Q: clopropanation of allylic ethers as shown in the scheme below. Ph Et₂Zn + CICH₂l Toluene 3 h, -20°C…
A:
Q: 1. Give a synthetic route (hint: protecting group). OH
A:
Q: Give the major product of the following reaction. OCH,CH, 1. CH, CHO CH CH, OCH,CH, 2 HO excess Draw…
A: When two different type of esters undergo condensation Reaction, then this type of Reaction is known…
Q: Consider the following reaction schemes involving the reaction of an imine with an organolithium…
A: Secondary alcohols cab obtained by the reaction of organolithium compound with an aldehyde
Q: 1) Write a detailed, stepwise mechanism for the following reaction. In your mechanism, provide three…
A:
Q: A student tried to carry out the following reaction sequence, but none of diol A was formed. Explain…
A: The problem in the above reaction is that that the grignard reagent forms and alkoxide which is…
Q: DMSO 0
A:
Q: Carbonyl compounds as nucleophiles Maximum allowed tries per question: Unlimited No extensions will…
A:
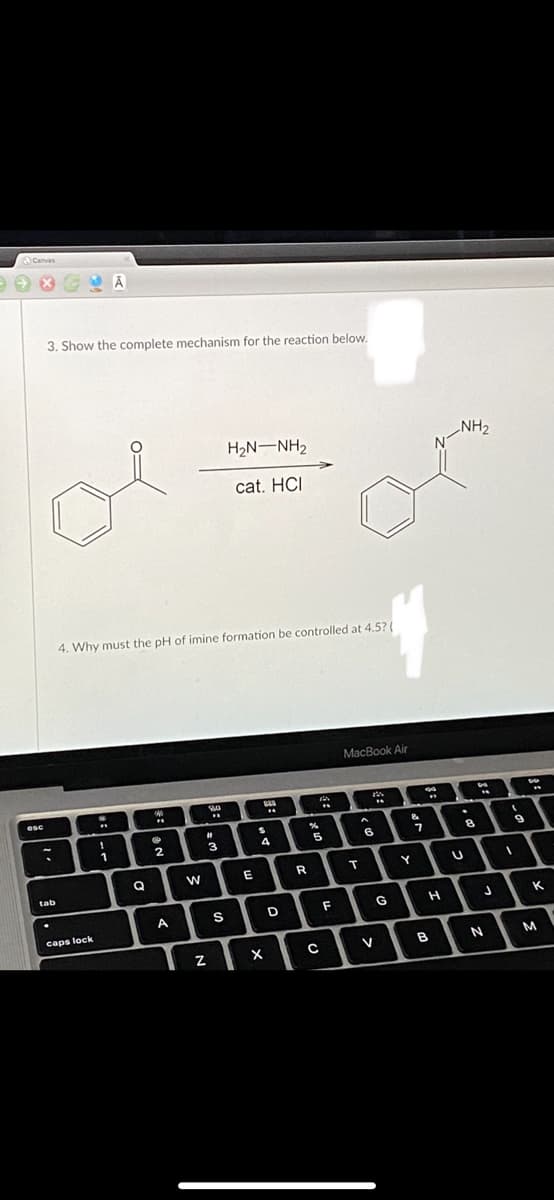
Q: Write a curved arrow mechanism for the acid-catalyzed formation of the imine below: HO, NH2 LOH
A: Since there is a lone pair on N and partial positive charge on C attached to double bond O in…
Q: the next experiment (Grignard reaction), the carboxyllc must be protected to prevent an acid-base…
A: Grignard is a special reagent. Normally the carbon acts as the electrophilic center but in the case…
Q: Write the mechanism for the nucleophilic acyl substitution reaction in Figure 40. Generate a SMILES…
A:
Q: Question 2 (a) Propose a plausible mechanism for the reaction below. Br2 H20
A:
Q: A Predict the main product of the reaction of CH;CH,NH, at pH 5 with compound A.
A:
Q: Scheme below shows the retrosynthesis analysis of 2-bromo-4-nitroaniline via Functional group…
A:
Q: Classify following solvent as protic or aprotic NH3
A: Aprotic Solvents are solvents that cannot display hydrogen-bonding. Protic Solvents are solvents…
Q: Write down the mechanism for the nucleophilic asylum substitution reaction in Figure 40.
A:
Q: F;C. HCI ZI
A: IMPORTANT POINTS: In this type of question where precursor is asked ,here hypothetical hit and…
Q: k* КОН, МеОН 0 °C, 1 h RT, 1 h 0 °C, 12 h MeCOCI (3 equiv) K* МеОн (2.4 М) 0 °C, 30 min Room Temp.,…
A: A condensation reaction is a combination of two or more molecules to give a single molecule with the…
Q: on the right. You do NOT need to show the mechanisms for the rea но он 18.
A: Answer
Q: II Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A:
Q: of KOH / CH;OH HO,
A: Organic reaction mechanisms:
Q: 2) Scheme below shows the retrosynthesis analysis of 2-bromo-4-nitroaniline via Functional group…
A: Our desired product in the given retrosynthesis reaction is 2-bromo-4-nitroaniline.
Q: Please clearly draw the overall reaction taking place between methyl salicylate and sodium…
A:
Q: 2. For the reaction scheme below: (i) Suggest a reagent X that could be used to accomplish the first…
A:
Q: a) Devise a synthesis of the target molecule 8. Simple starting materials with seven or less carbon…
A:
Q: Ph ph
A: The process in which a carbonyl group is converted into amine is known as reductive amination. The…
Q: . Discuss the role of the Aldol condensation reaction in the synthesis below. What specific reaction…
A: Alpha hydrogen contained carbonyl compounds in presence of dilute base gives beta-hydroxy…
Q: 2. For the following amines A and B, show all of the Hoffman elimination products. If one product is…
A:
Q: Provide a plausible arrow pushing mechanism for the reaction below. OH OMe TSOH, H20 OMe
A: The answer is mechanism
Q: а.) b.) с.)
