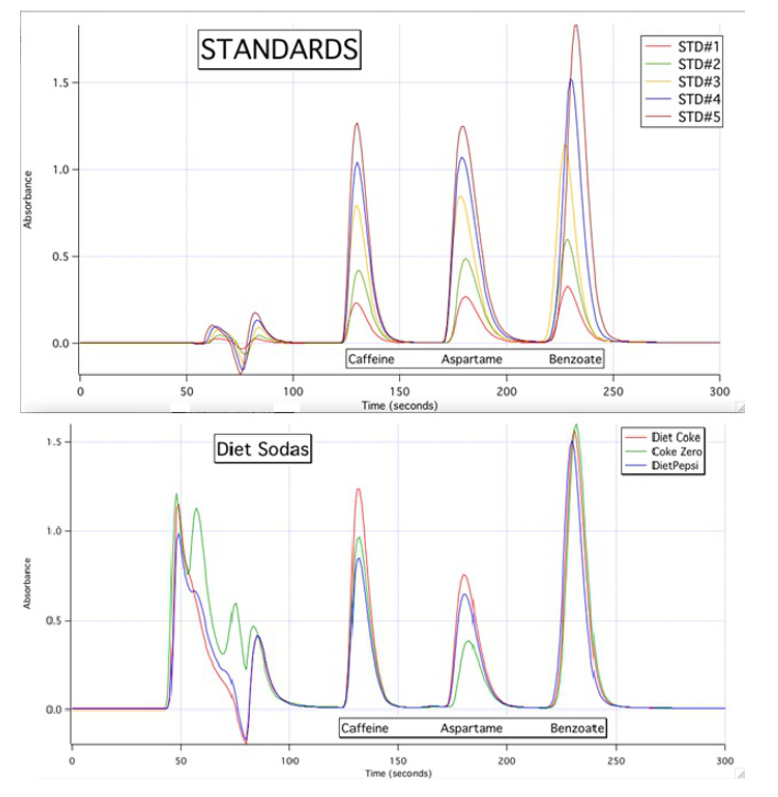
4) If the classification of the chromatography run is a reverse-phase chromatography, discuss the relative polarities of caffeine, aspartame and benzoate
Q: What effect does a decrease in pH have on the following equilibrium? CaCO3(s) + 2H+¹(a ¹(aq) →→→…
A:
Q: What is the theoretical oxygen demand in liters of air for a 50 mg/L solution of acetone, C3H6O to…
A:
Q: How would you make 1L of 0.5M total strength phosphate buffer (pKas are 2.1, 7.2, 12.7) at pH = 8…
A:
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2VO (aq) +…
A: Given, 2VO2+(aq) + 4H+(aq) + Fe(s) --------> 2VO2+(aq) + 2H2O(l) + Fe2+(aq) [VO2+] = 1.50 M…
Q: can you provide more explanation on impacts of SULFUR MANAGEMENT : explain the points below:…
A: Sulfur management
Q: Milk of Magnesia, a suspension of Mg(OH)2, a laxative, can be used as treatment for constipation…
A: We will find calculate solubility i.e. concentration of Mg(OH)2 in suspension, then find pH qnd…
Q: The cocoa bean has a [H3O¹] = 1.26x105 mol/L. What is the -POH? note: write the answer in 2…
A:
Q: SYNTHESIS! Devise a multistep synthesis of the target molecules from the given starting compound.…
A: Bromobenzene is converted into phenylmagnesiumbromide (grignard reagent) further it is treated…
Q: Predict whether aqueous solutions of the following compounds are acidic, basic, or neutral. compound…
A: The nature of aqueous solution of given salts depends on nature of acid and base given .
Q: NiCl, is a strong electrolyte. Determine the concentration of each of the individual ions in a 0.700…
A:
Q: 2. Explain the principle of ion exchange adsorption.
A: The adsorption results in the formation of a molecular layer of the adsorbate on the surface.…
Q: 1.Organic chemistry refer to OA. Study carbon compounds. O B. Study carbon and hydrogen compounds C.…
A: Hydrocarbon are the compound which contains hydrogen and carbon atoms.
Q: How many chiral centers are in the following molecule? 03 02 5 04 OH
A: We have given the organic compound we have to find how many chiral centre present in the given…
Q: "The IR absorption due to the stretn Stretching of which of these causon- hydrogen bonds occue at…
A:
Q: How many atoms of O there in 86.2 grams of cadmium (II) nitrite?
A: Here, we have to calculate the number of atoms of O there in 86.2 grams of cadmium (II) nitrite.
Q: identify the name of the compounds and it's functional group
A: here we are required to identify the name of the compounds and it's functional group
Q: Baking powder or sodium bicarbonate (NaHCO3), is used in baking to make cakes rise because the…
A: Baking powder is a leaving agent that is mainly used in the production of cakes, cookies, muffins,…
Q: Zn + Na2CrO4 + NaOH — NaZnO2 + NaCrO2 + H2O balance The redox reaction and include the…
A: Answer - According to the question - Given reaction - Zn + Na2CrO4 + NaOH — NaZnO2 + NaCrO2 + H2O…
Q: Assign Z and E configurations to these compounds. O HO HO O CI
A: Assign Z or E configuration of the following---
Q: When the concentration of reactant molecules is increased, the rate of reaction increases. TRUE OR…
A: The rate of the reaction is the amount of reactant reacted in given amount of time.
Q: If 4.90 mol of C5H₁2 reacts with excess O₂, how many moles of CO₂ will be produced by the following…
A: O2 is in excess so C5H12 will be the limiting reagent.The product will formed according to C5H12.
Q: The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown…
A:
Q: In water, HNO3 (aq) behaves as a: a) strong base b) weak acid c) amphoteric species d) strong…
A:
Q: Value of (5) is equal ат JP/V ат aP/V ӘР ( ат у ӘР as V to....
A:
Q: Question 1 Part A: Arrange the following metric units of weight order from smallest to largest.…
A:
Q: gy E required for deformation can be calculated using which formula? (L = length of d
A: According to the question, IN A CRYSTAL, ENERGY 'E' REQUIRED FOR DEFORMATION CAN BE CALCULATED USING…
Q: (Hand by written ans.)1. A buffer solution was prepared by dissolving 0.15 mol of sodium ethanoate…
A: here we are required to find the change in pH when 1.0 ml of 1.0 mol /L hydrochloric acid is added…
Q: Convert the structure below to a skeletal drawing. н н н н H-C H-c-o-C-H H-C C-H н н C-H /\ Н Н
A: The skeletal structure of the organic compound is representation in bond line form where only carbon…
Q: An acid increases the concentration of H+ in aqueous solution, and a base increases the…
A:
Q: Solve the water solubility of lead (II) iodide in 0.15M of potassium iodide. The Ksp is 8.4x10⁻⁹
A: Given, The molar solubility of lead (II) iodide in 0.15M of potassium iodide is:…
Q: Use partial orbital diagrams and Lewis symbols to depict the formation of the following ions from…
A:
Q: Consider the equilibrium: C(s) + CO2(g) ⇄ 2CO(g) When this system is at equilibrium at 700°C in a…
A:
Q: Part 2: 33g Na₂SO4 Step 1: mass to moles Molar mass of Na₂SO4 = 33g Na₂SO4 + molar mass of Na₂SO4 =…
A: Given: Mass of Na2SO4 = 33 g Known: Molar mass of Na = 23 g/mol Molar mass of S = 32 g/mol Molar…
Q: The diagram (Figure 1) represents a high-temperature reaction between CH4 and H₂O. Figure i Part A…
A: Here as 2 moles CH4 reacts with 2-Moles H2O so there is no excess reagent is present,this we can…
Q: -(NH4)2SO4 +-Pb(NO3)2 → PbSO4 + NH4 NO3 - 1
A:
Q: HC₂H3O2(aq) + + Ba(OH)₂(aq) →>>>
A: Given, HC2H3O2(aq) + Ba(OH)2(aq) -------->
Q: how many grams of magnesium
A:
Q: In Part B the given conditions were 1.00 mol of argon in a 0.500-L container at 26.0 °C. You…
A: The percent difference between ideal and real gas will be = [(49.1- 47.1atm)/ 49.1atm] x 100% =…
Q: catalyst: actually participates in the reaction. TRUE OR FALSE?
A: Catalyst takes part in Chemical reaction and increase the rate of reaction by decreasing activation…
Q: A chemist designs a galvanic cell that uses these two half-reactions: half-reaction 21O3(aq)+12 H+…
A:
Q: The aluminum in a 1.520-g sample of impure ammonium aluminum sulfate was precipitated with aqueous…
A: Given data : Mass of sample = 1.520 g Mass of Al2O3 = 0.2001 g
Q: the
A:
Q: Choose the word from the word bank below that best matches each statement. Constant Equilibrium…
A: Fill in the blanks.
Q: What would be the [H3O+]for a soda found to have a [OH−]of 1.26 × 10−13 mol/L? Is the solution…
A: Given, [OH-] = 1.26 × 10−13 mol/L Using, Kw = [H3O+][OH-] At 298K, Kw = 1.0×10-14
Q: a) Compare between equivalence point and endpoint. Distinguish which point comes first.
A:
Q: 4. Determine which of the following are mixtures. Water dissolved in alcohol Water dissolved in…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: What is the full name and 3-letter abbreviation of this peptide? b. Give the different forms…
A:
Q: For the reaction below, which change would cause the equilibrium to shift to the right? Tick all…
A: Le Chatelier's principle explains when the equilibrium is disturbed by external agent, then the…
Q: An aqueous solution has hydronium concentration [H3O+] = 6.3 x 10-3 mol L-1. What is the pH of the…
A:
Q: What would be the [H3O+] for a soda found to have a [OH-] of 1.26 x 10-¹3 mol/L? Is the solution…
A: Given concentration of OH- ion, [OH-] =1.26×10-13 moles/L concentration of H3O+ ion, [H3O+] =…
4) If the classification of the chromatography run is a reverse-phase chromatography, discuss the relative polarities of caffeine, aspartame and benzoate
Step by step
Solved in 2 steps with 2 images

- A solution of tryptophan has an absorbance of 0.64 at 280 nm. Given with of 6.04 x 103 Msolution of tryptophan and path length of 0.5 cm. What is the molar absorptivity oftryptophan?Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72As part of an investigation on Pb in biological ecosystems, some grasses that grew along the road and were exposed to emissions from motor gasoline were collected. 6.2501 g of grass were calcined to destroy all organic matter, the inorganic residue was properly treated and finally made up to 100 mL. An aliquot of 50 mL of this solution was read in an atomic absorption spectrophotometer registering an absorbance of 0.1250; then 0.10 mL of a standard solution of 55 µg of Pb / mL were added to the other 50 mL, with this fortified sample, another absorbance measurement was made, which was 0.1798. Calculate the µg of Pb / g of grass
- Attatched is UV-Vis Spectrum (Isopropanol, c= 1.65 * 10-4 M) Include the UV-Vis data in a table with wavelength, absorbance. and molar extinction coefficient Wavelength Absorbance Molar Extinction Coefficient 300-700 nmThe absorbance values at 250nm of 5 standard solutions, and sample solution of a drug are given below: Conc. (ug/ml) A 250 nm10 0.16820. 0.32930 0.50840. 0.66050 0.846Sample. 0.661Calculate the concentration of the sampleCalculate the concentration of a colored solution if the molar absorptivity is 39.80 M-1•cm-1 and the absorbance is 0.777. The path length of the sample is 1.00 cm.
- A calibration plot of absorbance vs concentration (ppm) was obatined with standard known Red 40 dye solution. - the slope of the best-fit straight line of the plot is 0.057 ppm^-1 - The absorbance of the dilute unknown sports drink was 0.88 What is the concentration of this dilute unknown sports drink?______ the answer is 15.4 ppm ( can you please show me how to calculate this problem)Sally obtains a standard calibration curve for their assigned food dye by plotting absorbance versus concentration (in M) of known solutions and finds the following slope and intercept: y = 1.646x + 0.026 Note: Copying and/or posting this or other questions without the express written permission from Dr. Burke and the Department of Chemistry & Biochemistry at the University of Delaware is a violation of intellectual property copyright law. Sally finds that her original Kool-aid sample is too concentrated, so they dilute it by transferring 13.00 mL of the original Kool-aid sample into a new container and diluting to a total volume of 37.00 mL. If the absorbance of the dilute Kool-aid sample at the appropriate wavelength for their assigned dye is 0.950, determine the concentration (in M) of the assigned food dye in the original Kool-aid sample. Report your answer with three places after the decimal.Compound X was dissolved in a 5-mL volumetric flask. A 1.00-mL aliquot of the compound was placed in a 10-mL volumetric flask and diluted to the mark. The solution was read in a UV-Vis spectrophotometer and an absorbance of 0.427 in a 1.000-cm cuvette was observed at 340 nm. Solve the following question. Data for Compound X:Molecular mass = 292.16 g/molMolar absorptivity (ɛ) at 340 nm= 6130 M -1 cm -1 a. Calculate the concentration of compound X in the cuvette. b. How many milligrams of compound X were used to make the 5-mL solution?
- Compound X was dissolved in a 5-mL volumetric flask. A 1.00-mL aliquot of the compound was placed in a 10-mL volumetric flask and diluted to the mark. The solution was read in a UV-Vis spectrophotometer and an absorbance of 0.427 in a 1.000-cm cuvette was observed at 340 nm. Solve the following question. Data for Compound X:Molecular mass = 292.16 g/molMolar absorptivity (ɛ) at 340 nm= 6130 M -1 cm -1 1. What was the concentration of compound X in the 5-mL flask?Calculate the concentration of a solution of peptide AWSME, in mM if the absorbance at 280 nm for the solution is 0.6 absorbance units, and a 0.2-cm cuvette is used. For the extinction coefficient, take into account that e280(Trp) = 5690 M-1 cm-1 and e280(Tyr) = 1280 M-1 cm-1. Round your answer to two decimals.A solution of Tryptophan has an absorbance at 280 nm of 0.54 in a 0.5 cm length cuvette. Given the absorbance coefficient of trp is 6.4 x 103 Lmol-1cm-1. What is the concentration of solution? The options are as follows A) 0.17 x10-3 mol/L B)2.56x10-3mol/L C)0.17x10+3mol/L D)2.56x10+3mol/L

