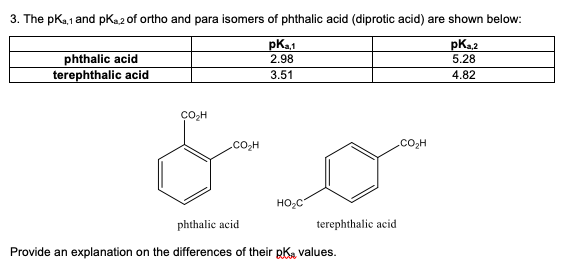
3. The pKa,1 and pKa,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below: Provide an explanation on the differences of their pKa values.
Q: Acid HNO3, 50°C O2N A B
A: Concept: it is a nitration reaction. pKa of HNO3 is -1.3, any acid lesser pKa than HNO3 will be used…
Q: In charts the pKa of acids are often given instead of the Ka values. For the 5 acids below predict…
A: There are two types of bases/acids. One is strong base/acid which is completely dissociable in into…
Q: Why was the sample being made alkaline with 3M KOH and heated for the qualitative test for NH4+?
A: The reason why was the sample being made alkaline with 3M KOH and heated for the qualitative test…
Q: A typical amino acid with one amino group and one carboxylic acid group, such as serine can exist in…
A: At lower pH that means in acidic conditions the carboxylate ion takes up proton and exists as…
Q: O) For a weak acid with a pKa of 5.5, at what pH would there be a 10-fold excess of the protonated…
A: Since you have asked multiple question, as per our company guidelines we are supposed to answer the…
Q: Look up the pKa values for trimethylamine, dimethylamine, methylamine, and ammonia. Predict the…
A: pKb tells about the basicity of any organic base. Higher the pKb value, lower would be the basicity.
Q: What fraction of iminodiacetic acid is in each of its four forms (H, A H,A, HA, A) at pH 3.357? The…
A: pH=3.35=-log [H+]or, [H+]=10-3.35=4.47×10-4
Q: Assume that for the reaction XTP XDP + Pi, the Keq =272024 (dimensionless). Calculate the standard…
A:
Q: How would the pKa values of carboxylicacids, alcohols, ammonium ions 1RNH32 + , phenol, and an…
A: The strength of a chemical substance acidity is determined by its pKa value.
Q: hat molar ratio of to H in solution would produce a pri Phosphoric acid BPO4), a triprotic acid, has…
A: Here the answer is 1.38
Q: Ka values of HA and HB weak acids are 4x10-7 and 8x10-4, respectively. These acids are water and…
A:
Q: me structure of Ethylenediaminetetraacetic acid (EDTA) is provided below. Provide the structure of…
A: From the above structure of EDTA given we can see that there are 2 N present in the EDTA which has…
Q: Sulfuric acid (H2SO4) is called a diprotic acid because it has two acidic protons. The pKa for the…
A:
Q: A student performs the pka of an Indicator experiment with phenol red, and obtains the following…
A: Given, Wavelength = 560 nm Aln-= 0.712AHln=0.0003
Q: Why is it easier to see all three pKa values in a tripolic acid but not all 3 equivalence points?
A:
Q: Write the mathematical expression for (c) metastable ion, indicating the terms involved. Explain the…
A: The mathematical expression of Metastable ion(m*) is given as m* = (m2)2 /m1 Here, m2 = mass(m/z) of…
Q: Calculate Ka values from the following pKa’s: (a) Acetone, pKa 5 19.3 (b) Formic acid, pKa 5 3.75
A: pKa = -log Ka Ka = 10-pKa (a) Acetone, pKa = 19.3 Ka = 10-pKa Ka = 10-19.3 Ka = 5.01 x 10-20 (b)…
Q: What is the dominant form of arsenic acid (H3A) at pH =9.23 ? Given: pKa1 = 2.24 , pKa2 = 6.96, pKa3…
A: At first equivalence point, pH will be given by: pH=pKa1+pKa22=2.24+6.962=4.6 At this point, the…
Q: Match the following tests to their corresponding positive visible result. v Ethanolic AGNO3 I.…
A: The test for detection of organic functional group and their confirmation is given below
Q: 2. Answer these questions about separating benzoic acid (pKa = 4.2) and aniline (which is basic, pKa…
A: Reverse phase high pressure liquid chromatography (HPLC) is a separating process where a non-polar…
Q: what is the purpose of hydroxylamine hydrochloride in the spectrophotometic determination of iron in…
A: (or) So, Hydroxylamine hydrochloride 10% solution (10 g in 100 mL of distilled water) was used to…
Q: Diclofenac acid has low aqueous solubility (and thus is formulated as diclotenaC sodium. It is…
A:
Q: Consider an amino acid with the following pKa values: pKa1 = 2.71 and pK, = 9.31 At what pH value in…
A: Amino acids are amphoteric in nature due to the presence their ionizable α-amino and α-carboxylic…
Q: Discuss what effect, if any, would you expect the following changes to experimental conditions to…
