300 Sugar (C12H22011) 260 KNO3 220 180 NaNO3 140 NaBr 100 KBr 60 KCI NaCI 20 Ce,($O)3 20 40 60 80 100 Temperature (°C) Figure 11.17 This graph shows how the solubility of several solids changes with temperature. Solubility (g solute per 100 g H20)
300 Sugar (C12H22011) 260 KNO3 220 180 NaNO3 140 NaBr 100 KBr 60 KCI NaCI 20 Ce,($O)3 20 40 60 80 100 Temperature (°C) Figure 11.17 This graph shows how the solubility of several solids changes with temperature. Solubility (g solute per 100 g H20)
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter12: Solutions
Section: Chapter Questions
Problem 12.65QE
Related questions
Question
Calculate the percent by mass of KBr in a saturated solution of KBr in water at 10 °C. as shown for useful data, and report the computed percentage to one significant digit.
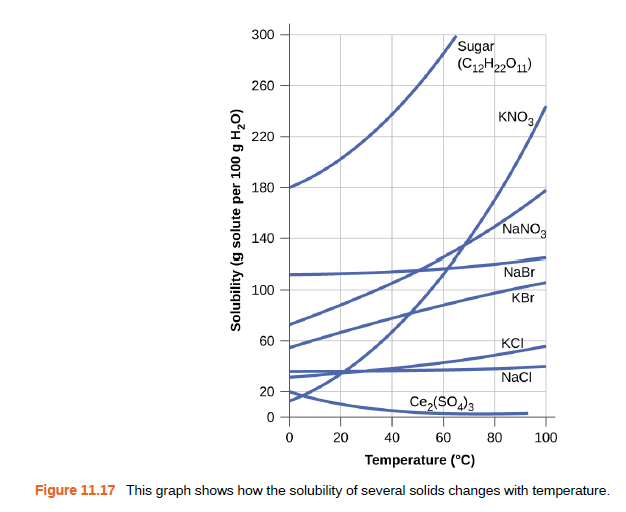
Transcribed Image Text:300
Sugar
(C12H22011)
260
KNO3
220
180
NaNO3
140
NaBr
100
KBr
60
KCI
NaCI
20
Ce,($O)3
20
40
60
80
100
Temperature (°C)
Figure 11.17 This graph shows how the solubility of several solids changes with temperature.
Solubility (g solute per 100 g H20)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
