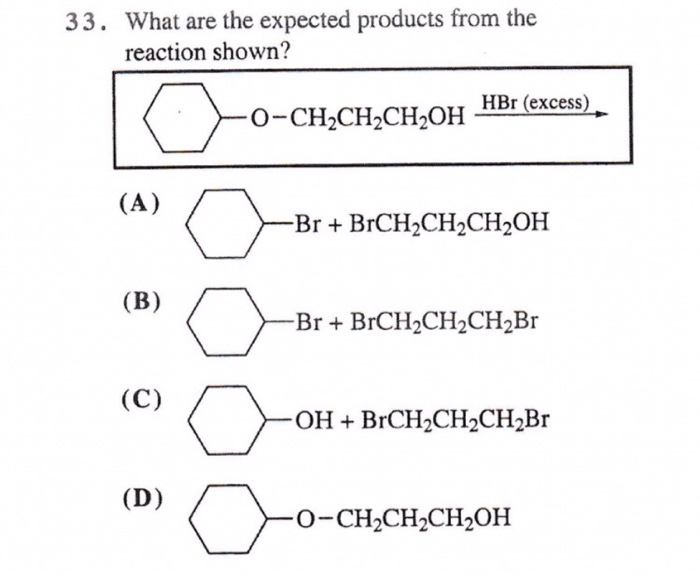
33. What are the expected products from the reaction shown? o (A) (B) (C) (D) -O-CH₂CH₂CH₂OH HBr (excess) -Br+ BrCH₂CH₂CH₂OH -Br+ BrCH₂CH₂CH₂Br -OH + BrCH₂CH₂CH₂Br o -0-CH₂CH₂CH₂OH
Q: Draw the most stable resonance form for the intermediate in the following electrophilic substitution…
A: Electrophilic aromatic substitution reactions are the characteristic feature of benzene and other…
Q: 2) Compare your calculated AH values with real "enthalpy of combustion" values using the links…
A: Enthalpy of combustion is defined as the heat produced during burning of one mole of a compound in…
Q: Draw the synthetic polymer that arises from reacting the two monomers shown below? NH
A: Polymerization is the process of formation of high molecular weight polymer from small molecules…
Q: Calculate the pH of a solution made by adding 200 mL of 1.00 M HCl to 400 mL of 1.90 M NH, solution.…
A: Answer :--
Q: What is the IUPAC name of the following compound? CH3 0 || CH₂CHCH₂CCH3
A: The naming of the organic compound is done using the IUPAC system. IUPAC system of nomenclature is…
Q: Nitrogen dioxide is one of the many oxides of nitrogen (often collectively called "NOx") that are of…
A:
Q: What is the major organic product for the following reaction? SO₂H CH3CH₂CI AICI3 ? Major Organic…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: Select the major product of the following reaction. 5- OH OH CrO/H OH II О Ш 5 OH IV OH "OH
A: The reaction of alcohols in the presence of CrO3 (chromium trioxide) and H+ (acid) is known as the…
Q: (c) H₂(g) H(aq) + H¯(aq) V spontaneity: O spontaneous O not spontaneous balanced equation: chemPad…
A: Sorry but this is according to Bartleby guidelines,you have asked multiple questions. We will answer…
Q: CH3 CH3 NC H CO₂CH3 H
A: Since you asked multiple images questions so as per Q&A guidelines of portal I solved first…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Answer:Compound that gives H+ ion in a solution is called as Bronsted-Lowry acid and any compound…
Q: Give detailed mechanism Solution with explanation needed..no need handwritten answer
A: The given reagent is sodium amide in liquid ammonia.It can act as a base or nucleophile.In this…
Q: Calculate values for the following cells. Which reactions are spontaneous as written (under standard…
A: The reaction is said to spontaneous, if the cell potential is positive.If E is negative, the…
Q: The mass of a proton is 1.00728 amu and that of a neutron is 1.00867 amu. What is the binding energy…
A: The formulas used to calculate the binding energy per nucleon are shown belowThe formula of the mass…
Q: Predict the dominant form of the amino acid shown below at pH 10.0. Given: pKa a-carboxyl 2.2, pka…
A: Amino acids are amphoteric biomolecules that have both the acidic carboxylic acid COOH group and…
Q: The source of Fe³+ ions in this lab was 0.00200 M Fe(NO3)3 and the source of SCN ions was 0.00200 M…
A: For the given experiment, determine the K value for beaker 1.
Q: 2. Draw the resonance structures of the following. " NH 3. Classify the following compounds as 1°,…
A:
Q: Conditions 1 Intermediate after Step 1 Ph3P 1. OsO4 2. NaHSO3 1. LDA 2. PhSeCl 3. H₂O₂ A D G M ? 3…
A: The detail synthetic pathway is below.
Q: Calculate the rate at which C3H8 is reacting if the average rate of formation at a given time of CO2…
A: The rate of the formation of the product or the rate of disappearance of the reactants is known as…
Q: A chemical reaction is run in which 523 Joules of heat are absorbed and 296 Joules of work are done…
A: According to the mathematical form of the first law of thermodynamics, Here, = the change in…
Q: Give detailed Solution with explanation needed
A: The given compound has an amide functional group.It has one carbonyl group present in it. Generally,…
Q: OH CH3CI AICI 3 ? Major Organic Product
A:
Q: 1) Which of the following compounds is stable under aqueous basic reaction conditions? Record your…
A: We have to identify the most stable compound under aqueous basic solution.