A: Methyl orange is a weak acid. It has a red colour in non-ionized from and is used as an indicator in…
Q: Deduce expressions for the fractions of each type of species present in an aqueous solution of…
A:
Q: 6. Carbonic acid (H2CO3) has two ionizable protons, with pKa around 6.1 and 10.2. (a) Which species…
A:
Q: Why does methyl orange have a lower pKa value (pKa~3.5) than methyl red (pKa ~5.1)? Provide detailed…
A:
Q: Nicotine (structure shown below) has pKa values of 3.2 and 8.1. Account for these values. In your…
A: According to the acid-base theory put forward by physical chemist Gilbert N. Lewis the chemical…
Q: The pKa values of the conjugate acids of 2-aminopyridine (6.71) and 4- aminopyridine (9.11) are as…
A: It is given that 2-amino pyridine conjugate acid has a pKa value of 6.71 and 4-aminopyridine…
Q: Ethylenediaminetetraacetic acid (EDTA), is one of the most popular chelating agents. It has two…
A: The pKa values denote the respective pH values where the proton gets deprotonated. The pKa values of…
Q: wnte the mathematical expression for metastabie ion indicating the terms invoived. Explain the…
A: The mathematical expression of Metastable ion(m*) is given as m* = (m2)2 /m1 Here, m2 =…
Q: You want to use acid-base extraction to separate compound A (pKa = 4.9) from compound B (pKa = 9.5).…
A: Based on the pKa values of compounds A and B, it is evident that compound A is more acidic than…
Q: Determine AHnn for the generic reaction below. 3AB2 (9) • C28 ()-2CAB3 (aq) + AB (9)
A: Standard enthalpy of formation for a reaction is equal to the sum of enthalpy of formation of…
Q: Given the following alpha fraction plot, what is the pK31 of the polyprotic acid? 1.0000 0.8000…
A:
Q: 3-(N-Morpholino)-2-hydroxy-propanesulfonic acid (MOPSO) is a common buffer used in biology and…
A: b. Molar concentration of the added acid =0.015 mol500×10-3 L=0.03 mol/L
Q: Explain the difference in acidity between p-methoxybenzoic acid (pKa = 4.46) and m-methoxybenzoic…
A: Methoxy group has both +R effect due to the lone pair of electrons present on the oxygen atom and -I…
Q: What are the structures of threonine in each part of its titration curve? Give a clear handwritten…
A:
Q: .Applications of conductivity measurements. )Determination of degree of dissociation )Determination…
A: The conductivity of a substance is defined as the ability or power to conduct or transmit heat,…
Q: Given the information about the following acids: i= HW pKa = 2 ii= HX pKa = 6 iii= HY pKa = 10…
A: The acid with higher pKa is lower acidic and more basic . And with Lower pKa is higher acidic and…
Q: What are the net charges of the following five-amino-acids peptides at pH 14.0 RRKKQ? (please…
A:
Q: Retinol is 83.84% C, 10.58% H, and 5.58% O by mass. What is the molecular formula of retinol? Add…
A: The empirical formula is the chemical formula of any compound that represents the proportions of…
Q: Most of the pKa values given in this text were determined in water. How would the pKa values of…
A: The strength of a chemical substance acidity is determined by its pKa value. It is actually the…
Q: The gas-phase basicity of triphenylamine (876.4 kJ mol-1) is less than that of triphenylphosphine…
A: Given that The gas-phase basicity of triphenylamine is 876.4 kJ mol-1 The gas-phase basicity of…
Q: Acetic acid (CH3COOH, abbrev. HA) is a very common weak acid with pKA = 4.75 (at 25oC and low ionic…
A:
Q: Write the significances of Poly Dispersity Index (PDI) and calculate the PDI for given data. Number…
A: Ploy Dispersity Index (PDI) tells us the distribution of molecular weights in a polymer. It is given…
Q: Consider the structure of quinine below and identify a suitable analytical technique for its…
A:
Q: and K2 = 5.42 x 10. In a solution made by dissolving 25.00 g of oxalic acid in enough water to a…
A:
Q: a solution of phenylhydrazine (C6H5-NH-NH2) with ionic strength 0.1 mol/L has pH = 8.13. Determines…
A: Reaction of phenyl hydrazine with acid gives phenyl hydrazinium ion and the reaction and the…
Q: Use Pauling’s First and Second Rules to determine the pKa values of bromic acid and selenous acid as…
A: Explanation to the correct answer is given below.
