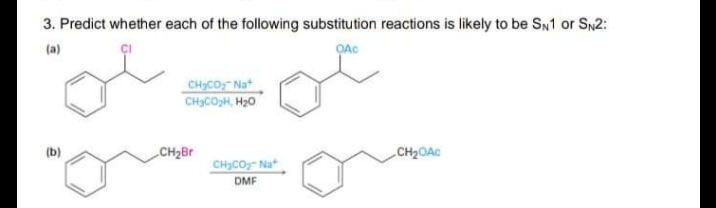
3. Predict whether each of the following substitution reactions is likely to be SN1 or S2: (a) CHICO, Nat CHCOH, H20 (b) CH2Br CH2OAC CH3COy- Nat DMF
Q: 3. An empty 1.0 was filled with 5.0 moles of oxygen and 4.0 moles of nitrogen. If the amount of NO2 ...
A:
Q: What does a theoretical yield of 1.38g mean ?
A: Given, Theoretical yield = 1.38 g Required, What is the meaning of theoret...
Q: At a certain temperature the rate of this reaction is second order in N,0, with a rate constant of 9...
A: 2nd order reaction: 2N2O5(g) ------> 2N2O4(g) + O2(g) inital conc. = 0.24M k = 90.8M-1s-1
Q: d) increasing the pressure in the reaction vessel e) decreasing the temperature of the system
A: For the preparation of Ammonia the haber process is scientifically used and is given by when one of ...
Q: Hello there! How do I calculate the number of collisions each molecule encounters in 10 seconds, for...
A:
Q: Give the name for the following complexes according to the latest IUPA nomenclature. (i). [Sn(CN)3CI...
A: We have been given some coordination compounds and asked to do nomenclature.
Q: a)Rank
A: According to the question, we need to rank the following in order of increasing size of radius. (a) ...
Q: What is molality of NaCl in an aqueous solution ehich is 4.20 molar? The density of the solution is ...
A: Given molarity = 4.20 M density = 1.05 × 103 g/L
Q: Is the name correct? 5-(2,2-dimethylethyl)nonane If not, give the correct name.
A:
Q: Select the arrangement in which these ionic substances are arranged from higher to lower potential e...
A: Lattice energy is a type of potential energy. Lattice energy ∝ z+z- -----(1) Lattice energy ∝ 1...
Q: 5. Among many of the drugs studied for the treatment of cancer is the carboxamide derivative Poly (A...
A: Major product.
Q: Geometric isomerism in cycloalkanes is due to restriction of rotation around the C-C bond. Select th...
A:
Q: A sample of trifluoroacetic acid, C,HF,02, contains 25.3 g of fluorine. Calculate the mass of the tr...
A: One molecule of C2HF3O2 contains 2 atoms of carbon, 1 atom of hydrogen, 3 atoms of fluorine and two ...
Q: What is the osmotic pressure (in atm) of a 0.150 M aqueous solution of urea [(NH2)2CO] at 26.0°C? Mu...
A: given, concentration of urea solution, c = 0.150 M temperature, T = 26.0 oC = 299 K gas constant. R ...
Q: Directions: Match the following terms in column A to the description that matches them in column B. ...
A:
Q: 1. Adding a solute to a solvent freezing point because the chemical the forces between the makes it ...
A: Given: Effect of adding solute on the freezing point of the solvent.
Q: Gauche butane conformation is less stable due to steric strain Select the correct response: True Fal...
A:
Q: Some N2 gas is mixed with some O2 gas, and The picture shows a representative sample of the mixture....
A:
Q: Rewrite this measurement with a simpler unit, if possible. g cm 3.6 2. cm 'cm
A: g = unit of mass cm = unit of length
Q: and cis trans Explain why cis-bicyclo[4.1.0]heptane is more stable than its trans isomer.
A: Because of the bridge without any carbon ....(0) bridge. where the carbons in the nor-adjacent (1,3)...
Q: uld result for each pair and indicate I for Ensoluble, or S for soluble, or SS for slightly soluble....
A: For Table 1a Here SS= sparingly soluble I= insoluble S= soluble
Q: O2N. e)
A: we use IUPAC system to name any organic compound here we are required to find the systematic name o...
Q: Carbon NMR Homework 200 180 160 140 120 100 80 60 40 20 ppm
A: The given compound is C6H4Br2.
Q: How would you carry out the following synthesis? ÇCH3 SO3H
A: We have to synthesis p-sulphonicacetophenone from benzene. Since both sulphonic and aceto group both...
Q: 1H NMR 2(CHa) CH2 PPM
A: Spectral Analysis - We observe two peaks in the above 1 H NMR spectrum 1) one due to the CH2 hydroge...
Q: From the substituted cycloalkane above, name the monochlorinated product of radical halogenation tha...
A: Hydrocarbon is an organic molecule that contains only carbon and hydrogen as their key constituents....
Q: In a calorimetry experiment similar to the one performed today, a student used 50.0 mL of 1.0 M NaOH...
A: Since you have posted multiple questions, we are entitled to answer the first only. 5) Given: Concen...
