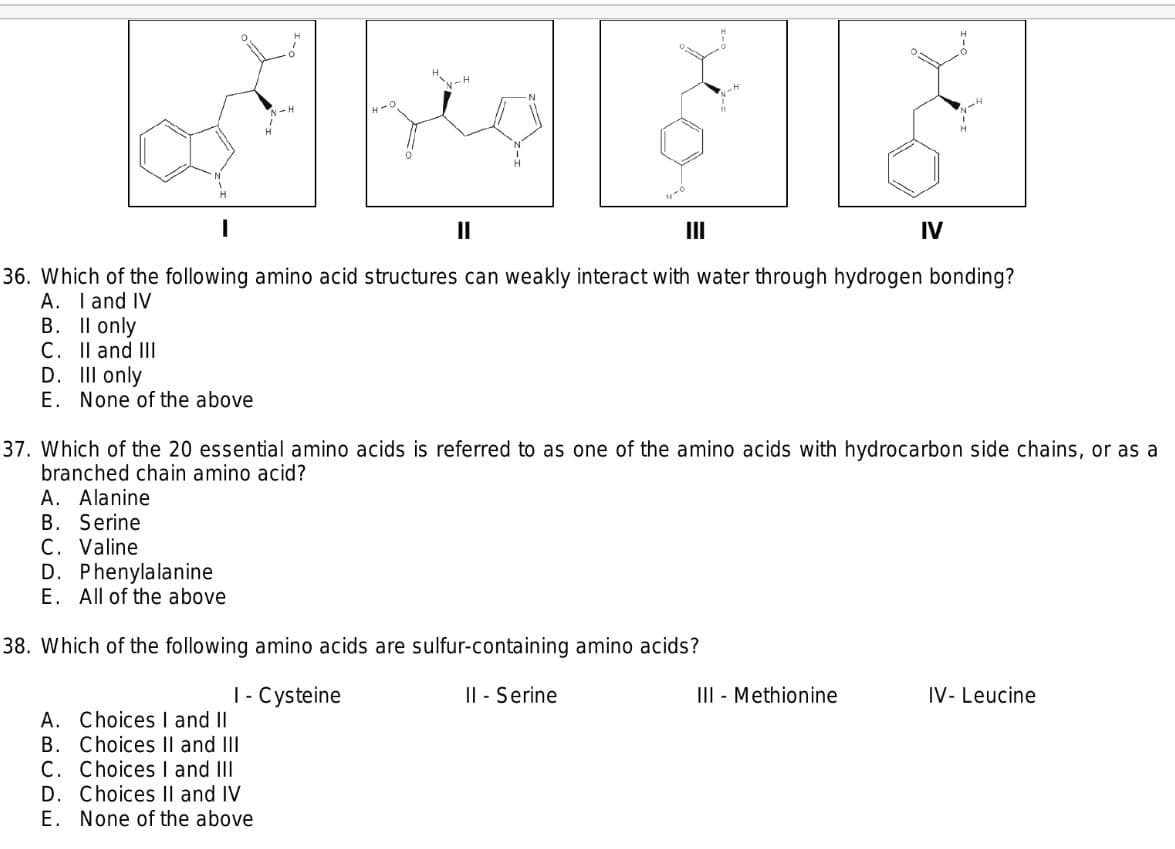
36. Which of the following amino acid structures can weakly interact with water through hydrogen bonding? A. I and IV B. Il only C. Il and III D. III only E. None of the above
36. Which of the following amino acid structures can weakly interact with water through hydrogen bonding? A. I and IV B. Il only C. Il and III D. III only E. None of the above
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter3: The Chemistry Of Life: Organic Compounds
Section3.1: Carbon Atoms And Organic Molecules
Problem 3C
Related questions
Question
11
Transcribed Image Text:N-H
II
II
IV
36. Which of the following amino acid structures can weakly interact with water through hydrogen bonding?
A. I and IV
B. Il only
C. Il and III
D. III only
E. None of the above
37. Which of the 20 essential amino acids is referred to as one of the amino acids with hydrocarbon side chains, or as a
branched chain amino acid?
A. Alanine
B. Serine
C. Valine
D. Phenylalanine
E. All of the above
38. Which of the following amino acids are sulfur-containing amino acids?
|- Cysteine
I| - Serine
III - Methionine
IV- Leucine
A. Choices I and II
B. Choices Il and III
C. Choices I and III
D. Choices Il and IV
E. None of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning