3A+4B-----2C is found to be second order with respect to A and the rate constant for the reaction is k = 1.56* 10^ {-5}k=1.56∗10−5 \frac{L}{mole\cdot\sec}mole⋅secL. If the initial concentration of A is 0.86 M0.86M how much many seconds will be required for the concentration of A to reach 0.46 M0.46M you can input your answer as a whole number
3A+4B-----2C is found to be second order with respect to A and the rate constant for the reaction is k = 1.56* 10^ {-5}k=1.56∗10−5 \frac{L}{mole\cdot\sec}mole⋅secL. If the initial concentration of A is 0.86 M0.86M how much many seconds will be required for the concentration of A to reach 0.46 M0.46M you can input your answer as a whole number
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 87QAP: A drug decomposes in the blood by a first-order process. A pill containing 0.500 g of the active...
Related questions
Question
100%
3A+4B-----2C
is found to be second order with respect to A and the rate constant for the reaction is k = 1.56* 10^ {-5}k=1.56∗10−5 \frac{L}{mole\cdot\sec}mole⋅secL. If the initial concentration of A is 0.86 M0.86M how much many seconds will be required for the concentration of A to reach 0.46 M0.46M
you can input your answer as a whole number
Expert Solution
Step 1
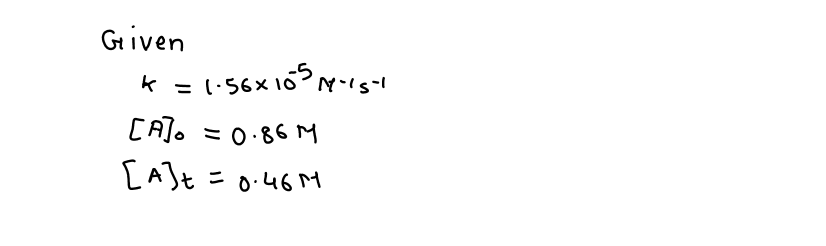
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
