Q: Tautomerization in base resembles tautomerization in acid, but deprotonation precedes protonation in...
A: Keto-enol tautomers are the isomeric forms which represents the same compound but variation in the p...
Q: Question attached
A: Since (CH3)3COK is a strong and bulky base, it only favours E2 elimination with secondary alkyl hali...
Q: Write the Lewis structure for each molecule/ion. H3COCH3
A: The given compound is dimethyl ether. The Lewis structure for this compound can be drawn as follows,
Q: Mg+ 02 --> MgO 13. Balance the following equation: Your answer
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: Name the element that corresponds to each electronic configuration. 1s^2, 2s^2, 2p^6, 3s^2, 3p^5
A: Electronic configuration refers to the systematic arrangement of electrons in the various shells and...
Q: Calorimetry is when two substances at different temperatures are mixed and allowed to come to equili...
A: Given data: Mass of ice(m1 ) = 38 g Initial temperature(T1 ) = -60CMass of water(m2 ) ...
Q: Determine the number of valence electrons in dimethyl ether ((CH₃)₂O) and then draw the correspondin...
A: Lewis structure is a structural representation of a molecule along with its valence electrons. Elect...
Q: THEOKETICAL QUESTIONS: I-what are the concentration cells? Explain an elec trolyte concentra tion ce...
A: Hello. Since there are more than one question, the first question shall be only answered in this cas...
Q: Draw the product for the attached reactions.
A: Alkenes react with water in the presence of sulphuric acid to form alcohols. The formation of major ...
Q: How many total atoms are in 0.50 moles of CO2? A:3.01 x 10^23 B:6.02 x 10^23 C:9...
A: Avogadro number (NA) The number that corresponds to 1 mole of a substance is called Avogadro number....
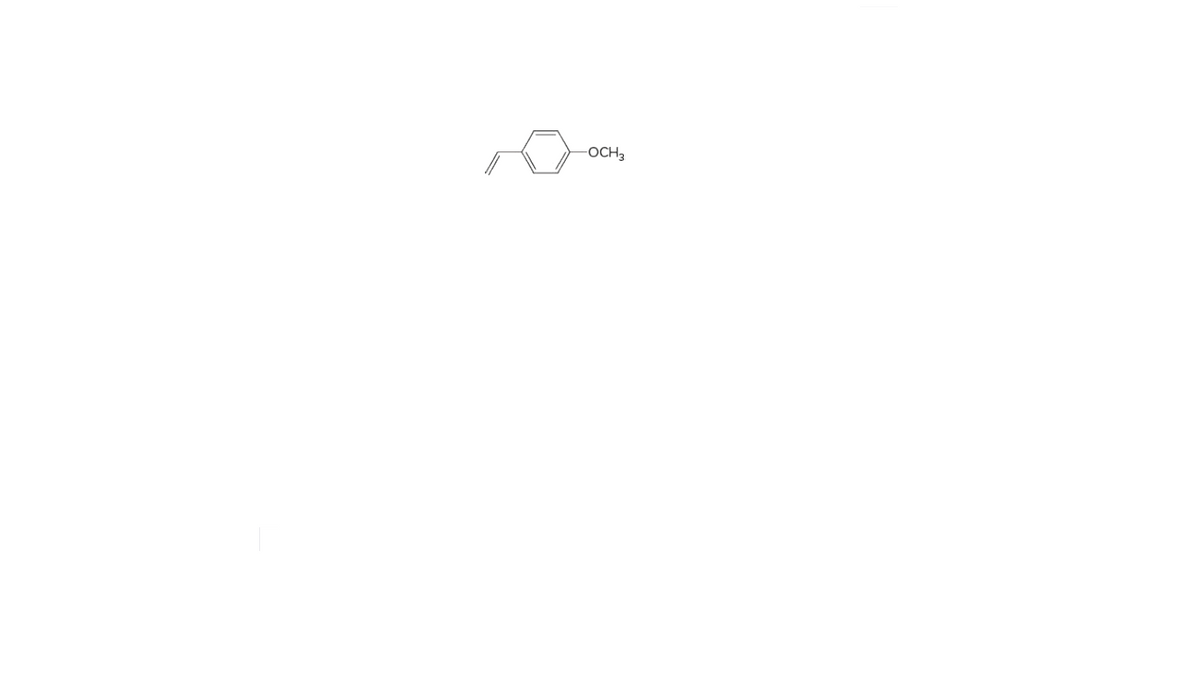
Q: Identify each organic compound as an alkane, alkene, alkyne, aromatic hydrocarbon, alcohol, ether, a...
A: Name of given compound is given as
Q: Can you please give me all 3 answers in detail and show the work please.
A:
Q: Which of these 4 major clades of living organisms (Bacteria, Plants, Animals, and Fungi) produces th...
A: A question to determine the specific clade of living organism which is the source of biologically im...
Q: What is the correct name for the following compound? H2O A. Hydrogen oxide B. Hydrogen oxolate C. D...
A: WaterH2O is a covalent compound containing hydrogen and oxygen.Almost all the compounds are easily s...
Q: (a) Draw the condensed structure of the tripeptide Gly-Gly-His. (b) How many different tripeptides c...
A:
Q: The gas-phase reaction between nitrogen and oxygen was carried out in a device designed to maintain ...
A:
Q: 10. What are shared in covalent bonds?* Cations Protons Electrons Anions 11. What type of reaction d...