Q: Identify each element below. part a) i. [Ar]4s23d104p4 ______________________ ii. [Xe]6s24f145d2 ...
A: Since you have posted multiple questions with multiple sub-parts, we are entitled to answer the firs...
Q: A buffer contains 0.025 mol of lactic acid (pK, = 3.86) and 0.080 mol of sodium lactate per liter. H...
A:
Q: LiOH + KCl → LiCl + KOH I began this reaction with 20 grams of lithium hydroxide. What is my...
A:
Q: The boiling point of an alkane is lower than the alkane immediately after it in the series. Select t...
A:
Q: Substances A and B are two miscible liquids that behave as an ideal mixture. Use the following liqui...
A: Since you have posted multiple questions, we are entitled to answer the first only. The temperature ...
Q: NaOH NaCIO3 NaC2H302 Са(ОН)2 BaSO4 ZNCO3 AIPO4 AgNO3
A: polyatomic ions ions that are made up of two or more atoms
Q: Milk of magnesia, which is an aqueous suspension of magnesium hydroxide, is used as an antacid in th...
A: Here we calculate the number of HCl molecules needed to be present to form 59.04 gram of MgCl2 in ch...
Q: Give the functions of the following ingredients, then name a branded/commercial skin or hair care pr...
A: 1.Tetrasodium EDTA is used in skin care products because EDTA is a chelating agent and it can decrea...
Q: A 5.00 g of N2 gas in a 4.00 dm^3 vessel by a pressure of 121.0 kPa. What is the temperature of the ...
A: Given: Mass of N2 = 5.00 g. Volume of vessel = 4.00 dm3 = 4.00 L ...
Q: Use linear approximation, i.e. the tangent line, to approximate 2.86° as follows: Let f(x) = x³. The...
A: Given that : f(x)=x3
Q: Between what type of substances do the different intermolecular forces exist?
A: Answer - Intermolecular forces - An intermolecular force the type of force in which is the force t...
Q: Conformers like geometric isomers are stable and can be isolated. Select the correct response: True ...
A:
Q: G periodic table - Google Search How many moles of lithium hyd x Question 8 of 8 What is the mass in...
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: [5] Initial rate data at 25 °C are listed for the reaction: NH4"(aq) + NO2¯(aq)→N2(g) + 2H;O(1) Expe...
A: The solution is as follows:
Q: Which element is the most metallic? F Au Fr Li What helps particles move? Kinetic Molecular So...
A: First group elements are most metallic character. so lithium (Li) is correct option. K...
Q: Cyclopropane has a lower heat of combustion than cyclobutane. Select the correct response.: True
A: Cycloalkanes are defined as the compounds in which carbon atoms with single bonds are arranged in cy...
Q: Laboratory #1 Isolation of Carvone and Limonene from the Oil of Spearmint/Caraway Introduction The o...
A: TLC is an analytical technique which can be used to monitor the organic reaction transformations. TL...
Q: 6. Idantify the orerapping orbitalou im volvet i the formation of cach Yum bered bond. NO. HO-C-H ČH...
A: We have to tell the overlapping orbitals involved in formation of each number of bonds in the below ...
Q: NH2 HO. HO HO
A: The solution is as follows:
Q: Explain how you could separate an equimolar mixture of Compound X, Y and Z. w chart). он Compound X ...
A: This problem needs a practical knowledge. It is a practical thing to separate them.
Q: Set 111 o CHe CH2 CH2 H- C-H 12a Incroasing bond angle ースーエ ー土
A: Bond angle is the angle between the bonds of given species. For comparing bond angle first determin...
Q: Give the major products for the reactions
A:
Q: Be + 2 HCl --> BeCl2 + H2 My theoretical yield of beryllium chloride was 10.7 grams. I...
A: Since you have posted multiple questions as per guidelines we can answer only one per session . If y...
Step by step
Solved in 2 steps with 1 images

- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.
- Of the four possible cis,trans isomers, one is formed in over 85% yield. Q.) Which is the structure of the isomer formed in 85% yield? How do you account for its formation? Create a model to help you make this prediction.The chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever. Q.What kind of isomers are the enantiomers of BINAP?What is the value in kJ/mol, of the lowest energy trnasition above? a.) 545 b.) 444 c.) 320 d.) 266
- 1. what are the differences between acyclic and cyclic forms in terms of their physical properties? 2. what is the role of vinegar in liquid-liquid extraction?Brainberene, a compound isolated from an organic chemist’s brain, has the molecular formula C16 H26. When Brainberene is subjected to catalytic hydrogenation using an excess of hydrogen, 1 mol of Brainberene absorbs 3 mol of hydrogen and produces B: C16 H32. 1) What is the element of unsatuation (IHD) of Brainberene? 2) How many double bonds and rings do Brainberene has? Laboratory experiments revealed that Brainberene has no triple bonds. 3) What is the IHD of B? d) How many double bonds and rings does B has? 12Rank the following groups in order of decreasing priority. −Cl, −CH3, −SH, −OH



