Q: Which of the following can undergo both radical and cationic polymerization? H2C=CH2 II OCH3 CN IV…
A: For cation and radical process the concerned cation and radical should form. That means these…
Q: Polymerisation reactions usually occur with molecules which have no carbon-carbon double bonds o…
A: Polymerisation is a process through which a large number of monomers molecule react together to form…
Q: What t pe of polymer would be produced in the following polymerization reaction? H,CO,C. HO co,CH,…
A: The polymer can be an addition polymer or condensation polymer. Addition polymer will be formed by…
Q: d) Polymer Product NC S AIBN
A: The polymer product is :
Q: 4. Predict the repeat unit for the following polymerizations: а. H2O2 CH3 b. NCO OCN
A: Since you have multiple questions we will answer the first one for you. To get the remaining…
Q: Which of the following pairs of compounds can be used directly for the synthesis of a step-growth…
A: We are to find out whi h of given pairs of reactants yield step growth polymers directly.
Q: #6: The monomer shown below is polymerized via condensation polymerization with loss of water to a…
A:
Q: Rank the following groups of monomers from most able to least able to undergo anionic polymerization
A: Polymers are formed from linking small units called monomers and they are classified into Chain…
Q: 3. Draw the structure of the product of polymerization of 1,2-dichloroethylene, CHCICHCI.
A: 1,2-dichloroethylene exist in two geometrical isomeric form : cis and trans
Q: 11) What happehs during the propagation step in a polymerization? A) Number of Radicals Increase B)…
A: In this question, we will see the correct answer for the propagation step in a polymerization. You…
Q: In each of the following pairs of monomers, which is the more reactive towards cationic…
A:
Q: (a) Hard contact lenses, which first became popular in the 1960s, were made by polymerizing methyl…
A: The polymerization of methyl methacrylate to form poly(methyl methacrylate) is an addition…
Q: Rank the following groups of monomers from most able to least able to undergo cationic…
A:
Q: Polymerization of 1 3 -hexadiene can yield in either cis (Z) or trans (E) polymer.on the other hand…
A: The structures of 1,3 -hexadiene and 1,4 -hexadiene are shown below: In 1,3 -hexadiene, the two C=C…
Q: Which polyrner shown below is formed via the base-initiated polymerisation of propylene oxide? Click…
A:
Q: Rank the following compounds in order of increasing ability to undergo anionic chain-growth…
A: The chain growth polymerization reaction which involves the polymerization of vinyl monomer along…
Q: Poly(ethylene terephthalate) (PET) can be prepared by this reaction. Propose a mecha- nism for the…
A: Poly(ethylene terephthalate) is made by reacting Dimethyl terephthalate and ethylene glycol in the…
Q: You are preparing polyesters from the following two monomers: CH,OH CH3CH2 C-CH,OH ČH,OH Co.H HO,C…
A: Polymerization can be described as a chemical process that results in the creation of polymers in…
Q: 25. (3) Draw the structure of the monomer from which the polymer can be synthesized. -CIN n
A: Monomers are the simple basic unit through which polymers with high molar mass is synthesied.
Q: From what monomer(s) is the following polymer made? CH, H. H. The correct answer choice is not…
A: In the polymerization of 2,3-dimethylbuta-1, 3-diene, acts as monomer, polymer is formed. The…
Q: 10. Complete the following polymerization reactions. Name the polymer that forms. a) H Mod b) c) H H…
A:
Q: What monomer(s) are needed to synthesize each polymer? naf а. b. е. F CIF CIF CI C. ČO,CH3 ČO2CH3
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Which set of monomers was used to produce the polymer shown? Click on a letter A through D to…
A: Manomers are the compounds that there repetative addition form the polymer.
Q: Draw a short segment of the polymer formed from cationic polymerization of…
A: In the cationic polymerization reaction, we react the reactant with a small amount of reagent which…
Q: Rank the following groups of monomers from most able to least able to undergo cationic…
A: Polymers are formed from linking small units called monomers and they are classified into chain…
Q: What polymer is formed by chain-growth polymerization of attached monomer ?
A: The monomer unit is:
Q: CH3 CH3 HO Direct Polycondensation но ČH3 'n H* Ring-opening polymerisation
A: The reaction D to E is a direct polycondensation reaction which means that the reactant is an…
Q: Draw short segments of the polymers obtained from the following monomers. indicate whether the…
A: Chain growth polymerization involves the formation of polymer using unsaturated compounds while…
Q: Rank the following groups of monomers from most able to least able to undergo cationic…
A: Given compounds are
Q: Draw short segments of the polymers obtained from the following monomers. indicate whether the…
A: The polymerization in which monomers having one or more double bonds undergo repeated addition in a…
Q: Rank the following groups of monomers from most able to least able to undergo anionic…
A: Polymers are formed from linking small units called monomers and they are classified into Chain…
Q: Draw short segments of the polymers obtained from the following monomers. indicate whether the…
A: The given monomers are:
Q: Draw short segments of the polymers obtained from the following monomers. indicate whether the…
A: A polymer is a large unit made by linking repeating small units called monomers. Chain growth…
Q: Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but…
A: Given: products of the radical polymerization of styrene.
