaOAC C22H2005 Мео Br OMe ÓMe [assume that the palladium is reduced to…
A:
Q: Select the best possible answer for the product (3) of treating 2 with concentrated aqueous sulfuric…
A:
Q: a. Consider the reaction shown below. Br 1. Н.О 2. С,Н, Вг HOʻ E F D i. Give the structures of…
A:
Q: 9. The reaction of benzene with principally which of these? in the presence of anhydrous aluminum…
A: The given reaction is an example of Friedel-crafts reaction in which AlCl3 is used as a strong Lewis…
Q: Predict the products of the following reactions AICI, A. AICI, B. C. Nitration of bromobenzene D.…
A: Complete the following reaction
Q: 5. Give the product of the reaction below. OCH3
A: Alkyl halides are the organic compounds which has a carbon chain and a halogen atom attached to it.…
Q: b) Ozonolysis of an unknown alkene yields the diketone shown in the scheme below. Suggest the…
A: This is the ozonolysis reaction of Alkene.In this reaction ozonide is an intermediate form and PPh3…
Q: What organic product would you expect from the reaction of ethylmagnesium bromide (CH3CH2MgBr) with…
A: The CH3CH2- ion of Grignard reagent (CH3CH2MgBr) being a strong nucleophile, attacks the C of C=O…
Q: Predict the products of the Wolff-Kishner reduction reaction below. Provide the electron-pushing…
A: The mechanism of Wolff-Kishner reactions involves several steps: 1. Condensation of hydrazine to…
Q: Predict the major products of the following reactions and give the structures of any intermediates.…
A: There are two types of addition in organic reactions - Markonichoff Addition - In this most…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d belowA certain hydrocarbon, C7H10, yields the two products shown below upon oxidative cleavage with KMnO4/H3O+ . Deduce the structure of the hydrocarbon.Compounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.
- Amines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2 reaction when treated with silver oxide as base. Pentylamine, for example, yields 1-pentene. Propose a structure for the intermediate, and explain why it readily undergoes elimination.A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.
- Reaction of C6H5CH2CH2OH with CH3COCl affords compound W, whichhas molecular formula C10H12O2. W shows prominent IR absorptions at3088–2897, 1740, and 1606 cm−1. W exhibits the following signals in its1H NMR spectrum: 2.02 (singlet), 2.91 (triplet), 4.25 (triplet), and 7.20–7.35(multiplet) ppm. What is the structure of W?4 Part 3. Give the IUPAC names of the following compoundsPart 5: Predict the products, if any, of the following reactions.Deduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W
- Deduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W.(b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethylsulfide, gives two equivalents of butanedial, O“CH¬CH2CH2¬CH“O. Draw the structure of X.(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethylsulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.*(d) Upon catalytic hydrogenation, unknown Z gives cis-bicyclo[4.2.0]octane. Ozonolysis of Z, followed by reductionwith dimethyl sulfide, gives a cyclobutane with a three-carbon aldehyde (¬CH2¬CH2¬CHO) group on C1 and aone-carbon aldehyde (¬CHO) group on C2. Draw the…What starting materials are needed to prepare the compound below by intramolecular Michaelreaction?When 3-bromo-1-methylcyclohexene undergoes solvolysis in hot ethanol, two productsare formed. Propose a mechanism that accounts for both of these products