Q: Give the major product formed in the following reaction. CH₂ CH CH₂ D e 1. OH 2.OH. heat
A: Reaction of cyclohexanone with vinyl methyl ketone in the presence of base,
Q: Which of he following molecules has a dipole moment that is not zero? Group of answer choices all of…
A:
Q: Fill-in the missing reagents and products for the synthesis of HH Br NaCN SN2 A B N 1 H H
A: Note: As per the guidelines, solution of first question has been made. For the expert solution of…
Q: Consider the complete combustion of butanol (C4H100), as indicated in the balanced reaction below.…
A:
Q: Click the "draw structure" button to launch the drawing utility. Draw one of the aldoses formed when…
A:
Q: N-H 1) KOH 2) 3) H₂NNH₂ Br J K [H*], NaBH₂CN L [H], NaBH,CN M [H], NaBH₂CN NH₂ HN R R
A: ketone compound reacts with amine to form imine product which is reduced by NaBH3CN to form amine…
Q: A 1.00 g sample of a radioactive isotope decays such that 0.675 g will remain after 5.43 yr.…
A: The initial amount of the sample The amount left at time t, [A] = 0.675 gTime (t) = 5.43 yearsHalf…
Q: Given the ff information calculate the acceptable range for 99.9% of the population. Mean=6.32 ;…
A: The formula for calculating the acceptable range for a given population isHere,Mean is the…
Q: Conditions 1 Intermediate after Step 1 1. OsO4 2. NaHSO3 ou Ph3P 1. LDA 2. PhSeCl 3. H₂O₂ 1. PhSeCl…
A: Conversion of benzene into the desired product in three steps using the given reagents.
Q: 2. Predict all products of the following reaction and identify the major product (explain your…
A:
Q: The following reaction represents what nuclear process? 214 Pb 214 Bi + 82 83 A. positron emission…
A: The various nuclear processes are discussed belowPositron emission: In this process, the mass number…
Q: What is the hybridization for each of the labeled carbon atom in the following molecule? O O O O O 8…
A:
Q: How many molecules are in 4.5558 g of Rh(SO4)? Answer
A: Number of moles iscalculated by dividing given mass with molecular mass.In the given question it has…
Q: In the reaction below, identify (in order left to right) the oxidizing agent and the substance being…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: How many sigma bonds and how many pi bonds are represented inthis structure? C3HNC1₂
A: The covalent bond is formed by the overlap of the atomic orbitals between the atoms of similar…
Q: Write the product for 3 out of 4 of the following reactions. гар Н30 ада оченно OH H", heat N Ди H2O…
A: To give the products of the given reactions, the following types of reactions are usedHydrolysis of…
Q: After a substance acting as a strong base reacts, what remains of the base? Select one: a. a weaker…
A:
Q: b. Perform each calculation to the correct number of significant figures i 89.3 x 77.0 x 0.08 ii.…
A: Dimensional analysis is a way to check the correctness of equations and to convert from one…
Q: ||| = Initial Knowledge Check g A chemistry student needs 45.0 mL of acetone for an experiment. By…
A: Note: 1 mL = 1 cm3volume of acetone = 45.0 mL = 45.0 cm3density of acetone = 0.790 g.cm-3mass of…
Q: What is ester linkage
A: The ester linkage is generally observed in organic molecules. The term can be explained as;
Q: The standard EMF for the cell using the overall cell reaction below is +0.84 V: 2Al(s) + 312 (s)-…
A:
Q: C) A) OH B(OH)2 HO off Pd-cat. Base D) B) SOCI₂ OCH 3 Lo ? CI
A:
Q: What are the hybridization of carbons 1 and 2 respectively in the following molecule?
A:
Q: Question 7 Using curved arrows, propose how each of the following compounds can be formed. CH3 F (a)…
A: The given compound is cyclohalogen.In the reagent first given condition is heating, so there must be…
Q: 3. Calculate the number of moles of hydrofluoric acid, HF (K. = 6.8 x 104) that must be added to 500…
A: Given:Volume of 0.30 M sodium fluoride = 500 cm3 = 500 mL ()Ka of HF = 6.8 x 10-4 pH of the…
Q: Multiply or divide the following measurements. Be sure each answer you enter contains the correct…
A: Rules for determining the number of significant figures in an answer :The result of multiplication…
Q: Indicate the mechanism of the reaction of cyclopentanone and cyclopentylamine.
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Give detailed mechanism Solution with explanation needed
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- What are the products and reactants of the following: a) Isobutylmagnesium bromide + H2O -> Ab) sec-butyl bromide + Li/CuI -> B c) B + CH3CH2Br -> CPredict the major products, and if reaction is not feasible then write NR.Which of the following is the correct step of protonation of tran-2-butene? A B C D
- predict the products: XCL2+ E3PO4 --->predict the stating materials, reagents, or products of the followinf reactionsArrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H₂SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene A 1.2.3.4.5 (B) 5.1,243 5.1.4.2.3 D 5.2.1.4.3
- Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d belowOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Suggest an explanation for the fact that the order of reactivity of the halides toward n-butylbrosylate in acetone is Cl” > Br” > I~ when (C4H9)4N* is the cation of the halide salt butI” > Br” > Cl” when Li’ is the cation35.) What would be the preferred product for each of the following reactions ( hint : ether formation , ether-cleavage, diene formation , 1,2/1,4-addition, epoxidation and Diels-Alder cycloaddition?An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a refrigerator, and left for aparty. When he returned from the party, he discovered that the refrigerator was broken and that it had gotten quite warminside. Although all the THF had evaporated from the flask, he treated the residue with basic hydrogen peroxide. Tohis surprise, he recovered a fair yield of decan-1-ol. Use a mechanism to show how this reaction might have occurred.(Hint: The addition of BH3 is reversible.)