A: The answer to the given three questions can be given as -
Q: Calculate the ΔHo for methane gas , CH4, produced from graphite and hydrogen gas according to the fo...
A: The enthalpy change is defined as the amount of heat absorbed or evolved in the transformation of th...
Q: ) A child blows a bubble that contains air at 28 C and has a volume of 23 cubic meters at 1 atm. As ...
A: Whether the bubble will get larger or smaller as well as the new volume of the bubble in this case c...
Q: According to the scheme below, How much energy is required to evaporate 54.06 g water (Molecular wei...
A:
Q: Sunscreen contains compounds that absorb ultraviolet light. When sunscreen is applied to skin, it pr...
A: First, find the wavelength corresponding to the maximum absorption of TDSA from the graph: It is ar...
Q: Synthesize attached compound from cyclohexanol using any other organicor inorganic compounds.
A: Cyclohexanol reacts with SOBr2 to give bromo-derivative which reacts with Mg, ether to form Grignard...
Q: 1.0L
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any s...
Q: Identify and describe the triple point of a gas from its phase diagram.
A: Any substance can exist in liquid, solid or gaseous form. Gaseous phases have the highest volume whe...
Q: 15a
A: Significant figures are the total number of meaningful digits that help in improving the accuracy of...
Q: Summarize each of the simple gas laws (Boyle’s law, Charles’s law, and Avogadro’s law). For each, ex...
A: Boyle's Law: Boyle’s law states that as long as the temperature and the amount of gas remain constan...
Q: In the reaction: 2H2O → 2H2 + O2 how many grams of O2 are formed from decomposing 18...
A:
Q: For the following, how would decreasing the temperature affect the solubility of the solute? NaCl (s...
A: ΔH = + 3.9 kJ/mol i.e. reaction is endothermic.
Q: Draw a structure for alkane. 4,4-diethyloctane
A: Given compound name, 4,4-diethyloctane
Q: explain Commercial Importance of Alkenes
A: Given Explain Commercial Importance of Alkenes
Q: Which two gas samples have the same total number of molecules?
A:
Q: Explain the procedure for synthesis of transition metal complexes of hydroxycinnamic acids? For the ...
A: P-coumaric acid and ferulic acid are used to synthesis the transition metal complexes of hydroxycin...
Q: Predict the products and write a balanced molecular equation for reaction. If no reaction occurs, wr...
A: The type of reactions in which the exchange of ions takes place on the reaction between the two comp...
Q: Draw the major product formed in each reaction.
A: major products formed in each reaction are
Q: Imagine you must prepare 50 mL of a 2M HCl solution. Provided that you only have a concentrated HCl ...
A: To prepare 50 ml of a 2M HCl solution:
Q: Based on what you just learned about ionization energies, explain why valence electrons are more imp...
A: The electrons in the innermost shell are called the core electrons while the electrons in the outerm...
Q: Predict the position of equilibrium and calculate the equilibrium constant, Keq, for acid-base rea...
A: We know that strong acids will donate protons easily and strong base will accept proton easily. That...
Q: 3. Pure (glacial) acetic acid doesn't conduct electricity while 1.0 M aqueous acetic acid does, some...
A: Acetic acid is a weak acid . It does not dissociate so readily into its constitute ions. Pure acetic...
Q: Molybdenum has a BCC crystal structure, an atomic radius of 0.1363 nm, and an atomic weight of 95.94...
A: For BCC structure, number of atoms per unit cell = 2 Radius = 0.1363 nm Edge, a∘ =43×radius=43×0.136...
Q: Cylinder A has a movable piston and contains hydrogen gas. An identical cylinder, B, contains methan...
A: The gases are found to obey the gas laws fairly well if the pressure is low or the temperature is hi...
Q: A piece of metal that is a cube with sides of 1.66 cm has a mass of 69.000 g. Calculate the density...
A: TO calculate the density of the metal, first of all we determine the volume of the metal cube.
Q: Describe how the particles change in position and movement as the two substances are mixed
A: The two important factors that are responsible in the determination of the arrangement of the partic...
Q: What is molar solubility?
A: Molar solubility, which is directly related to the solubility product, is the number of moles of the...
Q: A new linear temperature scale, degrees Zunzol (∘Z), is based on the freezing point and boiling poin...
A: Boiling point is defined as the temperature at which the vapor pressure of the liquid state will bec...
Q: Which property of carbon is related to its ability to form a large number of compounds?
A: Carbon atom has two main characteristic properties: Tetravalent nature and catenation.
Q: Is the change in enthalpy for a reaction an extensive property? Explain the relationship between ΔH ...
A: The enthalpy of the reaction is considered as the state function that is it depends only on the init...
Q: 7. What mass of nitrogen is required to react with 16 grams of oxygen? 2N2+ 502 --> 2N205 (Hint: 3 s...
A: as per Q&A guidelines of portal I solve first question because it comes under multiple question ...
Q: Sketch a periodic table (without element symbols). Include the correct number of rows and columns in...
A:
Q: Describe Thomson’s model of the atom. How might itaccount for the production of cathode rays?
A: The atomic model proposed by Thomson was the plum-pudding model. According to Thomson's atomic model...
Q: C
A: length is fundamental property of matter, its SI unit is meter.
What
Step by step
Solved in 2 steps with 2 images


