Q: 1. Fill in the missing products, reactants, and/or reagents. OH m-CPBA HBr EtONa, EtOH all products…
A: The objective of the question is to predict the reactant, product or reagents and fill in the…
Q: Q2/A solution with a volume of (250mL) contains (1.4g) of pure anhydrous sodium carbonate. If you…
A: The question is based on the concept of molarity.molarity is a method of expressing concentration of…
Q: 38) 8 d cl Start with benzene and Succinyl chloride as reactants of this transformation
A: The objective of this question is to complete the reaction.
Q: 33) a) Predict the major Le You ord, oor c.) Br product Bre Fe word al) AO reviction
A: The required answer is given belowExplanation:Step 1: Step 2: Step 3: Step 4:
Q: a) c) predict the major product Alcis - CH₂CH(CH3)₂ -CH3 b) d) cH3 CHCH₂CH3 -CH ₂ CH ₂ CH ₂ CH 3.
A: In first step of friedel craft reaction carbocation intermediate are form
Q: How many different molecules are drawn below? 0 X S
A: Identical molecules :- on the basis of the number of atoms present in molecules, those molecules…
Q: The major product of the following reaction sequence is: 1. B₂H6 2. H₂O2, HO a) b) ОН OH
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: a) 1) Brz, Fe 2)" Aldz ان. ود و به انسان الارت 2) Br₂, Fe 3) Zn (Hg), Hel, heat رط الا لط ان سن مدل…
A: The objective of this question is to find which reagents are used to convert benzene to .
Q: Complete the reaction below. 1) What are the names of the starting compound and product? 2) Draw out…
A: The given reaction is an acid catalysed lactone formation reaction from a hydroxy carboxylic acid.…
Q: Choose the correct structure for the major product of the following: or ox or or HO NO₂ NO₂ A HNO3…
A: Information about the question
Q: Benzodiazepines are known to have a wide therapeutic window. What makes benzodiazepines relatively…
A: The objective of the question is to understand why benzodiazepines are considered relatively safe…
Q: Consider this reaction: +H,O(aq) At a certain temperature it obeys this rate law. -¹) [NH₂OH]² NH,OH…
A: Consider the given reaction,Rate law of the above reaction,The power of the concentration of…
Q: Calculate the percentage by mass of the indicated element in the following compounds: (a) carbon in…
A: molar mass of CS(NH2)2 = 76.12 g/molmolar mass of C18H27NO3 = 305.41 g/molmolar mass of C2H6 = 30.07…
Q: 24) predict the major final product X CH3CH₂C Alcs H₂ 504 a) ******* c) "OH ex ass KMn Dy / NaoH /…
A: Find out major product of reactionStep 1& 2 is F. C. AlkylationStep3 is oxidation
Q: Answer the questions below about the highlighted atom in this Lewis structure: H I HCC=C-H 1 H In…
A: • Sigma bonds are formed by head to head overlapping of atomic orbitals.All single bonds are sigma…
Q: The structure of citric acid is shown. Click on the three most acidic protons in citric acid. HOH O…
A: The structure of citric acid given is
Q: You wish to prepare 250.0 mL of a 425.0 ppm w/v sodium (MW= 22.9897 g/mol) solution. How many grams…
A: Given:Volume of solution = 250 mLw/v concentration of Na = 425.0 ppmMolar mass of Na = 22.9897…
Q: Consider the following equilibria: 2A(g) 2B(g) + C(g) A(g) +E(g) B(g) + D(g) K1 K2 Calculate the…
A: If we reverse the chemical reactions ,the equilibrium constant of reverse reaction is inverse of…
Q: 100 mL of water containing 100 mg/L toluene is placed in a 150 mL (completely full, no headspace)…
A: Initial volume of water (Vw) = 100 mL = 0.1 LInitial concentration of toluene in water (Cw initial)…
Q: Choose all species that are Lewis acids in water. It may help to draw each Lewis structure.…
A: Lewis Acids are chemical species that have empty orbitals and can accept electron pairs from Lewis…
Q: Check the box under each molecule that has a total of two ß hydrogens. If none of the molecules fit…
A: In organic chemistry, a hydrogen refers to a hydrogen atom that is directly attached to the carbon…
Q: The leaves of the rhubarb plant contain high concentrations of diprotic oxalic acid (HOOCCOOH) and…
A: Answer:Value of pH is always equal to the negative of logarithm of hydrogen ion concentration, so,
Q: Question 21 Using the angular overlap model (AOM), determine the energies of the d orbitals of the…
A: The objective of the question is to determine the energies of the d orbitals of the metal for…
Q: For the compound below please identify the position of equilibrium (which chair conformation is more…
A: The cyclohexane molecule generally exists in chair form to minimize the angle strain. The general…
Q: Liquid sodium is being considered as an engine coolant. How many grams of liquid sodium (minimum)…
A:
Q: Calculate the [H+] and pH of a 3.31 x 10 M butanoic acid solution. The K₂ of butanoic acid is 1.52 ×…
A: Given that,The concentration of butanoic acid,The dissociation constant of butanoic acid,The…
Q: For the reaction 2A(g)+3B(g) → C(g) + 3D(g) the following data was collected at constant…
A: The objective of the question is to determine the rate law of the given reaction.
