Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
Solve as many as you can please
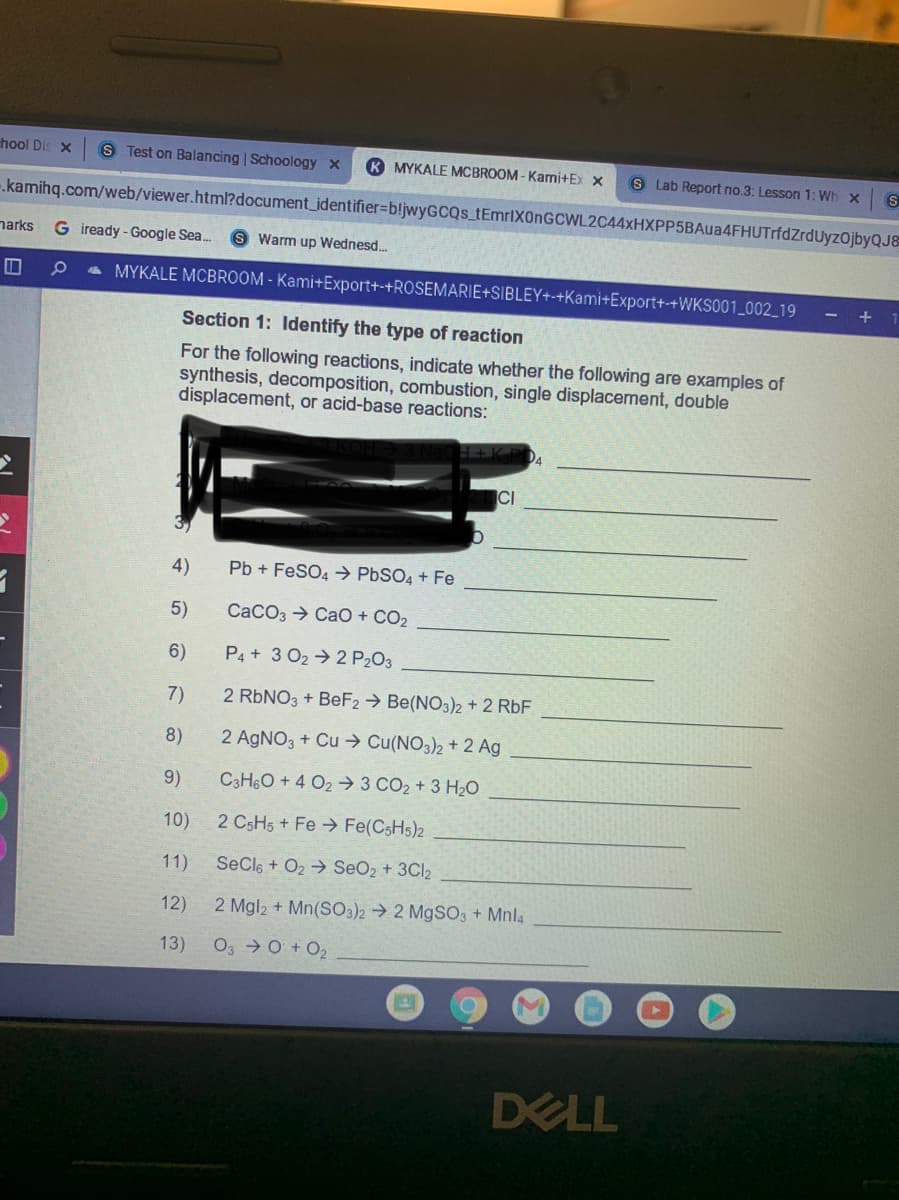
Transcribed Image Text:hool Dis X
S Test on Balancing | Schoology x
K MYKALE MCBROOM - Kami+Ex x
S Lab Report no.3: Lesson 1: Wh x
.kamihq.com/web/viewer.html?document_identifier%3Db!jwyGCQs_tEmrlXOnGCWL2C44xHXPP5BAua4FHUTrfdZrdUyzOjbyQJ&
narks
G iready - Google Sea.
S Warm up Wednesd..
- MYKALE MCBROOM - Kami+Export+-+ROSEMARIE+SIBLEY+-+Kami+Export+-+WKS001_002_19
Section 1: Identify the type of reaction
For the following reactions, indicate whether the following are examples of
synthesis, decomposition, combustion, single displacement, double
displacement, or acid-base reactions:
CI
4)
Pb + FeSO4 → PBSO4 + Fe
5)
CaCO3 → CaO + CO2
6)
P4 + 3 02→2 P203
7)
2 RBNO3 + BeF2 → Be(NO3)2 + 2 RbF
8)
2 AGNO3 + Cu → Cu(NO3)2 + 2 Ag
9)
C3H6O + 4 O2 3 CO2 + 3 H20
10)
2 CSH5 + Fe Fe(CSH5)2
11)
SeCle + O2 → SeO2 + 3CI2
12)
2 Mgl2 + Mn(SO3)2 → 2 M9SO3 + Mnla
13)
O3 0 + O2
DELL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