A: Acetals on hydrolysis gives corresponding aldehyde or ketone and alcohols. Let's see here,
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A:
Q: Na HO, Br
A: When an alcohol is treated with NaOH, the following acid-base equilibrium occurs. The…
Q: Who would compound X be in the following sequence of reactions? Select one:
A:
Q: 2. For the reaction scheme below: (i) Suggest a reagent X that could be used to accomplish the first…
A: Transformation of alcohol to it's halide, generally used SOX2 or PX3 (where, X = halogen atom). In…
Q: Give the expected product of the following Michael addition. base 6.
A: Michael addition is nucleophilic addition of a carbanion (enolate ion) to the alpha-beta carbonyl…
Q: HO OH H+ to
A:
Q: 4) Provide the major organic product(s) in the reaction below. Br/light
A: This reaction proceeds through free radical mechanism.
Q: Perform a retrosynthetic analysis by working backwards two steps in the synthesis below. Identify…
A: Given target molecule is alkyl halide.
Q: 2. For the following amines A and B, show all of the Hoffman elimination produc If one product is…
A: The Hofmann Elimination is an elimination reaction of alkylammonium salts that forms C-C double…
Q: Q2(a): mechanism of synthesis of [COCI(NH3);]Cl, starting from [COCO;(NH3),]NO; Write the and…
A: Coordination complexes compounds of Co(III) have kinetically inert nature and they undergo ligand…
Q: Provide a plausible arrow pushing mechanism for the reaction below. Note that the starting material…
A: The given reaction is diazotisation reaction. In this reaction a diazonium salt is formed as a…
Q: OH + + b) What direction would you expect the equilibrium to lie and by how much? c) If an amine was…
A: A question based on equilibrium concept that is to be accomplished.
Q: Complete the mechanism and draw the final product for the Grignard reaction below. It is not…
A: Its an organometallic reagent used as very strong nucleophile in organic chemistry.
Q: Provide a plausible arrow pushing mechanism for the reaction below. Note that the starting material…
A: Applying basic rule of reagents and chemical reaction.
Q: 1. Consider the acid-base equilibrium shown below on the left side. Here, the base abstracts a…
A:
Q: Base Compound 2 Compound 1 B.
A:
Q: HO
A: Alcohols are the hydrocarbon that contains a hydroxyl (-OH) functional group. Alcohols on treatment…
Q: 1. Excess LIAIH4 MeO НО 2. H2о
A: Given reaction: We have to write the mechanism of the reaction.
Step by step
Solved in 2 steps with 2 images

- What is the equation for the expected reaction between dimethylformamide (DMF) and sodium iodide? What are possible reaction products? (Please include structures in explanation) If you were to change the concentration of the solvent (DMF) how would that affect the reaction (would more product or less product be produced)? (Please make sure to make answers easy to follow)I want the sequence of the reaction to get to the target for number 3 thank uMost imine formation reactions are performed in the presence of molecular sieves ormagnesium sulfate. What is the purpose of these added reagents?
- Why is it important to thoroughly vortex the contents of the vial after adding reagent 4 (Steps 3 and 4., stage 1). To ensure that the two immiscible solvents come into contact with each other, permitting the derivatization reactions to occur. B.To ensure that the solution is mixed thoroughly. To ensure that all the amino acids are transferred to the lower aqueous layer so that a full extraction occurs. To remove all the interferring compounds by transferring them to the upper organic layer Hint: Reagent 4 is the derivatizing agent dissolved in octane.Please show mechanims and explain the procedure for the following image.Given the information pictured, The final step of the reaction sequence uses PhLi. Provide a reason and possible mechanism by which this final elimination and hydrolysis reaction would occur.
- On the axes below, sketch the graphs which show the result of adding: 0.1M NaOH to 25cm3 of 0.1M ethanoic acid. 0.1M NaOH to 25cm3 of 0.1M H2SO42) Another method for the above reaction starts with 4-aminophenol hydrochloride (the conjugate acid of 4-aminophenol). The treatment of this the hydrochloride salt with sodium ethanoate (acetate) buffer produces 4-aminophenol which is then able to react with ethanoic anhydride as per our method. i. Why is 4-aminophenol hydrochloride not suitable for direct reaction with ethanoic anhydride? ii. Draw a mechanism (i.e. curly arrows) showing the deprotonation of 4-aminophenol hydrochloride by sodium ethanoate to form 4-aminophenol (the free base).based on this video: on Hinsberg Test Tests for Amines - MeitY OLabs (8.5min) https://www.youtube.com/watch?v=j5jgMUWri8U Write the reaction (two-step, in skeletal) of each test amine when tested in the Hinsberg Test.
- Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…Experimental observations show that thiocyanate (SCN- ), the conjugate base of thiocyanic acid (pKa = 1.1 at 25 °C), is quite soluble in neutral water at pH > 2. In the space provided below, explain this experimental observation. That is, why does SCN– readily dissolve in water when the pH > 2?At the start of lab, Anthony adds vanillin to his Erlenmeyer flask, then adds sodium hydroxide, and lets the suspension cool. i. With minimal words, identify the reaction that takes place between sodium hydroxide and vanillin. ii. Is sodium borohydride capable of inducing the same reaction as (i)? Explain.