Q: Eight patients are treated for hypoparathyroidism using a drug therapy. Before the treatment, the…
A: The objective of the question is to determine if there was a significant increase in serum calcium…
Q: A 340.00 mL solution of 0.00175 M AB2 is added to a 140.00 mL solution of 0.00235 M CD4. What is…
A: Answer:For any sparingly soluble compound, value of pQsp is equal to the negative of logarithm of…
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A: The objective of the question is to calculate the equilibrium pressure.
Q: Give correct typing answer with explanation
A: Compound have higher melting point than alcohal. Explanation:Given compound is Symmetrical They have…
Q: PQ-29. What is the major product of this reaction? ОН (B) ОН ОН 1) excess CH3Li 2) H30+ (D) H
A: Givena carboxylic acid and reagent CH3Li.Determine the product.
Q: Draw the most and least stable Newman projections for the following molecules ( focus on C2 and C3).…
A:
Q: Given the AH values of the two allotropes, 12 (s) and I (g), determine which is more stable at this…
A: Given two allotropes as I2 (s) and I(g)The enthalpy of formation of both of them is 0 kJ/mol and 107…
Q: Nitrogen monoxide and hydrogen react to form nitrogen and water, like this: 2 NO(g) + 2 H₂(g) →…
A: The equilibrium constant Kp is defined as the ratio of partial pressure of products divided by the…
Q: Describe a-dislocations, c-dislocations and c+a dislocations, which may be possible in HCP crystals.…
A: In the given question we have to describe the -dislocations, c-dislocations and dislocations which…
Q: NaBH4 PQ-18. In which reaction would the addition of the nucleophile be reversible? CH3OH (A) (B) 0…
A: Nucleophilic addition reaction mechanism:In step-1 nucleophile Y- attack to electrophilic carbon…
Q: Depending on the size and complexity of the molecule, small chemical alterations can impart…
A: A compound that can mimic the function of acetyl choline or butyrl choline is called a cholinergic…
Q: 6. Complete this reaction NBS w/ hu ts):
A: Given that, the reaction is:
Q: H This molecule:
A: Given is organic compound. Diastereomers are isomers that are not mirror images of each other.…
Q: O ketone Oester O carboxylic acid O aldehyde O alcohol O nitrile (i.e., RCN) None of these other…
A: We are given at IR spectroscopy data, we have to identify the compound's functional group from…
Q: 4) a) Attempting to alkylate t-Butylchloride with the enolate of acetone gives an undesired product.…
A:
Q: 3) Complete the following: Stereochemistry and regiochemistry may be important. OSO4 THF a) b) c) d)…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The C-13 NMR shown below represents. 200 a benzil b benzoin 180 160 140 120 100 PPM 80 60 40 20
A: In 13C - NMR spectra, equivalent carbon atoms give same peak.Equivalent carbon atoms are the carbon…
Q: Draw the product structure and label each unique hydrogen atom with a letter. Label the NMR spectral…
A: Isopentyl acetate, also known as isoamyl acetate, has the chemical formula C7H14O2 and an average…
Q: 2. Assuming that the polymerization reactions are feasible, write repeating units for the polymers…
A: A polymer is defined as a high molecular weight substance that is made up of a repetition of small…
Q: Give the major product Хон H2S04
A: Given is organic reaction.The given starting compound is benzene.The given reagents is 1,4-diol.The…
Q: 2. Draw the most stable conjugate base and most stable conjugate acids for the following compounds.…
A: The objective is to determine the most stable conjugate base and conjugate acid of the given…
Q: PQ-3. What is the major product of this reaction? مو (A) CN H (B) CN (C) CN CN H (D) HCN OH CN
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: H₂O Br
A: The reaction of alkyl bromide with water is an example of solvolysis and a SN1 reaction.
Suggest an efficient synthesis for the porblem in the image, Show reagents and products of each step. Ensure that the reagents are indicated on the arrows pointing to the respective product. Please only utilize reactions that are part of the
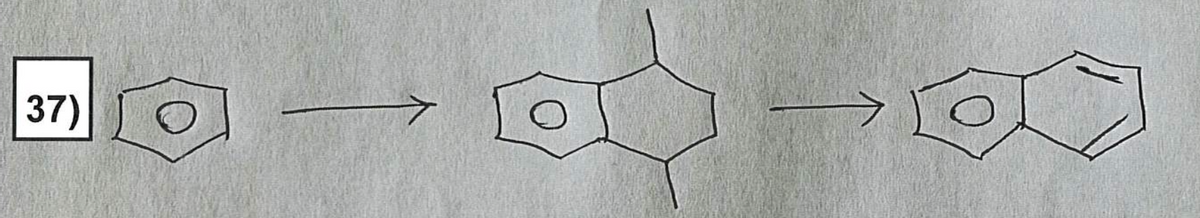
Step by step
Solved in 3 steps with 1 images


