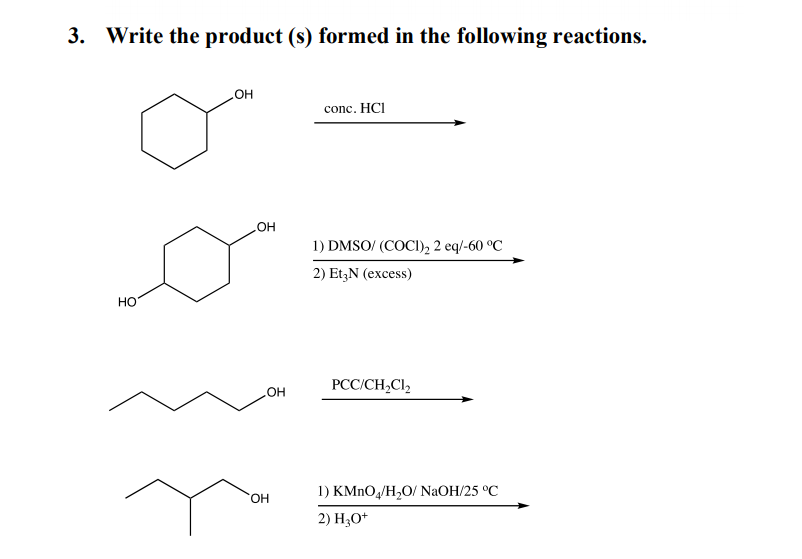
3. Write the product (s) formed in the following reactions. но conc. HCI 1) DMSO/ (COCI), 2 eq/-60 °C 2) Et;N (excess) HO PCC/CH,Cl, 1) KMnO,/H,О/ NaOH/25 °C HO. 2) H,O*
Q: Complete the missing Reagent and proclucts in the reactron Sequien ce, the questver Marlc indeecbe…
A: The species that are electron deficient in nature are termed as electrophiles. When an electrophile…
Q: 14. Acetylene gan, CH, can be prodaced by the seachion of cala CaC, () + 2H,0 - How many lites of…
A:
Q: Conc. KMNO, heat HO. Cs Scanned with Camicanner
A: Here the alkene oxidises with KMnO4 yield a mixture of carbonyl compound and carboxylic acid.
Q: Provide the missing compounds and reagents in the reaction scheme. Identify reagent 5. он reagent 2…
A:
Q: Write down land/or the reaction conditions for the fallowinng major producto), ar other recie…
A:
Q: What product (or product mixture) is expected for the following reaction? На, Pd/C ???
A: Hydrogenation of a double bond in the presence of H2 and a metal catalyst occurs through SYN…
Q: What is the final product, 'Z', of the following synthesis? of a 1. KMnO4, OH, heat 2. H₂O* I a) I…
A:
Q: -B2 → products in vessels (a)-(d) are 1:2:4:8. Unshaded spher ent B2 molecules present at the…
A: For this reaction, Rate law is : Rate = k • [A2]^m • [B2]^n
Q: In each reaction box, place the best reagent and conditions from the list provided. 1) Mg, Et,O Br…
A: The detail explanation of the organic reaction is given below
Q: Amion Elimination Test Oburmations added heagenter) Obreyation Ba (NO3), while ppt HNO, Fe (NC),…
A: Anion is a negatively charged species and it is prepared by the acceptance of electrons by the…
Q: A reaction for converting ketones to lactones, called theBaeyer–Villiger reaction, is used in the…
A: The reaction for the oxidation of ketone to lactone by hydrogen peroxide is given as, In the above…
Q: What are the volume (uL), moles, and/or weight (mg) of each reagent and solvent in: 50 mg of the…
A:
Q: a) H,CO. AICI,
A:
Q: Determine the theoretical yield of the oxidation of cyclohexanol to cyclohexanone. 1.0g (0.01 mol)…
A:
Q: Chemical Engineering 7) Propose multistep syntheses for the following molecules. i EtO OEt MeO OMe
A: Note - Since the given question is a multiple question, hence I solved first question according to…
Q: of this reaction. Ignore ir yproducts.
A: Alcohol can be defined as the organic compound in which carbon atom is attached to hydroxyl group.
Q: to yield compound B Based oe the data peovided in a lable below caicalete the volume and the number…
A: The solution is as follows:
Q: Mg THE CH,OD Br 1. 2 Li, THF 2. Cul, THF 1. ethylene axide 2.dilute H2SO4 Fill in the following…
A:
Q: Which of the following correctly completes the reaction? AGNO3 + CaCl2→?
A: To find: The correct equation for the given reaction
Q: the following reaction. concentrated fuming NO2 H2SÓ4 OH ON O ON HO NO.
A: This is the concept of Organic reaction
Q: 2. Write the product(s) for the following chemical reactions. а. + Br2 + Br2 H2O H3C- -CH3 + KMNO4…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: If a student isolates 213 mg of the E,E-DBA (MW = 235.1 g/mol) product from the following reaction,…
A: Please find the answer attached.