Q: What polymer is formed by the radical polymerization of each monomer?
A: The radical polymerization reaction occurs in the presence of radical initiator which helps in…
Q: heat HN ElN (weak base) OCH conc. conditions draw 2 or 3 repeat units of the polymer product H2SO4…
A:
Q: Rank the following groups of monomers from most able to least able to undergo cationic…
A: To rank the below monomers from the most able to least able to undergo cationic polymerization.
Q: What will be the most suitable initiator used to polymerize the following monomers? Give ONE (1)…
A: Polymerization is a procedure to make large molecules using corresponding smaller bits. This process…
Q: (B) HO Co,H OH HO2C
A: Polymerisation of acid and alcohol forms polyesters . They are commercially useful fibres in…
Q: 4.52 Plexiglas, a clear plastic used to make many molded articles, is made by polymerization of…
A: Polymerization is the process of formation of polymer from the maneric units .
Q: Choose the correct option. Guess the type of polymerization if you are provided with…
A: Polymerization is the process of combining small molecules to form macromolecules of higher mass…
Q: 3. Why is therèñð išôtáčtíć or syndiótacticform of polyethylene? a. mon omer is symmetrical. b. mon…
A: Answer - The correct option is (d) monomer can form only graft or random polymer Explanation -…
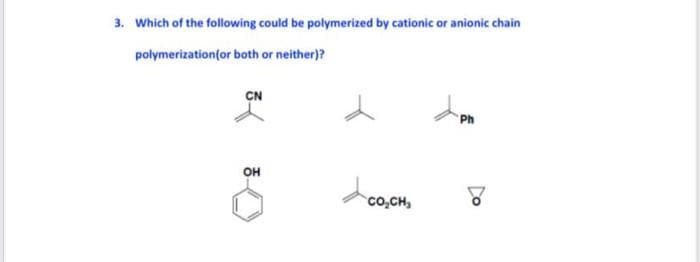
Q: Which of the following compounds can either undergo cationic, anionic, or free radical…
A: Unsaturated alkene can undergo cation anion and free radical addition polymerization reaction.
Q: Rank the following groups of monomers from most able to least able to undergo anionic…
A: Polymers are formed from linking small units called monomers and they are classified into Chain…
Q: Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but…
A: The radical formed in radical-Polymerization of styrene is The radical formed in X is benzyl free…
Q: Rank the following compounds in order of increasing ability to undergo anionic chain-growth…
A: Alkenes bearing anion stabilizing groups like Phenyl, Vinyl, Cyano, Nitro and Ester can react with…
Q: 10. What is the product of the following step-growth polymerization? OH HO"
A: Bifunctional monomers are combined to form the polymer chain by the step-growth polymerization. In…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Rank the following monomers in order of increasing reactivity toward cationic polymerization (least reactive to most reactive).Rank the following monomers in order of increasing reactivity toward anionic polymerization (least reactive to most reactive).Please show the product and mechanism for the cationic polymerization of 2-butene using BF3/H2O to catalyze polymerization. Do you expect this to be an effective polymerization strategy for this monomer? Why or why not?
- Styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic polymerization is an example of electrophilic addition to an alkene involving carbocations. a.) Draw a short segment of the polymer formed by the polymerization ofA.b.) Why does A react faster than styrene (C6H5CH=CH2) in a cationicpolymerization?What small molecule is generated during the polymerization in the above molecule?List the following group of monomers in order of decreasing ability to undergo cationic polymerization.
- Which reaction—dehydration synthesis or hydrolysis—converts a polymer to its monomers? Which one convertsmonomers to a polymer? Explain your answerDraw a structural formula of the polymer resulting from base-catalyzed polymerization of each compound. Would you expect the polymers to be optically active? (S)-(+)-lactide is the dilactone formed from two molecules of (S)-(+)-lactic acid.When certain cycloalkenes are used in metathesis reactions, ringopening metathesis polymerization (ROMP) occurs to form a highmolecular- weight polymer, as shown with cyclopentene as the starting material. The reaction is driven to completion by relief of strain in the cycloalkene.What products are formed by ring-opening metathesis polymerization of each alkene?
- 1.- How does the molecular weight change vs % conversion in a step polymerization? Explain this behavior and why does it occur? 2.- Name at least 3 requirements for obtaining a high molecular weight polymer by step polymerization. 3.- How can you minimize cyclization reactions in a step polymerization? Explain with equations and narrative Why do you want to minimize cyclization? Is it possible to eliminate cyclic products completely?(a) Which monomer is most likely to undergo anionic polymerization? Justify your choice. ( b)Which one ismost likely to undergo cationic polymerization? Justify your choice.Which monomer and which type of initiator can you use to synthesize each of the following polymers?