Q: spectrochemical series which is the interaction relationship of Lewis bases with metals…
A:
3. The pKa,1 and pKa,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below:
Provide an explanation on the differences of their pKa values.
Step by step
Solved in 3 steps with 2 images

- A typical amino acid with one amino group and one carboxylic acid group, such as serine can exist in water in several iconic forms. Suggest the forms of the amino acid at low pH and at a high pH. Amino acids generally have two pKa values, one in the range of 2 to 3 and the other in the range of 9 to 10. Serene, for example, has pKa values of 2.91 and 9.21. Using species such as acetic acid and ammonia as models, suggest the origin of the two pKa values. Glutamic acid is an amino acid that has three pKa’s: 2.10, 4.07, and 9.47. Draw the structure of glutamic acid and assign each pKa to the appropriate part of the molecule. An unknown amino acid is titrated with strong base, producing the following titration curve. Which amino acid are likely candidates for the unknown?Acetic acid (CH3COOH, abbrev HA) is a very common weak acid with pKa = 4.75 (at 25oC and low ionic strength) A] Write down the dissociation equation of the weak acid HA in aqueous solution, derive the respective formulae for the dissociation constant Ka and pKa. B] Calculate pH of the diluted solution of acetic acid with concentration, c = 0.05 mol/dm3 C] What is the approximate pH of the aqueous acetate buffer solution composed of dissolved mixture of acetic acid (HA) and sodium acetate (NaA) in the same respective concentrations? What is the working pH range of acetate buffers? D] How can you prepare exactly 1 dm3 of acetate buffer solution with required pH 5.0 and the total concentration [HA + NaA], c = 0.01 mol/dm3 if the stock solution of acetic acid (HA) with concentration, cHA = 0.1 mol/dm3, anhydrous sodium acetate (NaA, MT = 82.03 g/mol) and purified water are available? E] Acetate buffer solution may be also prepared by the partial neutralization of HA with NaOH: 20 cm3 of…Acetic acid (CH3COOH, abbrev. HA) is a very common weak acid with pKA = 4.75 (at 25oC and low ionic strength). Calculate pH of the diluted solution of acetic acid with concentration c = 0.05 mol dm-3.
- The pKa values of the conjugate acids of 2-aminopyridine (6.71) and 4- aminopyridine (9.11) are as shown. Which nitrogen is the more basic in these structures and why?Deduce expressions for the fractions of each type of species present in an aqueous solution of lysine as a function of pH and plot the appropriate speciation diagram. Use the following values of the acidity constants: pKa (H3Lys2+) = 2.18. pKa (H2Lys+) = 8.95, pKa(Hlys) = 10.53.spectrochemical series which is the interaction relationship of Lewis bases with metals oftransition: a) Explain the order of each base in relation to Fe3+. Why?b) Determine the order of bases with respect to NH4+. Why?
- Phosphoric acid (H3PO4) has 3 dissociable protons with the pKa values shown. Which form of phosphoric acid predominates in a solution at pH 4? Explain. Acid pKa H3PO4 2.14 H2PO4- 6.86 HPO42- 12.42 What can be described if a sample analyzed by AAS shows the following results:Phosphotungstic acid is one of a number of heteropolyacids. Name additional elements you can bind into heteropolyacids. Why are certain heteropolyacids so important for gravimetric determination of the heteroelement? Briefly describe, draw and discuss at least three of their structures.
- A newly synthesized drug was found to have a carboxyl group, an imide and a sulfonamide group. The first type of method to consider in this drug molecule’s quantitatve analysis is: a. Non-aqueous Alkalimetryb. Non-aqueous Acidimetryc. Aqueous Alklaimetryd. Aqueous AcidimetryA typical amino acid with one amino group and one carboxylicacid group, such as serine, can exist in water in several ionic forms. (a) Suggest the forms of the amino acidat low pH and at high pH. (b) Amino acids generally havetwo pKa values, one in the range of 2 to 3 and the other inthe range of 9 to 10. Serine, for example, has pKa values of2.19 and 9.21. Using species such as acetic acid and ammoniaas models, suggest the origin of the two pKa values.(c) Glutamic acid is an amino acid that has threepKa's: 2.10, 4.07, and 9.47. Draw the structure of glutamicacid, and assign each pKa to the appropriate part of themolecule. (d) An unknown amino acid is titrated withstrong base, producing the following titration curve.Which amino acids are likely candidates for the unknown?Consider the following data. For the acid: HA- Ka = 7.46⋅10−87.46⋅10-8 Calculate the Kb for A2-. Report your answer in scientific notation using 3 sig figs.