Q: Create sywthesis worng at least your owN 6or the following reagants: ) Towes 2) LIAIRY )…
A:
Q: Union, Elimination Test Obuuations Added heaqenter) Oberation while ppt HNO, Fe(NO), NO 3, KMnO4…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Br HO H+ OH Br OF 1. PPh3 2. Buli ppms 2. H₂O, H+ H
A: This is a multistep reaction. In first step protection of carbonyl group occurred. In second step…
Q: WRite down the expression of Root mean squane end to end distence Of linean poly merRe GoR breely…
A: A model that describes polymer chains in the absence of long-range self-interactions is the freely…
Q: At constant pressure and 25 C, what is the Delta rH for the following reaction: 2C2H6 (g) + 7O2(g)…
A: Given that 12.5 g of C₂H₆ releases 752.3 kJ of heat. You want to calculate the heat liberated by 2…
Q: 5.11 (a) Develop an electron-equivalent fermentation energy-reaction for conversion of propionate…
A: Since you have asked multiple questions questions, we will solve the first question for you. If you…
Q: Match the following methods to their endpoint. * B. Appearance of the first C. A distinctive color…
A:
Q: what a schematic diagram is, and what processes/contents should be in it. thank you
A: In this question we have to clearfy about a schematic diagram and the process or contents should be…
Q: 2. Write suitable detailed mechanisms to account for the formation of the products in the following…
A:
Q: Added Reagenter) Obuymation while ppt Bo (NO), HNOg. Fe(NO), HNO3, KMnO4 Blood red aqueour layen;…
A: Qualitative analysis is a part of analytical chemistry. Anion analysis is a systematic approach to…
Q: 2 C5H10 (1)+15 O₂ (g) → 10 CO₂ (g) + 10 H₂O(l) a. Calculate 4H of the reaction. b. Calculate 4S of…
A: Dear student , since you have posted multiple parts questions we will allow to solve only first…
Q: 10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH,Br + H2S CH3SH…
A: a. The Keq at room temperature (25°C) is 2.335. GivenCH3Br+H2S⇌CH3SH+HBr∆G=2.1KJmol=-2100…
Q: Write theproduets of the Sn2 reaction Sn OCN+CH; Gr 4- a) write the meehanism b) drow į la leel the…
A: SN2 Reaction - Biomolecular nucleophilic substitution reaction between alkyl halide and strong…
Q: 1 Complete the following table by filing in the boses with the appropriate starting material,…
A: The reaction of an alkyne with sodium amide leads to the formation of a carbanion by removal of…
Q: 1. What conclusions can be reached given the following information? [Be specific regarding kinetic…
A: The explanation of kinetic and thermodynamic controlled reaction along with ΔG at equilibrium and at…
Q: ater could destroy the reagent before it has a chance to react with the est Grignard reagents also…
A:
Q: Write the products structures and mechanisms CO2H A = (cat.) HO (a) NC CN LOME ? solvent Ph (i)…
A: The Diels-Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene…
Q: which product would be generated in highest yield from the reaction sequence below
A: Nucleophilic substitution reactions are the reactions in which a nucleophile attacks the another…
Q: write a mechanism for the Pollow ing ) HzO, solvolysis A Na H OCHS 3) CH3I write a detailed synmesis…
A: We only answer one question from multiple questions. Please resubmit the other questions separately…
Q: Taking Ka for acidic acid as 1.8 x 10-5, calculate H3O+ , using the known concentraions of acidic…
A: Note : From the Ka value given for the acid, the correct name of the acid will be acetic acid not…
Q: following alkoxymercuration-demercuration reaction. 2) Na C .CH Close H,C 10:08 4/25/2 of
A: Applying concept oxymercuration of alkene.
Q: How to determine ΔHo for the attached reaction.
A: The enthalpy for the reaction is calculated as shown below.
Q: In each reaction box, place the best reagent and conditions from the list. 2. H,C H CH;CH, он но…
A: The reaction given is,
Q: does the reaction anyway. Provide the product of the reaction below, and identify why it's a problem…
A: These are the list of alcohol deprotecting agents: Benzyl .............H2/Pd-C Allylcarbonate…
Q: H,SO, (A) но 180° K,Cr,0, H,SO, (B) он (F) (E) (D) (C) NaH (G) + ethyl chloride -OH (H) (1) + )…
A: here we are required to predict the product of the reaction when different reagents are used
Q: 7:39 AM Fri Nov 6 * 39% A learn-us-east-1-prod-fleet01-xythos.s3.amazonaws.com Consider each of the…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Step by step
Solved in 2 steps with 1 images

- Please complete reactions in clear handwritten of all subpartsFill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.cmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)
- I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Solution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaF
- complete this reaction Fe³= ........agent (from-to) S2O3= .........agrnt (from-to).Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configurationIn the reaction below, 3-Buten-2-one reacts with 2,3-Dimethyl-1,3-butadieneand 1,3-Butadieneat 140°C and 30°C, respectively. Explain why these two dienes react at different temperatures.
- The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?In the reaction of NaBH4 with formaldehyde in methanol sovlent, the products are BH3, CH3OH and sodium methoxide. what is the transition state for the slow step of this reaction? please give structure.Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?

